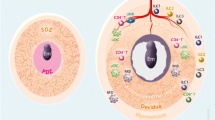
Abstract
The tolerance of the semi-allogeneic fetus by the maternal immune system is an eternal topic of reproductive immunology for ensuring a satisfactory outcome. The maternal–fetal interface serves as a direct portal for communication between the fetus and the mother. It is composed of placental villi trophoblast cells, decidual immune cells, and stromal cells. Decidual immune cells engage in maintaining the homeostasis of the maternal–fetal interface microenvironment. Furthermore, growing evidence has shown that decidual macrophages play a crucial role in maternal–fetal tolerance during pregnancy. As the second largest cell population among decidual immune cells, decidual macrophages are divided into two subtypes: classically activated macrophages (M1) and alternatively activated macrophages (M2). M2 polarization is critical for placentation and embryonic development. Cytokines, exosomes, and metabolites regulate the polarization of decidual macrophages, and thereby modulate maternal–fetal immunotolerance. Explore the initial relationship between decidual macrophages polarization and maternal–fetal immunotolerance will help diagnose and treat the relevant pregnancy diseases, reverse the undesirable outcomes of mothers and infants.



Similar content being viewed by others
Data availability
All relevant data are within the paper. The data are available from the corrsponding author on reasonable request.
Abbreviations
- DM\(\phi\) :
-
Deciduous macrophages
- dSC:
-
Decidual stromal cell
- dDC:
-
Decidual dendritic cell
- M1:
-
Classically activated macrophage
- M2:
-
Alternative activated macrophage
- dM1:
-
Classically activated decidual macrophage
- dM2:
-
Alternative activated decidumacrophage
- dNK:
-
Decidual nature killer cell
- LILR:
-
Leukocyte Ig-like receptors
- KIR:
-
Killer cell Ig-like receptor
- MSC:
-
Mesenchymal stem cell
- CCR2:
-
CC chemokine receptor 2
- CD11c:
-
Integrin\(\alpha\)-X
- HMOX1:
-
Heme oxygenase-1
- IL:
-
Interleukin
- NLRP3:
-
NOD-like receptor thermal protein domain associated protein 3
- GM-CSF:
-
Macrophage colony-stimulating factor 2
- M-CSF:
-
Macrophage colony-stimulating factor
- IFN-\(\gamma\) :
-
Interferon \(\gamma\)
- Tumor necrosis factor-\(\alpha\) :
-
TNF-\(\alpha\)
- TGF-\(\beta\) :
-
Transforming growth factor \(\beta\)
- STAT:
-
Signal transducer and activator of transcription
- NOS2:
-
Nitric oxide synthase 2
- IRF4:
-
Interferon regulatory factor 4
- RSA:
-
Recurrent spontaneous abortion
- PE:
-
Preecampsia
- CRH:
-
Corticotropin-releasing hormone
- hCG:
-
Human chorionic gonadotropin
- VIP:
-
Vasoactive intestinal peptide
- HDAC9:
-
Histone deacetylase 9
- Tim-3:
-
T cell immunoglobulin mucin 3
- Gal-9:
-
Galactoglutinin-9
- UHRF1:
-
Ubiquitin like with PHD and ring finger domains 1
- FBP:
-
Fructose-1,6-bisphosphate
- EVT:
-
Extravillous trophoblasts
- EXOC2:
-
Exocyst complex component 2
- GAS6:
-
Growth arrest specific 6
- NLRP7:
-
NLR family pyrin domain containing 7
- PlGF:
-
Placental growth factor
- PPAR\(\gamma\) :
-
Peroxisome proliferator-activated receptor gamma
References
Medawar PB (1961) Immunological tolerance: the phenomenon of tolerance provides a testing ground for theories of the immune response. Science 133(3449):303–306
Krop J, van der Zwan A, Ijsselsteijn ME et al (2022) Imaging mass cytometry reveals the prominent role of myeloid cells at the maternal–fetal interface. Iscience 25(7):104648
Li C, Houser BL, Nicotra ML et al (2009) HLA-G homodimer-induced cytokine secretion through HLA-G receptors on human decidual macrophages and natural killer cells. Proc Natl Acad Sci 106(14):5767–5772
Gomez-Lopez N, Garcia-Flores V, Chin PY et al (2021) Macrophages exert homeostatic actions in pregnancy to protect against preterm birth and fetal inflammatory injury. JCI Insight 6(19):e146089
Ding J, Zhang Y, Cai X et al (2021) Crosstalk between trophoblast and macrophage at the maternal–fetal interface: current status and future perspectives. Front Immunol 12:758281
Murray PJ, Allen JE, Biswas SK et al (2014) Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41(1):14–20
Bai K, Lee CL, Liu X et al (2022) Human placental exosomes induce maternal systemic immune tolerance by reprogramming circulating monocytes. J Nanobiotechnol 20(1):86
Ma C, Zhao M, Wang X et al (2023) Downregulation of miR-455-3p in decidual cells promotes macrophage polarization and suppresses trophoblasts invasion. Exp Cell Res 425(2):113510
Hx Ye, Gn Liao, Yj Dong et al (2022) miR-146a-5p enhances embryo survival in unexplained recurrent spontaneous abortion by promoting M2 polarization of decidual macrophages. Int Immunopharmacol 110:108930
Zhang J, Tao Y, Cai R et al (2022) miR-196a-5p-rich extracellular vesicles from trophoblasts induce M1 polarization of macrophages in recurrent miscarriage. J Immunol Res 2022:6811632
Bai K, Li J, Lin L et al (2023) Placenta exosomal miRNA-30d-5p facilitates decidual macrophage polarization by targeting HDAC9. J Leukoc Biol 113(5):434–444
Liu X, Fei H, Yang C et al (2022) Trophoblast-derived extracellular vesicles promote preeclampsia by regulating macrophage polarization. Hypertension 79(10):2274–2287
Ying X, Jin X, Zhu Y et al (2020) Exosomes released from decidual macrophages deliver miR-153-3p, which inhibits trophoblastic biological behavior in unexplained recurrent spontaneous abortion. Int Immunopharmacol 88:106981
Ding J, Zhang Y, Cai X et al (2021) Extracellular vesicles derived from M1 macrophages deliver miR-146a-5p and miR-146b-5p to suppress trophoblast migration and invasion by targeting TRAF6 in recurrent spontaneous abortion. Theranostics 11(12):5813
Ying X, Jin X, Zhu Y et al (2020) Exosomes released from decidual macrophages deliver miR-153-3p, which inhibits trophoblastic biological behavior in unexplained recurrent spontaneous abortion. Int Immunopharmacol 88:106981
Ding J, Zhang Y, Cai X et al (2021) Extracellular vesicles derived from m1 macrophages deliver miR-146a-5p and miR-146b-5p to suppress trophoblast migration and invasion by targeting TRAF6 in recurrent spontaneous abortion. Theranostics 11(12):5813
Shan Y, Shen S, Long J et al (2022) Term and preterm birth initiation is associated with the macrophages shifting to M1 polarization in gestational tissues in mice. Biology 11(12):1759
Schonkeren D, van der Hoorn ML, Khedoe P et al (2011) Differential distribution and phenotype of decidual macrophages in preeclamptic versus control pregnancies. Am J Pathol 178(2):709–717
Jiang X, Du MR, Li M et al (2018) Three macrophage subsets are identified in the uterus during early human pregnancy. Cell Mol Immunol 15(12):1027–1037
Lasch M, Sudan K, Paul C et al (2022) Isolation of decidual macrophages and Hofbauer cells from term placenta-comparison of the expression of CD163 and CD80. Int J Mol Sci 23(11):6113
Mills CD, Kincaid K, Alt JM et al (2000) M-1/M-2 macrophages and the Th1/Th2 paradigm. J Immunol 164(12):6166–6173
Vondra S, Höbler AL, Lackner AI et al (2023) The human placenta shapes the phenotype of decidual macrophages. Cell Rep 42(1):111977
Zheng Y, Pan J, Xia C et al (2022) Characterization of placental and decidual cell development in early pregnancy loss by single-cell RNA sequencing. Cell Biosci 12(1):1–15
Wei P, Dong M, Bi Y et al (2022) Identification and validation of a signature based on macrophage cell marker genes to predict recurrent miscarriage by integrated analysis of single-cell and bulk rna-sequencing. Front Immunol 13:1053819
Houser BL, Tilburgs T, Hill J et al (2011) Two unique human decidual macrophage populations. J Immunol 186(4):2633–2642
Li M, Sun F, Xu Y et al (2022) Tim-3+ decidual M\(\varphi\)s induced Th2 and Treg bias in decidual CD4+ T cells and promoted pregnancy maintenance via CD132. Cell Death Dis 13(5):454
Lv X, Li X, Dai X et al (2020) Investigation heme oxygenase-1 polymorphism with the pathogenesis of preeclampsia. Clin Exp Hypertens 42(2):167–170
Sun T, Cruz GI, Mousavi N et al (2022) HMOX1 genetic polymorphisms display ancestral diversity and may be linked to hypertensive disorders in pregnancy. Reprod Sci 29(12):3465–3476
Gallardo V, González M, Toledo F et al (2020) Role of heme oxygenase 1 and human chorionic gonadotropin in pregnancy associated diseases. Biochimica et Biophysica Acta (BBA)-Mol Basis Dis 1866(2):165522
Wang H, He M, Hou Y et al (2016) Role of decidual CD14+ macrophages in the homeostasis of maternal–fetal interface and the differentiation capacity of the cells during pregnancy and parturition. Placenta 38:76–83
Romanowska-Próchnicka K, Felis-Giemza A, Olesińska M et al (2021) The role of TNF-\(\alpha\) and anti-TNF-\(\alpha\) agents during preconception, pregnancy, and breastfeeding. Int J Mol Sci 22(6):2922
Svensson J, Jenmalm MC, Matussek A et al (2011) Macrophages at the fetal–maternal interface express markers of alternative activation and are induced by M-CSF and IL-10. J Immunol 187(7):3671–3682
Valero-Pacheco N, Tang EK, Massri N et al (2022) Maternal IL-33 critically regulates tissue remodeling and type 2 immune responses in the uterus during early pregnancy in mice. Proc Natl Acad Sci 119(35):e2123267119
Lin Z, Shi JL, Chen M et al (2023) CCL2: an important cytokine in normal and pathological pregnancies: a review. Front Immunol 13:1053457
Lindau R, Mehta RB, Lash G et al (2018) Interleukin-34 is present at the fetal–maternal interface and induces immunoregulatory macrophages of a decidual phenotype in vitro. Hum Reprod 33(4):588–599
Toniolo A, Fadini GP, Tedesco S et al (2015) Alternative activation of human macrophages is rescued by estrogen treatment in vitro and impaired by menopausal status. J Clin Endocrinol Metab 100(1):E50–E58
Evans P, Wheeler T, Anthony F et al (1997) Maternal serum vascular endothelial growth factor during early pregnancy. Clin Sci 92(6):567–571
Vishnyakova P, Poltavets A, Nikitina M et al (2021) Expression of estrogen receptor \(\alpha\) by decidual macrophages in preeclampsia. Biomedicines 9(2):191
Furcron AE, Romero R, Mial TN et al (2016) Human chorionic gonadotropin has anti-inflammatory effects at the maternal–fetal interface and prevents endotoxin-induced preterm birth, but causes dystocia and fetal compromise in mice. Biol Reprod 94(6):136
Furcron AE, Romero R, Plazyo O et al (2015) Vaginal progesterone, but not 17\(\alpha\)-hydroxyprogesterone caproate, has antiinflammatory effects at the murine maternal–fetal interface. Am J Obstet Gynecol 213(6):846-e1
Motomura K, Miller D, Galaz J et al (2023) The effects of progesterone on immune cellular function at the maternal–fetal interface and maternal circulation. J Steroid Biochem Mol Biol 229:106254
Arenas-Hernandez M, Romero R, Xu Y et al (2019) Effector and activated T cells induce preterm labor and birth that is prevented by treatment with progesterone. J Immunol 202(9):2585–2608
Tsai YC, Tseng JT, Wang CY et al (2017) Medroxyprogesterone acetate drives M2 macrophage differentiation toward a phenotype of decidual macrophage. Mol Cell Endocrinol 452:74–83
Paparini DE, Grasso E, Fernandez LdC et al (2021) Decidual factors and vasoactive intestinal peptide guide monocytes to higher migration, efferocytosis and wound healing in term human pregnancy. Acta Physiol 232(1):e13579
Paparini DE, Choudhury RH, Vota DM et al (2019) Vasoactive intestinal peptide shapes first-trimester placenta trophoblast, vascular, and immune cell cooperation. Br J Pharmacol 176(7):964–980
Carrión M, Pérez-García S, Martínez C et al (2016) VIP impairs acquisition of the macrophage proinflammatory polarization profile. J Leucoc Biol 100(6):1385–1393
Pedersen-White JR, Chorich LP, Bick DP et al (2008) The prevalence of intragenic deletions in patients with idiopathic hypogonadotropic hypogonadism and Kallmann syndrome. MHR Basic Sci Reprod Med 14(6):367–370
Wu S, Liu H, Zhou M et al (2022) The miR-410-5p/ITGA6 axis participates in the pathogenesis of recurrent abortion by regulating the biological function of trophoblast. J Reprod Immunol 152:103647
Sheng YR, Hu WT, Shen HH et al (2022) An imbalance of the IL-33/ST2-AXL-efferocytosis axis induces pregnancy loss through metabolic reprogramming of decidual macrophages. Cell Mol Life Sci 79(3):173
Jung J, Zeng H, Horng T (2019) Metabolism as a guiding force for immunity. Nat Cell Biol 21(1):85–93
O’Neill LA, Pearce EJ (2016) Immunometabolism governs dendritic cell and macrophage function. J Exp Med 213(1):15–23
Gao L, Xu QH, Ma LN et al (2022) Trophoblast-derived lactic acid orchestrates decidual macrophage differentiation via SRC/LDHA signaling in early pregnancy. Int J Biol Sci 18(2):599
Ma LN, Huang XB, Muyayalo KP et al (2020) Lactic acid: a novel signaling molecule in early pregnancy? Front Immunol 11:279
Li ZH, Wang LL, Liu H et al (2019) Galectin-9 alleviates LPS-induced preeclampsia-like impairment in rats via switching decidual macrophage polarization to M2 subtype. Front Immunol 9:3142
Zhang Y, Ma L, Hu X et al (2019) The role of the PD-1/PD-L1 axis in macrophage differentiation and function during pregnancy. Hum Reprod 34(1):25–36
Löb S, Knabl J, Vattai A et al (2023) Obesity in pregnancy is associated with macrophage influx and an upregulated GRO-alpha and IL-6 expression in the decidua. J Reprod Immunol 156:103800
Huang SJ, Zenclussen AC, Chen CP et al (2010) The implication of aberrant GM-CSF expression in decidual cells in the pathogenesis of preeclampsia. Am J Pathol 177(5):2472–2482
Li M, Piao L, Chen CP et al (2016) Modulation of decidual macrophage polarization by macrophage colony-stimulating factor derived from first-trimester decidual cells: implication in preeclampsia. Am J Pathol 186(5):1258–1266
Liu H, Wang LL, Xu QH et al (2022) UHRF1 shapes both the trophoblast invasion and decidual macrophage differentiation in early pregnancy. FASEB J 36(4):e22247
Wei CY, Li MQ, Zhu XY et al (2021) Immune status of decidual macrophages is dependent on the CCL2/CCR2/JAK2 pathway during early pregnancy. Am J Reprod Immunol 86(5):e13480
Petroff MG, Sedlmayr P, Azzola D et al (2002) Decidual macrophages are potentially susceptible to inhibition by class Ia and class Ib HLA molecules. J Reprod Immunol 56(1–2):3–17
Vento-Tormo R, Efremova M, Botting RA et al (2018) Single-cell reconstruction of the early maternal–fetal interface in humans. Nature 563(7731):347–353
Wang L, Wang H, Luo J et al (2022) Decorin promotes decidual M1-like macrophage polarization via mitochondrial dysfunction resulting in recurrent pregnancy loss. Theranostics 12(17):7216
Yang HL, Lai ZZ, Shi JW et al (2022) A defective lysophosphatidic acid-autophagy axis increases miscarriage risk by restricting decidual macrophage residence. Autophagy 18(10):2459–2480
Mittelberger J, Seefried M, Löb S et al (2023) The programmed cell death protein 1 (PD1) and the programmed cell death ligand 1 (PD-L1) are significantly downregulated on macrophages and Hofbauer cells in the placenta of preeclampsia patients. J Reprod Immunol 157:103949
Laskewitz A, van Benthem K, Kieffer T et al (2019) The influence of maternal obesity on macrophage subsets in the human decidua. Cell Immunol 336:75–82
Lindau R, Vondra S, Spreckels J et al (2021) Decidual stromal cells support tolerance at the human foetal–maternal interface by inducing regulatory M2 macrophages and regulatory T-cells. J Reprod Immunol 146:103330
Fernandez-Santamaria R, Satitsuksanoa P (2022) Engineered IL-10: a matter of affinity. Allergy 77(3):1067–1069
Yang L, Zhang X, Gu Y et al (2022) SEC5 is involved in M2 polarization of macrophages via the stat6 pathway, and its dysfunction in decidual macrophages is associated with recurrent spontaneous abortion. Front Cell Dev Biol 10:891748
Dai JC, Yang JY, Chang RQ et al (2022) GAS6-mediated dialogue between decidual stromal cells and macrophages is essential for early pregnancy maintenance by inducing M2-like polarization and cell proliferation of decidual macrophages. Mol Hum Reprod 28(3):gaac006
Tsai PY, Chen KR, Li YC et al (2019) NLRP7 is involved in the differentiation of the decidual macrophages. Int J Mol Sci 20(23):5994
Chang R, Dai J, Wang L et al (2023) PLGF/FLT-1 deficiency leads to reduced STAT3-C/EBP\(\beta\) signaling and aberrant polarization in decidual macrophages during early spontaneous abortion. Front Immunol 14:1061949
Kolben TM, Rogatsch E, Vattai A et al (2018) PPAR\(\gamma\) expression is diminished in macrophages of recurrent miscarriage placentas. Int J Mol Sci 19(7):1872
Wang S, Liu T, Nan N et al (2023) Exosomes from human umbilical cord mesenchymal stem cells facilitates injured endometrial restoring in early repair period through miR-202-3p mediating formation of ECM. Stem Cell Rev Rep 19(6):1954–1964
Zhao G, Miao H, Li X et al (2016) TGF-\(\beta\)3-induced miR-494 inhibits macrophage polarization via suppressing PGE 2 secretion in mesenchymal stem cells. FEBS Lett 590(11):1602–1613
Zhou H, Wang H, Liu X et al (2023) Downregulation of miR-92a in decidual stromal cells suppresses migration ability of trophoblasts by promoting macrophage polarization. DNA Cell Biol 42(8):507–514
Wei R, Lai N, Zhao L et al (2021) Dendritic cells in pregnancy and pregnancy-associated diseases. Biomed Pharmacotherapy 133:110921
Freitag N, Zwier M, Barrientos G et al (2014) Influence of relative NK-DC abundance on placentation and its relation to epigenetic programming in the offspring. Cell Death Dis 5(8):e1392–e1392
Cortés-Morales VA, Chávez-Sánchez L, Rocha-Zavaleta L et al (2023) Mesenchymal stem/stromal cells derived from cervical cancer promote M2 macrophage polarization. Cells 12(7):1047
Babazadeh S, Nassiri SM, Siavashi V et al (2021) Macrophage polarization by MSC-derived CXCL12 determines tumor growth. Cell Mol Biol Lett 26(1):30
Li Y, Zhang D, Xu L et al (2019) Cell–cell contact with proinflammatory macrophages enhances the immunotherapeutic effect of mesenchymal stem cells in two abortion models. Cell Mol Immunol 16(12):908–920
Li Y, Zhang D, Xu L et al (2019) Cell–cell contact with proinflammatory macrophages enhances the immunotherapeutic effect of mesenchymal stem cells in two abortion models. Cell Mol Immunol 16(12):908–920
Muñoz-Fernández R, De La Mata C, Requena F et al (2019) Human predecidual stromal cells are mesenchymal stromal/stem cells and have a therapeutic effect in an immune-based mouse model of recurrent spontaneous abortion. Stem Cell Res Therapy 10(1):1–13
Zhou WJ, Yang HL, Mei J et al (2022) Fructose-1,6-bisphosphate prevents pregnancy loss by inducing decidual COX-2+ macrophage differentiation. Sci Adv 8(8):eabj2488
Yang HL, Lai ZZ, Shi JW et al (2022) A defective lysophosphatidic acid-autophagy axis increases miscarriage risk by restricting decidual macrophage residence. Autophagy 18(10):2459–2480
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
XTJ: Data collection, Manuscript writing. LL: Project Development and Manuscript Editing. All authors contributed to the study conception and design. Data collection was performed by XJ. The first draft of the manuscript was written by XJ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, X., Li, L. Decidual macrophage: a reversible role in immunotolerance between mother and fetus during pregnancy. Arch Gynecol Obstet 309, 1735–1744 (2024). https://doi.org/10.1007/s00404-023-07364-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-023-07364-3