Abstract
Purpose
Τhat β2-adrenergic receptor (β2AR) haplotypes may play a key role in clinical response to β2-agonists and haplotype Cys-19Gly16Gln27 (CysGlyGln) is reported to be associated with desensitization of β2AR to β-agonists in lymphocytes isolated from patients with asthma and septic shock. We sought to determine whether haplotypic variation of the β2AR affects the functional outcomes of long-acting β2-agonist (LABA) treatment for chronic obstructive pulmonary disease (COPD) when used as monotherapy.
Methods
Treatment-naïve patients with COPD (n = 36) were prospectively treated with two kinds of LABA—inhaled salmeterol and transdermal tulobuterol patch—for 12 weeks in crossover study, and changes in pulmonary function data and 6-minute walk distance (6MWD) were compared between groups stratified by the CysGlyGln.
Results
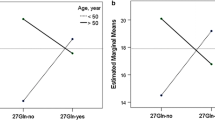
Frequencies of haplotype and diplotype for the CysGlyGln were 0.51 and 0.36, respectively. The individuals homozygous for CysGlyGln showed less improvement in FEV1, %FEF25−75 %, and IC/TLC than those with 0 or 1 copy of CysGlyGln after treatment with both LABAs despite initial bronchodilator responses to albuterol being similar in these groups. The response in these parameters was not significantly different between two types of LABA. Overall changes in 6MWD in individuals with 2 copies of CysGlyGln versus 0 or 1 copy for salmeterol were 2.8 and 11 m, and for tulobuterol were −1.3 and 16 m, respectively.
Conclusions
Homozygous haplotype for the CysGlyGln of β2AR may be associated with susceptibility to desensitization to LABA in patients with COPD.


Similar content being viewed by others
References
National Heart, Lung, and Blood Institute/World Health Organization (2010) Global strategy for the diagnosis, management, and prevention of chronic obstructive lung disease (GOLD): NHLBI/WHO workshop report. NIH Publication, National Institutes of Health, Bethesda
Taylor DR (2007) beta-Adrenergic receptor polymorphisms: relationship to the beta-agonist controversy and clinical implications. Expert Opin Pharmacother 18:3195–3203
Cazzola M, Calzetta L, Matera MG (2011) β2-adrenoceptor agonists: current and future direction. Br J Pharmacol 163:4–17
Drysdale CM, McGraw DW, Stack CB, Stephens JC, Judson RS, Nandabalan K, Arnold K, Ruano G, Liggett SB (2000) Complex promoter and coding region beta 2-adrenergic receptor haplotypes alter receptor expression and predict in vivo responsiveness. Proc Natl Acad Sci USA 97:10483–10488
Hawkins GA, Tantisira K, Meyers DA, Ampleford EJ, Moore WC, Klanderman B, Liggett SB, Peters SP, Weiss ST, Bleecker ER (2006) Sequence, haplotype, and association analysis of ADRbeta2 in a multiethnic asthma case-control study. Am J Respir Crit Care Med 174:1101–1109
Panebra A, Wang WC, Malone MM, Pitter DRG, Weiss ST, Hawlins GA, Liggett SB (2010) Common ADRB2 haplotypes derived from 26 polymorphic sites direct β2-adrenergic receptor expression and regulation phenotypes. PLoS One 5:e11819
Green SA, Turki J, Innis M, Liggett SB (1994) Amino-terminal polymorphisms of the human beta 2-adrenergic receptor impart distinct agonist-promoted regulatory properties. Biochemistry 33:9414–9419
McGraw DW, Forbes SL, Kramer LA, Liggett SB (1998) Polymorphisms of the 5′ leader cistron of the human beta2-adrenergic receptor regulate receptor expression. J Clin Invest 102:1927–1932
Dishy V, Sofowora GG, Xie HG, Kim RB, Byrne DW, Stein CM, Wood AJ (2001) The effect of common polymorphisms of the beta2-adrenergic receptor on agonist-mediated vascular desensitization. N Engl J Med 345:1030–1035
Turki J, Pak J, Green SA, Martin RJ, Liggett SB (1995) Genetic polymorphisms of the beta 2-adrenergic receptor in nocturnal and nonnocturnal asthma. Evidence that Gly16 correlates with the nocturnal phenotype. J Clin Invest 95:1635–1641
Martinez FD, Graves PE, Baldini M, Solomon S, Erickson R (1997) Association between genetic polymorphisms of the beta2-adrenoceptor and response to albuterol in children with and without a history of wheezing. J Clin Invest 100:3184–3188
Israel E, Drazen JM, Liggett SB, Boushey HA, Cherniack RM, Chinchilli VM, Cooper DM, Fahy JV, Fish JE, Ford JG, Kraft M, Kunselman S, Lazarus SC, Lemanske RF, Martin RJ, McLean DE, Peters SP, Silverman EK, Sorkness CA, Szefler SJ, Weiss ST, Yandava CN (2000) The effect of polymorphisms of the beta(2)-adrenergic receptor on the response to regular use of albuterol in asthma. Am J Respir Crit Care Med 162:75–80
Wechsler ME, Lehman E, Lazarus SC et al (2006) beta-Adrenergic receptor polymorphisms and response to salmeterol. Am J Respir Crit Care Med 173:519–526
Bleecker ER, Nelson HS, Kraft M, Corren J, Meyers DA, Yancey SW, Anderson WH, Emmett AH, Ortega HG (2010) Beta2-receptor polymorphisms in patients receiving salmeterol with or without fluticasone propionate. Am J Respir Crit Care Med 181:676–687
Taylor DR, Epton MJ, Kennedy MA et al (2005) Bronchodilator response in relation to β2-adrenoreceptor haplotype in patients with asthma. Am J Respir Crit Care Med 172:700–703
Oostendorp J, Postma DS, Volders H, Jongepier H, Kauffman HF, Boezen HM, Meyers DA, Bleecker ER, Nelemans SA, Zaagsma J, Meurs H (2005) Differential desensitization of homozygous haplotypes of the beta2-adrenergic receptor in lymphocytes. Am J Respir Crit Care Med 172:322–328
Nakada TA, Russell JA, Boyd JH, Aguirre-Hernandez R, Thain KR, Thair SA, Nakada E, McConechy M, Walley KR (2010) beta2-Adrenergic receptor gene polymorphism is associated with mortality in septic shock. Am J Respir Crit Care Med 181:143–149
Bleecker ER, Yancey SW, Baitinger LA et al (2006) Salmeterol response is not affected by β2-adrenergic receptor genotype in subjects with persistent asthma. J Allergy Clin Immunol 118:809–816
Bleecker ER, Postma DS, Lawrance RM, Meyers DA, Ambrose HJ, Goldman M (2007) Effect of ADRB2 polymorphisms on response to long-acting beta2-agonist therapy: a pharmacogenetic analysis of two randomised studies. Lancet 370:2118–2125
Sugawara T, Nanjo Y, Yamazaki M, Higashihara K, Tsuda Y, Mochizuki H, Noguchi T, Takahashi H (2009) Comparison of adherence and efficacy between inhaled salmeterol and transdermal tulobuterol patch in elderly COPD patients. J Am Geriatr Soc 57:919–920
Lipworth BJ, Hall IP, Aziz I, Tan KS, Wheatley A (1999) Beta2-adrenoreceptor polymorphism and bronchoprotective sensitivity with regular short- and long-acting beta2-agonist therapy. Clin Sci (Lond) 96:253–259
Munakata M, Harada Y, Ishida T, Saito J, Nagabukuro A, Matsushita H, Koga N, Ohsaki M, Imagawa K, Shiratsuchi T (2006) Molecular-based haplotype analysis of the beta 2-adrenergic receptor gene (ADRB2) in Japanese asthmatic and non-asthmatic subjects. Allergol Int 55:191–198
Kay LJ, Rostami-Hodjegan A, Suvarna SK, Peachell PT (2007) Influence of beta2-adrenoceptor gene polymorphisms on beta2-adrenoceptor-mediated responses in human lung mast cells. Br J Pharmacol 152(3):323–331
Silverman EK, Kwiatkowski DJ, Sylvia JS, Lazarus R, Drazen JF, Lange C, Liard NM, Weiss ST (2003) Family-based association analysis of β-adrenergic receptor polymorphisms in the childhood asthma management program. J Allergy Clin Immunol 112:870–876
Taylor DR, Hall IP (2007) ADRB2 polymorphisms and beta2 agonists. Lancet 370:2975–2976
Wechsler ME, Lunselman S, Chincillli VM et al (2009) Effect of β2-adrenoceptor polymorphism on response to long-acting β2-agonist in asthma (LARGE trial): a genotype-stratified, randomized, placebo-controlled, crossover trial. Lancet 374:1754–1764
Acknowledgments
No source of funding was used to assist in the preparation of this manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Clinical trial registered with www.umin.ac.jp/ctr/index/htm (UMIN000002629).
Rights and permissions
About this article
Cite this article
Mochizuki, H., Nanjo, Y., Kawate, E. et al. β2-Adrenergic Receptor Haplotype may be Associated with Susceptibility to Desensitization to Long-Acting β2-Agonists in COPD Patients. Lung 190, 411–417 (2012). https://doi.org/10.1007/s00408-012-9387-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-012-9387-7