Abstract
Objectives
The aim of this study was to identify clinical, magnetic resonance imaging (MRI) and biological markers predictive of long–term clinical response to interferon beta (IFN beta) therapy in patients with relapsing–remitting multiple sclerosis (RRMS).
Methods
Sixty–eight patients treated with IFN beta were followed over a 6–year period. Relapse rate and disability progression were evaluated throughout the study. We considered suboptimal clinical response to be either the presence of sustained disability progression, or more than two relapses. Baseline and 12–month demographic, clinical and MRI findings, as well as the development of neutralizing antibodies (NAbs) against IFN beta during the first year of therapy were analyzed as predictors of long–term clinical outcome.
Results
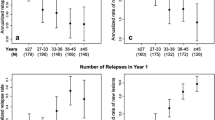
"Black holes" on MRI were the best baseline predictor of disability progression (odds ratio [OR] 6.8; p < 0.001).At 1 year, both male gender (OR 4.9; p = 0.009) and NAbs (OR 7.3; p = 0.003) were independently associated with a high risk of developing subsequent disability. The presence of gadolinium enhancement, both at baseline (OR 4.7; p = 0.005) and on the 1–year MRI scan (OR 7.9; p = 0.002), was the unique variable associated with the number of relapses over the study period.
Conclusions
Variables assessable within the first year of treatment significantly influence relapse rate and disability progression in patients with RRMS treated with IFN beta. These findings may help clinicians to make decisions regarding therapy regimen over time, and highlight the need for a prognostic algorithm.
Similar content being viewed by others
References
Confavreux C, Aimard G, Devic M (1980) Course and prognosis of multiple sclerosis assessed by the computerized data processing of 349 patients. Brain 103:281–300
Confavreux C, Vukusic S, Adeleine P (2003) Early clinical predictors and progression of irreversible disability in multiple sclerosis: an amnesic process. Brain 126:770–782
Weinshenker BG, Bass B, Rice GP, et al. (1989) The natural history of multiple sclerosis: a geographically based study. I. Clinical course and disability. Brain 112:133–146
Weinshenker BG, Bass B, Rice GP, et al. (1989) The natural history of multiple sclerosis: a geographically based study. II. Predictive value of the early clinical course. Brain 112:1419–1428
Runmarker B, Andersen O (1993) Prognostic factors in a multiple sclerosis incidence cohort with twenty-five years of follow-up. Brain 116:117–134
Trojano M, Avolio C, Manzari C, et al. (1995) Multivariate analysis of predictive factors of multiple sclerosis course with a validated method to assess clinical events. J Neurol Neurosurg Psychiatry 58:300–306
Kantarci O, Siva A, Eraksoy M, et al. (1998) Survival and predictors of disability in Turkish MS patients. Turkish Multiple Sclerosis Study Group (TUMSSG). Neurology 51:765–772
Filippi M, Horsfield MA, Morrissey SP, et al. (1994) Quantitative brain MRI lesion load predicts the course of clinically isolated syndromes suggestive of multiple sclerosis. Neurology 44:635–641
O’Riordan JI, Thompson AJ, Kingsley DP, et al. (1998) The prognostic value of brain MRI in clinically isolated syndromes of the CNS. A 10-year follow-up. Brain 121:495–503
Brex PA, Ciccarelli O, O’Riordan JI, Sailer M, Thompson AJ, Miller DH (2002) A longitudinal study of abnormalities on MRI and disability from multiple sclerosis. N Engl J Med 346:158–164
Barkhof F, Filippi M, Miller DH, et al. (1997) Comparison of MRI criteria at first presentation to predict conversion to clinically definite multiple sclerosis. Brain 120:2059–2069
Kappos L, Moeri D, Radue EW, et al. (1999) Predictive value of gadoliniumenhanced magnetic resonance imaging for relapse rate and changes in disability or impairment in multiple sclerosis: a meta-analysis.Gadolinium MRI Meta-analysis Group. Lancet 353:964–969
CHAMPS Study Group (2002) MRI predictors of early conversion to clinically definite MS in the CHAMPS placebo group. Neurology 59:998–1005
The IFN beta Study Group (1993) Interferon beta-1b is effective in relapsing- remitting multiple sclerosis. I. Clinical results of a multicenter, randomized, double-blind, placebo-controlled trial. The IFNB Multiple Sclerosis Study Group. Neurology 43:655–661
Jacobs LD, Cookfair DL, Rudick RA, et al. (1996) Intramuscular interferon beta-1a for disease progression in relapsing multiple sclerosis. The Multiple Sclerosis Collaborative Research Group (MSCRG). Ann Neurol 39:285–294
Randomised double-blind placebocontrolled study of interferon beta-1a in relapsing/remitting multiple sclerosis (1998) PRISMS (Prevention of Relapses and Disability by Interferon beta-1a Subcutaneously in Multiple Sclerosis) Study Group. Lancet 352:1498–1504
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Koudriavtseva T, Pozzilli C, Fiorelli M, et al. (1998) Determinants of Gdenhanced MRI response to IFN-beta- 1a treatment in relapsing-remitting multiple sclerosis. Mult Scler 4:403–407
Waubant E, Vukusic S, Gignoux L, et al. (2003) Clinical characteristics of responders to interferon therapy for relapsing MS. Neurology 61:184–189
Vermersch P, de Seze J, Stojkovic T; GSEP (Groupe Septentrional d’Etudes et de Recherches sur la SEP) (2002) Interferon beta 1a (Avonex) treatment in multiple sclerosis: similarity of effect on progression of disability in patients with mild and moderate disability. J Neurol 249:184–187
The IFNB Multiple Sclerosis study group and the University of British Columbia MS/MRI analysis group (1996) Neutralizing antibodies during treatment of multiple sclerosis with interferon beta-1b: experience during the first three years. Neurology 47: 889–894
Soelberg Sorensen P, Ross C, Clemmesen KM, et al. (2003) Clinical importance of neutralising antibodies against interferon beta in patients with relapsing-remitting multiple sclerosis. Lancet 362:1184–1191
Pozzilli C, Bastianello S, Koudriavtseva T, et al. (1996) Magnetic resonance imaging changes with recombinant human interferon-beta-1a: a short term study in relapsing-remitting multiple sclerosis. J Neurol Neurosurg Psychiatry 61:251–258
Paolillo A, Pozzilli C, Giugni E, et al. (2002) A 6-year clinical and MRI follow-up study of patients with relapsing- remitting multiple sclerosis treated with interferon-beta. Eur J Neurol 9:645–655
Bellomi F, Scagnolari C, Tomassini V, et al. (2003) Fate of neutralizing and binding antibodies to IFN beta in MS patients treated with IFN beta for 6 years. J Neurol Sci 215:3–8
WHO Expert Committee on Biological Standardisation (1985) Thirty-fifth report. WHO technical report series 725. Geneva: World Health Organization
Kawade Y (1986) Quantitation of neutralization of interferon by antibody. Methods Enzymol 119:558–573
Grossberg SE, Kawade Y, Kohase M, Klein JP (2001) The neutralization of interferon by antibody. II.Neutralizing antibody unitage and its relationship to bioassay sensitivity: the tenfold reduction unit. J Interferon Cytokine Res 21:743–755
Grimaud J, Lai M, Thorpe J, et al. (1996) Quantification of MRI lesion load in multiple sclerosis: a comparison of three computer-assisted techniques. Magn Reson Imaging 14:495–505
Miller DH, Grossman RI, Reingold SC, McFarland HF (1998) The role of magnetic resonance techniques in understanding and managing multiple sclerosis. Brain 121:3–24
Rio J, Nos C, Tintore M, et al. (2002) Assessment of different treatment failure criteria in a cohort of relapsingremitting multiple sclerosis patients treated with interferon beta: implications for clinical trials. Ann Neurol 52:400–406
Rudick RA, Fisher E, Lee JC, Duda JT, Simon J (2000) Brain atrophy in relapsing multiple sclerosis: relationship to relapses, EDSS, and treatment with interferon beta-1a. Mult Scler 6: 365–372
McFarland HF, Barkhof F, Antel J, Miller DH (2002) The role of MRI as surrogate outcome measure in multiple sclerosis. Mult Scler 8:40–51
Van Walderveen MA, Kamphorst W, Scheltens P, et al. (1998) Histopathologic correlate of hypointense lesions on T1-weighted spin-echo MRI in multiple sclerosis. Neurology 50:1282–1288
Van Waesberghe JH, Kamphorst W, De Groot CJ, et al. (1999) Axonal loss in multiple sclerosis lesions: magnetic resonance imaging insights into substrates of disability. Ann Neurol 46:747–754
Truyen L, Van Waesberghe JH, Van Walderveen MA, et al. (1996) Accumulation of hypointense lesions (“black holes”) on T1 spin-echo MRI correlates with disease progression in multiple sclerosis. Neurology 47:1469–1476
Simon JH, Lull J, Jacobs LD, et al. (2000) A longitudinal study of T1 hypointense lesions in relapsing MS: MSCRG trial of interferon beta-1a. Multiple Sclerosis Collaborative (Research Group). Neurology 55:185–192
Secondary Progressive Efficacy Clinical Trial of Recombinant Interferonbeta-1a in MS (SPECTRIMS) Study Group (2001) Randomized controlled trial of interferon beta-1a in secondary progressive MS: clinical results. Neurology 56:1496–1504
Li DK, Zhao GJ, Paty DW, University of British Columbia MS/MRI Analysis Research Group, The SPECTRIMS Study Group (2001) Randomized controlled trial of interferon-beta-1a in secondary progressive MS: MRI results. Neurology 56:1505–1513
De Cicco Nardone F, Rossiello F, Iacopino F, et al. (1996) Effects of interferon- beta on steroid receptors, prostaglandins and enzymatic activities in human endometrial cancer. Anticancer Res 16:161–169
Barak V, Kalickman I, Nisman B, et al. (1998) Changes in cytokine production of breast cancer patients treated with interferons. Cytokine 10:977–983
van Walderveen MA, Lycklama A, Nijeholt GJ, et al. (2001) Hypointense lesions on T1-weighted spin-echo magnetic resonance imaging: relation to clinical characteristics in subgroups of patients with multiple sclerosis. Arch Neurol 58(1):76–81
Pozzilli C, Tomassini V, Marinelli F, et al. (2003) “Gender gap” in multiple sclerosis: magnetic resonance imaging evidence. Eur J Neurol 10:95–97
Perini P, Calabrese M, Biasi G, Gallo P (2004) The clinical impact of interferon beta antibodies in relapsingremitting MS. J Neurol 251:305–309
Malucchi S, Sala A, Gilli F, et al. (2004) Neutralizing antibodies reduce the efficacy of beta IFN during treatment of multiple sclerosis. Neurology 62:2031–2037
Polman C, Kappos L, White R, et al. (2003) European Study Group in Interferon Beta-1b in Secondary Progressive MS.Neutralizing antibodies during treatment of secondary progressive MS with interferon beta-1b. Neurology 60:37–43
Molyneux PD, Filippi M, Barkhof F, et al. (1998) Correlations between monthly enhanced MRI lesion rate and changes in T2 lesion volume in multiple sclerosis. Ann Neurol 43:332–339
Rudick RA, Lee JC, Simon J, et al. (2004) Defining interferon beta response status in multiple sclerosis patients. Ann Neurol 56:548–555
Author information
Authors and Affiliations
Corresponding author
Additional information
The study was supported by Industria Farmaceutica Serono Italy.
Rights and permissions
About this article
Cite this article
Tomassini, V., Paolillo, A., Russo, P. et al. Predictors of long–term clinical response to interferon beta therapy in relapsing multiple sclerosis. J Neurol 253, 287–293 (2006). https://doi.org/10.1007/s00415-005-0979-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-005-0979-5