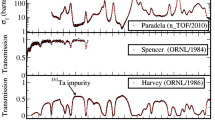
Abstract
Space radiation contains a complex mixture of particles comprised primarily of protons and high-energy heavy ions. Radiation risk is considered one of the major health risks for astronauts who embark on both orbital and interplanetary space missions. Ionizing radiation dose-dependently kills cells, damages genetic material, and disturbs cell differentiation and function. The immediate response to ionizing radiation-induced DNA damage is stimulation of DNA repair machinery and activation of cell cycle regulatory checkpoints. To date, little is known about cell cycle regulation after exposure to space-relevant radiation, especially regarding bone-forming osteoblasts. Here, we assessed cell cycle regulation in the osteoblastic cell line OCT-1 after exposure to various types of space-relevant radiation. The relative biological effectiveness (RBE) of ionizing radiation was investigated regarding the biological endpoint of cellular survival ability. Cell cycle progression was examined following radiation exposure resulting in different RBE values calculated for a cellular survival level of 1 %. Our findings indicate that radiation with a linear energy transfer (LET) of 150 keV/μm was most effective in inducing reproductive cell killing by causing cell cycle arrest. Expression analyses indicated that cells exposed to ionizing radiation exhibited significantly up-regulated p21(CDKN1A) gene expression. In conclusion, our findings suggest that cell cycle regulation is more sensitive to high-LET radiation than cell survival, which is not solely regulated through elevated CDKN1A expression.




Similar content being viewed by others
References
Alpen EL, Powers-Risius P, Curtis SB, DeGuzman R (1993) Tumorigenic potential of high-Z, high-LET charged-particle radiations. Radiat Res 136:382–391
Anastasov N, Hofig I, Vasconcellos IG, Rappl K, Braselmann H, Ludyga N, Auer G, Aubele M, Atkinson MJ (2012) Radiation resistance due to high expression of miR-21 and G2/M checkpoint arrest in breast cancer cells. Radiat Oncol 7:206
Anderson JA, Harper JV, Cucinotta FA, O’Neill P (2010) Participation of DNA-PKcs in DSB repair after exposure to high- and low-LET radiation. Radiat Res 174:195–205
Asaithamby A, Uematsu N, Chatterjee A, Story MD, Burma S, Chen DJ (2008) Repair of HZE-particle-induced DNA double-strand breaks in normal human fibroblasts. Radiat Res 169:437–446
Autsavapromporn N, Suzuki M, Plante I, Liu C, Uchihori Y, Hei TK, Azzam EI, Murakami T (2013) Participation of gap junction communication in potentially lethal damage repair and DNA damage in human fibroblasts exposed to low- or high-LET radiation. Mutat Res 756:78–85
Bandstra ER, Pecaut MJ, Anderson ER, Willey JS, De CF, Stock SR, Gridley DS, Nelson GA, Levine HG, Bateman TA (2008) Long-term dose response of trabecular bone in mice to proton radiation. Radiat Res 169:607–614
Baumstark-Khan C, Heilmann J, Rink H (2003) Induction and repair of DNA strand breaks in bovine lens epithelial cells after high LET irradiation. Adv Space Res 31:1583–1591
Belli M, Sapora O, Tabocchini MA (2002) Molecular targets in cellular response to ionizing radiation and implications in space radiation protection. J Radiat Res 43(Suppl):S13–S19
Blakely EA (2000) Biological effects of cosmic radiation: deterministic and stochastic. Health Phys 79:495–506
Chang PY, Bjornstad KA, Rosen CJ, McNamara MP, Mancini R, Goldstein LE, Chylack LT, Blakely EA (2005) Effects of iron ions, protons and X rays on human lens cell differentiation. Radiat Res 164:531–539
Chen DJ, Tsuboi K, Nguyen T, Yang TC (1994) Charged-particle mutagenesis II. Mutagenic effects of high energy charged particles in normal human fibroblasts. Adv Space Res 14:347–354
Chen D, Chen H, Feng JQ, Windle JJ et al (1995) Osteoblastic cell lines derived from a transgenic mouse containing the osteocalcin promotor driving SV40 T antigen. Molec Cell Differ 3:193–212
Chen H, Kolman K, Lanciloti N, Nerney M, Hays E, Robson C, Chandar N (2012) p53 and MDM2 are involved in the regulation of osteocalcin gene expression. Exp Cell Res 318:867–876
Cucinotta FA, Durante M (2006) Cancer risk from exposure to galactic cosmic rays: implications for space exploration by human beings. Lancet Oncol 7:431–435
Di LA, Linke SP, Clarkin K, Wahl GM (1994) DNA damage triggers a prolonged p53-dependent G1 arrest and long-term induction of Cip1 in normal human fibroblasts. Genes Dev 8:2540–2551
Durante M, Cucinotta FA (2008) Heavy ion carcinogenesis and human space exploration. Nat Rev Cancer 8:465–472
Durante M, Reitz G, Angerer O (2010) Space radiation research in Europe: flight experiments and ground-based studies. Radiat Environ Biophys 49:295–302
el-Deiry WS, Harper JW, O’Connor PM, Velculescu VE, Canman CE, Jackman J, Pietenpol JA, Burrell M, Hill DE, Wang Y (1994) WAF1/CIP1 is induced in p53-mediated G1 arrest and apoptosis. Cancer Res 54:1169–1174
Elkind MM, Sutton H (1960) Radiation response of mammalian cells grown in culture. 1. Repair of X-ray damage in surviving Chinese hamster cells. Radiat Res 13:556–593
Fournier C, Taucher-Scholz G (2004) Radiation induced cell cycle arrest: an overview of specific effects following high-LET exposure. Radiother Oncol 73(Suppl 2):S119–S122
Geyer RK, Nagasawa H, Little JB, Maki CG (2000) Role and regulation of p53 during an ultraviolet radiation-induced G1 cell cycle arrest. Cell Growth Differ 11:149–156
Gogineni VR, Nalla AK, Gupta R, Dinh DH, Klopfenstein JD, Rao JS (2011) Chk2-mediated G2/M cell cycle arrest maintains radiation resistance in malignant meningioma cells. Cancer Lett 313:64–75
Hada M, Georgakilas AG (2008) Formation of clustered DNA damage after high-LET irradiation: a review. J Radiat Res 49:203–210
Hada M, Sutherland BM (2006) Spectrum of complex DNA damages depends on the incident radiation. Radiat Res 165:223–230
Hall EJ, Marchese M, Hei TK, Zaider M (1988) Radiation response characteristics of human cells in vitro. Radiat Res 114:415–424
Hamilton SA, Pecaut MJ, Gridley DS, Travis ND, Bandstra ER, Willey JS, Nelson GA, Bateman TA (2006) A murine model for bone loss from therapeutic and space-relevant sources of radiation. J Appl Physiol 101:789–793
Hellweg CE, Baumstark-Khan C, Schmitz C, Lau P, Meier MM, Testard I, Berger T, Reitz G (2011) Carbon-ion-induced activation of the NF-kappaB pathway. Radiat Res 175:424–431
Houtgraaf JH, Versmissen J, van der Giessen WJ (2006) A concise review of DNA damage checkpoints and repair in mammalian cells. Cardiovasc Revasc Med 7:165–172
Hu Y, Lau P, Baumstark-Khan C, Hellweg CE, Reitz G (2012) X-ray induced alterations in the differentiation and mineralization potential of murine preosteoblastic cells. Adv Space Res 49:1422–1431
Jung YS, Qian Y, Chen X (2010) Examination of the expanding pathways for the regulation of p21 expression and activity. Cell Signal 22:1003–1012
Kronenberg A, Cucinotta FA (2012) Space radiation protection issues. Health Phys 103:556–567
Lang T, LeBlanc A, Evans H, Lu Y, Genant H, Yu A (2004) Cortical and trabecular bone mineral loss from the spine and hip in long-duration spaceflight. J Bone Miner Res 19:1006–1012
Lau P, Baumstark-Khan C, Hellweg CE, Reitz G (2010) X-irradiation-induced cell cycle delay and DNA double-strand breaks in the murine osteoblastic cell line OCT-1. Radiat Environ Biophys 49:271–280
Liu H, Li B (2010) p53 control of bone remodeling. J Cell Biochem 111:529–534
Lloyd SA, Bandstra ER, Travis ND, Nelson GA, Bourland JD, Pecaut MJ, Gridley DS, Willey JS, Bateman TA (2008) Spaceflight-relevant types of ionizing radiation and cortical bone: potential LET effect? Adv Space Res 42:1889–1897
Mason AJ, Giusti V, Green S, af Rosenschöld PM, Beynon TD, Hopewell JW (2011) Interaction between the biological effects of high- and low-LET radiation dose components in a mixed field exposure. Int J Radiat Biol 87:1162–1172
Mirzayans R, Andrais B, Scott A, Murray D (2012) New insights into p53 signaling and cancer cell response to DNA damage: implications for cancer therapy. J Biomed Biotechnol 2012:170325
Pawlik TM, Keyomarsi K (2004) Role of cell cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol Biol Phys 59:928–942
Pfaffl MW, Horgan GW, Dempfle L (2002) Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res 30:e36
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper–Excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515
Puck TT, Marcus PI (1956) Action of x-rays on mammalian cells. J Exp Med 103(5):653–666
Rana T, Schultz MA, Freeman ML, Biswas S (2012) Loss of Nrf2 accelerates ionizing radiation-induced bone loss by upregulating RANKL. Free Radic Biol Med 53:2298–2307
Reitz G, Berger T, Bilski P, Facius R, Hajek M, Petrov V, Puchalska M, Zhou D, Bossler J, Akatov Y, Shurshakov V, Olko P, Ptaszkiewicz M, Bergmann R, Fugger M, Vana N, Beaujean R, Burmeister S, Bartlett D, Hager L, Palfalvi J, Szabo J, O’Sullivan D, Kitamura H, Uchihori Y, Yasuda N, Nagamatsu A, Tawara H, Benton E, Gaza R, McKeever S, Sawakuchi G, Yukihara E, Cucinotta F, Semones E, Zapp N, Miller J, Dettmann J (2009) Astronaut’s organ doses inferred from measurements in a human phantom outside the international space station. Radiat Res 171:225–235
Roninson IB (2002) Oncogenic functions of tumour suppressor p21(Waf1/Cip1/Sdi1): association with cell senescence and tumour-promoting activities of stromal fibroblasts. Cancer Lett 179:1–14
Rydberg B, Cooper B, Cooper PK, Holley WR, Chatterjee A (2005) Dose-dependent misrejoining of radiation-induced DNA double-strand breaks in human fibroblasts: experimental and theoretical study for high- and low-LET radiation. Radiat Res 163:526–534
Sakurai T, Sawada Y, Yoshimoto M, Kawai M, Miyakoshi J (2007) Radiation-induced reduction of osteoblast differentiation in C2C12 cells. J Radiat Res 48:515–521
Schimmerling W (1992) Radiobiological problems in space. An overview. Radiat Environ Biophys 31:197–203
Sherr CJ, Roberts JM (1999) CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 13:1501–1512
Sorensen BS, Overgaard J, Bassler N (2011) In vitro RBE-LET dependence for multiple particle types. Acta Oncol 50:757–762
Tsuboi K, Yang TC, Chen DJ (1992) Charged-particle mutagenesis. 1. Cytotoxic and mutagenic effects of high-LET charged iron particles on human skin fibroblasts. Radiat Res 129:171–176
Tsuruoka C, Suzuki M, Kanai T, Fujitaka K (2005) LET and ion species dependence for cell killing in normal human skin fibroblasts. Radiat Res 163:494–500
Tsuruoka C, Suzuki M, Hande MP, Furusawa Y, Anzai K, Okayasu R (2008) The difference in LET and ion species dependence for induction of initially measured and non-rejoined chromatin breaks in normal human fibroblasts. Radiat Res 170:163–171
Ugenskiene R, Prise K, Folkard M, Lekki J, Stachura Z, Zazula M, Stachura J (2009) Dose response and kinetics of foci disappearance following exposure to high- and low-LET ionizing radiation. Int J Radiat Biol 85:872–882
Wang X, Kua HY, Hu Y, Guo K, Zeng Q, Wu Q, Ng HH et al (2006) p53 functions as a negative regulator of osteoblastogenesis, osteoblast-dependent osteoclastogenesis, and bone remodeling. J Cell Biol 172:115–125
White RJ, Averner M (2001) Humans in space. Nature 409:1115–1118
Willey JS, Lloyd SA, Nelson GA, Bateman TA (2011) Ionizing radiation and bone loss: space exploration and clinical therapy applications. Clin Rev Bone Miner Metab 9:54–62
Wilson GD (2004) Radiation and the cell cycle, revisited. Cancer Metastasis Rev 23:209–225
Xiong Y, Zhang H, Beach D (1993) Subunit rearrangement of the cyclin-dependent kinases is associated with cellular transformation. Genes Dev 7:1572–1583
Yao KC, Komata T, Kondo Y, Kanzawa T, Kondo S, Germano IM (2003) Molecular response of human glioblastoma multiforme cells to ionizing radiation: cell cycle arrest, modulation of the expression of cyclin-dependent kinase inhibitors, and autophagy. J Neurosurg 98:378–384
Yumoto K, Globus RK, Mojarrab R, Arakaki J, Wang A, Searby ND, Almeida EA, Limoli CL (2010) Short-term effects of whole-body exposure to (56)fe ions in combination with musculoskeletal disuse on bone cells. Radiat Res 173:494–504
Acknowledgments
The authors thank liaison scientists Jonas Klinger and Aljona Groo at the accelerator facility GSI Helmholtzzentrum für Schwerionenforschung GmbH in Darmstadt, Germany for excellent technical assistance. We are grateful to the ESA-IBER consortium for providing funding. Furthermore, the authors thank the Centre de Recherche sur les Ions, les Matériaux et la Photonique physicists and biologists, especially Isabelle Testard, at the heavy ion accelerator Grand Accélérateur National d’Ions Lourds in Caen, France. We are also grateful for the help and support of the Helmholtz Space Life Sciences Research School (SpaceLife), which is funded in equal parts by the Helmholtz Association and the German Aerospace Center and the Chinese Science Council (CSC) for funding Yueyuan Hu.
Conflict of interest
The authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, Y., Hellweg, C.E., Baumstark-Khan, C. et al. Cell cycle delay in murine pre-osteoblasts is more pronounced after exposure to high-LET compared to low-LET radiation. Radiat Environ Biophys 53, 73–81 (2014). https://doi.org/10.1007/s00411-013-0499-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00411-013-0499-0