Abstract
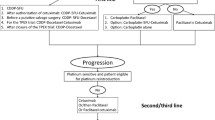
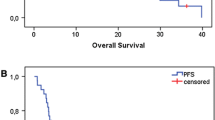
Few therapeutic options are available for recurrent/metastatic head and neck cancer when progression occurs after initial chemotherapy. We analyzed retrospectively the efficacy of weekly Paclitaxel plus Cetuximab as second line of palliative chemotherapy. Patients with squamous carcinoma of head and neck with documented progression after initial treatment were enrolled. Tumor response was evaluated through the response evaluation criteria in solid tumor criteria. The retrospective analysis focused on overall survival (OS) and progression-free survival (PFS). Between 2008 and 2011, 33 consecutive patients were treated. A response rate of 55 % was observed, with median response duration of 5.0 months (95 % CI 3.3–11.1). The median PFS was 4.0 months (95 % CI 2.9–5.0) and the median OS time was 10.0 months (95 % CI 7.9–12.0). Acne-like rash/Folliculitis and chronic anemia were the most common adverse events. A weekly schedule of Paclitaxel plus Cetuximab is a promising regimen for patients with advanced head and neck cancer after failure of platinum-based therapy. Good tolerance of this treatment suggests that would be used in fragile patients.




Similar content being viewed by others
References
Hitt R, Amador ML, Quintela-Fandino M et al (2006) Weekly docetaxel in patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck. Cancer 106:106–111
Vermorken JB, Mesia R, Rivera F et al (2008) Platinum-based chemotherapy plus cetuximab in head and neck cancer. N Engl J Med 359:1116–1127
Vermorken JB, Herbst RS, Leon X, Amellal N, Baselga J (2008) Overview of the efficacy of cetuximab in recurrent and/or metastatic squamous cell carcinoma of the head and neck in patients who previously failed platinum-based therapies. Cancer 112:2710–2719
Molin Y, Fayette J (2011) Current chemotherapies for recurrent/metastatic head and neck cancer. Anticancer Drugs 22:621–625
Baselga J, Trigo JM, Bourhis J et al (2005) Phase II multicenter study of the antiepidermal growth factor receptor monoclonal antibody cetuximab in combination with platinum-based chemotherapy in patients with platinum-refractory metastatic and/or recurrent squamous cell carcinoma of the head and neck. J Clin Oncol 23:5568–5577
León X, Hitt R, Constenla M et al (2005) A retrospective analysis of the outcome of patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck refractory to a platinum-based chemotherapy. J Clin Oncol (R Coll Radiol) 17:418–424
Licitra L, Mesia R, Rivera F et al (2011) Evaluation of EGFR gene copy number as a predictive biomarker for the efficacy of cetuximab in combination with chemotherapy in the first-line treatment of recurrent and/or metastatic squamous cell carcinoma of the head and neck: EXTREME study. Ann Oncol 22:1078–1087
Tejani MA, Cohen RB, Mehra R (2010) The contribution of cetuximab in the treatment of recurrent and/or metastatic head and neck cancer. Biologics 4:173–185
Kerbel RS, Klement G, Pritchard KI, Kamen B (2002) Continuous low-dose anti-angiogenic/metronomic chemotherapy: from the research laboratory into the oncology clinic. Ann Oncol 13:12–15
Caballero M, Grau JJ, Blanch JL et al (2007) Serum vascular endothelial growth factor as a predictive factor in metronomic (weekly) Paclitaxel treatment for advanced head and neck cancer. Arch Otolaryngol Head Neck Surg 133:1143–1148
Fayette J, Montella A, Chabaud S et al (2010) Paclitaxel is effective in relapsed head and neck squamous cell carcinoma: a retrospective study of 66 patients at a single institution. Anticancer Drugs 21:553–558
Grau JJ, Caballero M, Verger E, Monzó M, Blanch JL (2009) Weekly paclitaxel for platin-resistant stage IV head and neck cancer patients. Acta Otolaryngol 129:1294–1299
Inoue K, Slaton JW, Perrotte P et al (2000) Paclitaxel enhances the effects of the anti-epidermal growth factor receptor monoclonal antibody ImClone C225 in mice with metastatic human bladder transitional cell carcinoma. Clin Cancer Res 6:4874–4884
Bernier J (2008) Drug Insight: cetuximab in the treatment of recurrent and metastatic squamous cell carcinoma of the head and neck. Nat Clin Pract Oncol 12:705–713
Caballero M, E Sosa A, Tagliaplietra A, Grau JJ (2013) Metastatic adenoid cystic carcinoma of the salivary gland responding to cetuximab plus weekly paclitaxel after no response to weekly paclitaxel alone. Head Neck 35:E52–E54
Hitt R, Irigoyen A, Cortes-Funes H, Grau JJ, García-Sáenz JA, Cruz-Hernandez JJ, The Spanish Head and Neck Cancer Cooperative Group (TTCC) (2012) Phase II study of the combination of cetuximab and weekly paclitaxel in the first-line treatment of patients with recurrent and/or metastatic squamous cell carcinoma of head and neck. Ann Oncol 23:1016–1022
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumors: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Constenla DO, Hill ME, A’Hern RP et al (1997) Chemotherapy for symptom control in recurrent squamous cell carcinoma of the head and neck. Ann Oncol 8:445–449
Acknowledgments
We would like to thank the members of the Spanish Head and Neck Cancer Cooperative Group, TTCC, for their interest in this study and their collaboration in the redaction of this manuscript.
Conflict of interest
There are no actual or potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sosa, A.E., Grau, J.J., Feliz, L. et al. Outcome of patients treated with palliative weekly Paclitaxel plus Cetuximab in recurrent head and neck cancer after failure of platinum-based therapy. Eur Arch Otorhinolaryngol 271, 373–378 (2014). https://doi.org/10.1007/s00405-013-2537-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-013-2537-6