Abstract
Objective
To present the clinicopathological features of metastatic ovarian neoplasms with emphasis in the diagnostic challenge.
Methods
This is a retrospective study including 97 patients with pathological diagnosis of metastatic ovarian neoplasms, examined during the decade 2000–2009. The gross, microscopical and immunohistochemical characteristics as well as the clinical data (age of the patients, origin of the neoplasm, symptoms, treatment options) and 5-year survival rates were examined.
Results
The mean age of the patients is 55 years (range 26–78 years). 62.89% of the tumors were metastatic from extragenital organs (from stomach 21.65%, breast 15.46%, colon 15.46%, appendix 3.09%, pancreas 2.06%, lung 1.03% and kidney 1.03%, sarcoma 1.03% melanoma 1.03%) and 37.11% tumors originated from the genital tract. The 3-year survival rates ranged from 25.39% for metastatic ovarian neoplasms originating outside the genital tract up to 29.41% for those originating from the genital tract. Tumor immunohistochemistry is a helpful aid in the differential diagnosis mainly between primary mucinous ovarian tumors and metastatic colon cancers and in the recognition of metastatic breast cancers and other neoplasms of the GI tract.
Conclusion
The management of metastatic ovarian neoplasms should include specific immunohistochemical methods in order to identify the primary neoplasm site. The differential diagnosis of a pelvic mass should always include metastatic neoplasms of the ovaries.
Similar content being viewed by others
Introduction
Ovarian carcinoma is the fifth most common carcinoma in women [1], and is the leading cause of death from gynecologic malignancies [2, 3]. Metastatic ovarian tumors are neoplasms of the ovaries which usually originate from distant organs such as the stomach, the breast or the colon and constitute 5% of the total ovarian tumors and 10–30% of the total malignant ovarian tumors [4–10].
This difference in the incidence of metastatic ovarian neoplasms may be explained by various factors such as geographic distribution (e.g. great incidence of gastric cancer in Japan), the age groups, the experience of the laboratory personnel examining the specimens, and the laboratory methods used in the correct diagnosis [11, 12].
In many cases, metastatic ovarian tumors are detected by the pathology examination before the clinical detection of the primary tumor. It is reported that metastatic ovarian tumors preceded the primary tumor detection in 38% of patients [13].
In certain cases there are difficulties in the differential diagnosis of primary or metastatic ovarian neoplasms by routine histopathological examination. The main diagnostic problems include metastatic ovarian neoplasms from colon carcinomas and primary ovarian mucinous cystadenocarcinomas. Up to 45% of metastatic ovarian neoplasms from colon cancer have been primarily diagnosed as primary ovarian neoplasms [14, 15].
In cases of breast carcinomas, microscopic ovarian metastases have been diagnosed after prophylactic oophorectomy in 50% of the cases and the ovaries in 63.3% of cases present a grossly normal appearance [16].
In this study, we present the clinical and histopathological characteristics of metastatic ovarian neoplasms and the overall 3-year survival of those patients and the immunohistochemical characteristics of the tumors that are helpful in the correct diagnosis
Materials and methods
A retrospective study was conducted in the 2nd Department of Obstetrics and Gynecology, University of Athens, Aretaieion Hospital, Athens, Greece. During the period January 2000 to December 2009, 1,055 ovarian neoplasms were managed in our department from which 520 were primary malignant tumors and 97 were metastatic ovarian neoplasms. All the patients were operated in our department and the histopathological examination was performed in the Histopathology Department of our hospital. The available clinical records of patients with metastasis to the ovary were reviewed for age at diagnosis, origin of the neoplasm, consistency and appearance of the neoplasm, histological type and the 3-year survival. The gross, microscopic and immunohistochemical findings of metastatic ovarian neoplasms were reviewed from the pathological files.
All specimens were formalin fixed, routinely processed and stained by hematoxylin and eosin. The immunohistochemical study was performed by Ventana automated system with the use of following abs: CK7 (Monosan, 1:25), CK20 (DAKO, 1:50), CA125 (Monosan 1:25), CEA (Monosan 1:25), ER (Novocastra 1:50), PgR (Thermoscience 1:50), TTF1 (Cellmarque 1:25).
The survival rates were calculated in months from the time of surgical intervention.
Results
Ovarian metastatic neoplasms comprise 97/1055 (9.19%) of all ovarian neoplasms and 97/520 (18.65%) of the total malignant ovarian neoplasms treated in our Clinic.
The median age of the patients was 55 years (range 26–78 years).
43/97 (44.33%) patients were premenopausal, whereas 54/97 (55.67%) were postmenopausal. 88/97 (90.72%) had a previous cancer history, 21/97 (21.65%) patients reported oral contraceptive use and 9/97 (9.28%) patients reported hormone replacement treatment (Table 1). In relation to localization of the metastatic ovarian neoplasms, 51/97 cases (52.58%) had bilateral development, 24/97 (24.74%) localized in the right and 22/97 (22.68%) in the left ovary.
Of the total study group, 61 cases (62.89%) originated from non-genital tract malignancies and 36 cases (37.11%) from genital tract malignancies (Table 2).
Forty-four cases (%) originated from the gastrointestinal tract (24 cases of gastric carcinoma, 15 cases of colon carcinoma, 3 cases of appendix carcinoma, 2 cases of pancreatic carcinoma) and 15 cases originated from the breast.
Of the primary genital tract malignancies that metastasized to the ovary, endometrial neoplasms were the most common (18/97).
Gross morphology
45/97 of the metastatic ovarian neoplasms were ovoid or kidney-shaped, often with cystic areas occupying <50% of the tumor mass. The external surface was generally smooth and nodular in 43 cases and irregular in 2 cases. In 40 cases extensive necrosis was observed.
Carcinomas metastatic from colon cancers were large and cystic (15/15 cases) with the exception of gastric carcinomas which were large and solid tumors presenting the typical gross and microscopical morphology of Krukenberg tumors in 20/24 cases.
The mean diameter of the metastatic ovarian neoplasms was 7.1 cm with a range of 1–24 cm.
The smallest ovarian neoplasms originated from the breast, whereas the largest ovarian neoplasms originated from the colon with the biggest recorded metastatic neoplasm weighting 1,580 g (Figs. 1, 2, 3).
Histology
All cases of the primary gastrointestinal tumors were adenocarcinomas (100%) and in 20/24 cases of gastric cancer a signet ring cell morphology was observed (Figs. 4, 5).
The predominant histological type of the metastatic breast tumors was that of infiltrating ductal cell carcinoma (NOS) (13 cases) and in two cases a lobular type was observed.
Metastatic genital tract tumors were endometrioid adenocarcinomas in 12/18 cases (66.67%) serous papillary carcinomas in 6/18 cases (33.33%) squamous cell cervical cancers in 9 cases and in 3 cases a primary fallopian tube cancer was diagnosed.
The cases of metastatic sarcoma, melanoma and lung cancer presented a typical morphology that was consistent with the previously reported primary tumors.
Metastatic GI tract cancers that presented the main diagnostic problem because of their morphological similarity with primary ovarian mucinous tumors had a distinct immunoprofile (CK7−, CK20+, CA125−, TTF1−, ER−). Metastatic breast cancer also presented a distinct immunoprofile (ER+, PGR+, CK7+, CA125−, CK20−, TTF1− (Fig. 6). The rest of the cases presented an histological morphology that in combination with the clinical history permitted the correct diagnosis.
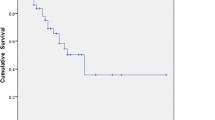
The 3-year survival of patients with metastatic ovarian neoplasms was 26.8% after surgical resection of the neoplasms.
The 3-year survival rate of the 63 patients with metastatic ovarian neoplasms from a non-genital tract site were 25.4%, and of the 34 patients with metastatic ovarian neoplasms from a genital tract site the survival rates were 29.4% (Tables 3, 4).
Discussion
The differential diagnosis between primary and metastatic ovarian neoplasms is important and the primary site of the latter should be diagnosed by the histopathological examination of the ovary. However, according to Yada-Hashimoto et al. [16] not all histopathological examinations lead to a diagnosis of the primary site. About 4.4% of female patients who died from malignant neoplasms were found to have ovarian metastases at autopsy [18], while 25% of patients with breast carcinoma who underwent prophylactic oophorectomy had ovarian metastases [16].
In our Department, the incidence of metastatic ovarian neoplasms was 97/520 malignant ovarian tumors (18.65%).
However, the incidence of such malignancies ranged from 6 to 30% in previous studies [14, 17, 19–21]. It should be mentioned that our material did not include cases of prophylactic oophorectomy for breast carcinomas.
The main primary sites of ovarian metastatic neoplasms were as follows in our study: gastrointestinal tract 44/97 (45.36%), breast 15/97 (15.46%) and genital tract 36/97 (37.11%). Our study is consistent with previous studies which showed that the most common origin for metastatic ovarian neoplasms is the gastrointestinal tract [19]. However, the Mediterranean diet in comparison to the Japanese diet is considered responsible for the difference in the incidence of ovarian metastatic gastric carcinoma (24/97—24.74% of the cases in our study) compared to the results of the study of Yada-Hashimoto et al. [16]. Moreover, in our study, Krukenberg tumors were identified in 20/97 cases (20.62%), a finding which is in accordance with previous studies [21]. It should be noticed that we characterized neoplasms as Krukenberg tumors according to the Woodruff and Novak criteria [22] which defined that Krukenberg tumor is a tumor with fibrous stroma infiltrated by characteristic mucin-filled, signet ring adenocarcinoma cells.
The median age of the patients in this study was 55 years ranging from 26 to 78 years. The finding seems to be higher than previous studies; however, the high incidence of metastatic ovarian neoplasms originating from the genital tract could explain it as such malignancies are found in older ages [15]. Moreover, in our study, the younger median age (46 years) of metastatic ovarian neoplasms originating from the stomach should be mentioned.
Localization of the neoplasms tends to be bilateral and is attributed to lymphatic dissemination of breast carcinoma and neoplasms of the upper abdomen [5, 15].
Cystic tumors were found in 48/97 cases (49.48%) in our study and 39/48 of them (81.25%) originated from tumors of the gastrointestinal tract and the finding is in consistence with previous studies [20, 23]. Breast metastases tend to create small nodules in the ovary and usually are inconspicuous on observation.
The diameter of most ovaries in our study did not exceed 7 cm and is in accordance to the reported size of metastatic ovarian neoplasms described in previous studies [24, 25].
The immunohistochemical investigation showed that colorectal cancers metastatic to the ovaries, which present the main differential diagnostic problem from primary mucinous tumor, present a distinct immunoprofile [tumor cells CK20 (+), CEA (+), CK7 (−), ER (−), PR (−), CA125 (−)] that permit the accurate diagnosis [16].
The survival of patients with metastatic ovarian neoplasms is rather poor ranging from 19 to 47% in neoplasms originating from non-genital tract to genital tract organs, respectively [17]. In that study, the median survival time of patients with ovarian metastatic neoplasms with origin genital tract tumors was 48 months, while for those originating from non-genital tract tumors was 12 months. In our study, the overall 3-year survival for surgically managed ovarian metastatic neoplasms was 31/97 (31.96%), while 15/36 (41.67%) and 16/63 (25.4%) were the survival rates in the genital and non-genital tract tumors, respectively.
In conclusion, patients with ovarian metastatic neoplasms are in the fifth to sixth decade and present bilateral lesions of the ovaries. The primary sites are more frequently the gastrointestinal tract which create cystic mucinous tumors, the breast, and the endometrium. Immunohistochemistry is a valuable aid in the correct diagnosis of metastatic tumors especially in cases of neoplasms originating from the colon.
The presence of metastatic ovarian neoplasms should always be included in the differential diagnosis of a pelvic tumor to avoid diagnostic or treatment pitfalls. The prognosis is generally poor since these tumors represented cases in late stages of the disease.
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ (2009) Cancer statistics, 2009. CA Cancer J Clin 59(4):225–249
Colombo N, Van Gorp T, Parma G, Amant F, Gatta G, Sessa C, Vergote I (2006) Ovarian cancer. Crit Rev Oncol Hematol 60(2):159–179
Lenhard SM, Bufe A, Kümper C, Stieber P, Mayr D, Hertlein L, Kirschenhofer A, Friese K, Burges A (2009) Relapse and survival in early-stage ovarian cancer. Arch Gynecol Obstet 280(1):71–77
Holtz F, Hart WR (1982) Krukenberg tumors of the ovary: a clinicopathological analysis of 27 cases. Cancer 50:2438–2447
Janovski NA, Paramandlem XL (1973) Ovarian tumors. In: Major problems in obstetrics and gynecology. Saunders Co, Philadelphia
Furukawa N (2007) Solitary splenic metastasis of ovarian cancer. Arch Gynecol Obstet 275(6):499–502
García A, De la Torre J, Castellví J, Gil A, López M (2004) Ovarian metastases caused by cholangiocarcinoma: a rare Krukenberg’s tumour simulating a primary neoplasm of the ovary: a two-case study. Arch Gynecol Obstet 270(4):281–284
Studzinski Z, Zajewski W (2002) Bilateral metastatic ovarian tumors (Krukenberg’s tumors) in the course of stomach cancer. Arch Gynecol Obstet 267(2):95–97
Andresen DM, Pedersen FH, Rasmussen KL (2001) Adenocarcinoma of the small intestine mistaken as a primary ovarian cancer. Arch Gynecol Obstet 265(4):214–215
Ozsaran AA, Dikmen Y, Terek MC, Ulukus M, Ozdemir N, Orgüc S, Erhan Y (2000) Bilateral metastatic carcinoma of the breast from primary ovarian cancer. Arch Gynecol Obstet 264(3):166–167
Young RH, Path FRC, Scully RE (1994) Metastatic tumors of the ovary. In: Blausteins pathology of the female genital tract, 4th edn. Springer, Berlin
Petru E, Pickel H, Heydarfadai M, Lahousen M, Haas J, Schaider H, Tamussino K (1992) Nongenital cancers metastatic to the ovary. Gynecol Oncol 44(1):83–86
Ulbright TM, Roth LM, Stehman FB (1984) Secondary ovarian neoplasia. A clinicopathologic study of 35 cases. Cancer 53(5):1164–1174
Yakushiji M, Tazaki T, Nishimura H, Kato T (1987) Krukenberg tumors of the ovary: a clinicopathologic analysis of 112 cases. Nippon Sanka Fujinka Gakkai Zasshi 39(3):479–485
Lumb G, Mackenzie DH (1959) The incidence of metastases in adrenal glands and ovaries removed for carcinoma of the breast. Cancer 12(3):521–526
Yada-Hashimoto N, Yamamoto T, Kamiura S, Seino H, Ohira H, Sawai K, Kimura T, Saji F (2003) Metastatic ovarian tumors: a review of 64 cases. Gynecol Oncol 89(2):314–317
Fox H, Langley FA (1976) Tumors of the ovary. Heinemann, London
Webb MJ, Decker DG, Mussey E (1975) Cancer metastatic to the ovary: factors influencing survival. Obstet Gynecol 45(4):391–396
Khunamornpong S, Suprasert P, Chiangmai WN, Siriaunkgul S (2006) Metastatic tumors to the ovaries: a study of 170 cases in northern Thailand. Int J Gynecol Cancer 16(Suppl 1):132–138
Santesson L, Kottmeier HL (1968) General classification of ovarian tumors. In: Gentil F, Junqueira AC (eds) Ovarian cancer UICC monograph series, vol 11. Springer, Berlin, pp 1–8
Latour JP, Soloway I, Young MH (1956) Krukenberg tumors of the ovary. Obstet Gynecol 8:636–638
Woodruff JD, Novak ER (1960) The Krukenberg tumor: study of 48 cases from the ovarian tumor registry. Obstet Gynecol 15:351–360
Choi HJ, Lee JH, Seo SS, Lee S, Kim SK, Kim JY, Lee JS, Park SY, Kim YH (2005) Computed tomography findings of ovarian metastases from colon cancer: comparison with primary malignant ovarian tumors. J Comput Assist Tomogr 29(1):69–73
Fujiwara K, Ohishi Y, Koike H, Sawada S, Moriya T, Kohno I (1995) Clinical implications of metastases to the ovary. Gynecol Oncol 59(1):124–128
Karsh J (1951) Secondary malignant disease of the ovaries; a study of 72 autopsies. Am J Obstet Gynecol 61:154–160
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kondi-Pafiti, A., Kairi-Vasilatou, E., Iavazzo, C. et al. Metastatic neoplasms of the ovaries: a clinicopathological study of 97 cases. Arch Gynecol Obstet 284, 1283–1288 (2011). https://doi.org/10.1007/s00404-011-1847-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-011-1847-4