Abstract
Purposes
Uterine cervical carcinosarcoma (CS) is very rare. To date, only 40 cases have been reported. It seems to have a more aggressive clinical behavior than does cervical squamous cell carcinoma (SCC). The purposes of our study were to characterize the clinicopathologic characteristics and human papillomavirus (HPV) status of the rare tumor and to analyze the molecular features in cervical CS that may account for its aggressive behavior.
Methods
Three patients were diagnosed with uterine cervical CS at West China Second Hospital of Sichuan University between 1995 and 2009. Data were retrospectively analyzed from available charts and pathological reports. Twelve patients with FIGO stage Ib–IIa cervical SCC were enrolled as the controls, and the expression profiling of p53, Ki-67, bcl-2, survivin and apoptosis index between cervical CS and SCC was compared. Immunohistochemical and apoptosis results were scored separately for the carcinomatous and sarcomatous components.
Results
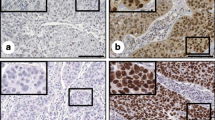
All three patients were shown to be negative for HPV infection by Hybribio HPV genoarray assay. Expression of p53 was observed in one patient in both carcinomatous and sarcomatous components in a similar proportion; in contrast, the Ki67, bcl-2 and survivin expressions were higher in carcinomatous components than in sarcomatous components in all three cases. Compared to cervical SCC, stronger immunostaining for bcl-2, survivin and lower apoptosis was observed in cervical CS.
Conclusions
Cervical CS is a peculiar tumor with many different clinicopathologic characteristics from cervical SCC. Dysregulation of apoptosis may confer tumor cells of cervical CS with survival and growth advantages, and thereby facilitate the aggressive behavior of cervical CS.




Similar content being viewed by others
References
Abell MR, Ramirez JA (1973) Sarcomas and carcinosarcomas of the uterine cervix. Cancer 31:1176–1192. doi:10.1002/1097-0142(197305)
Clement PB, Zubovits JT, Young RH, Scully RE (1998) Malignant Mullerian mixed tumors of the uterine cervix: a report of nine cases of a neoplasm with morphology often different from its counterpart in the corpus. Int J Gynecol Pathol 17:211–222
Takeshima Y, Amatya VJ, Nakayori F, Nakano T, Iwaoki Y, Daitoku K et al (2002) Co-existent carcinosarcoma and adenoid basal carcinoma of the uterine cervix and correlation with human papillomavirus infection. Int J Gynecol Pathol 21:186–190
Iida T, Yasuda M, Kajiwara H, Minematsu T, Osamura RY, Itoh J et al (2005) Case of uterine cervical carcinosarcoma. J Obstet Gynaecol Res 31:404–408
Laterza R, Seveso A, Zefiro F, Formenti G, Mellana L, Donadello N et al (2007) Carcinosarcoma of the uterine cervix: case report and discussion. Gynecol Oncol 107:98–100. doi:10.1016/j.ygyno.2007.07.038
Grayson W, Taylor LF, Cooper K (2001) Carcinosarcoma of the uterine cervix: a report of eight cases with immunohistochemical analysis and evaluation of human papillomavirus status. Am J Surg Pathol 25:338–347
Kadota K, Haba R, Ishikawa M, Kushida Y, Katsuki N, Hayashi T et al (2009) Uterine cervical carcinosarcoma with heterologous mesenchymal component: a case report and review of the literature. Arch Gynecol Obstet 280:839–843. doi:10.1007/s00404-009-1017-0
Schindl M, Schoppmann SF, Ströbel T, Heinzl H, Leisser C, Horvat R et al (2003) Level of Id-1 protein expression correlates with poor differentiation, enhanced malignant potential, and more aggressive clinical behavior of epithelial ovarian tumors. Clin Cancer Res 9:779–785
Li J, Jia H, Xie L, Wang X, He H, Lin Y et al (2009) Correlation of inhibitor of differentiation 1 expression to tumor progression, poor differentiation and aggressive behaviors in cervical carcinoma. Gynecol Oncol 114:89–93. doi:10.1016/j.ygyno.2009.03.016
Semczuk A, Skomra D, Rybojad P, Jeczen R, Rechberger T (2006) Endometrial carcinoma with pleural metastasis: a case report. Acta Cytol 50:697–700
Solanas M, Grau L, Moral R, Vela E, Escrich R, Escrich E (2009) Dietary olive oil and corn oil differentially affect experimental breast cancer through distinct modulation of the P21 ras signaling and the proliferation-apoptosis balance. Carcinogenesis (Epub ahead of print). doi:10.1093/carcin/bgp243
Zehbe I, Wilander E (1997) Human papillomavirus infection and invasive cervical neoplasia: a study of prevalence and morphology. J Pathol 181:270–275. doi:10.1002/(SICI)1096-9896(199703)181:3<270:AID-PATH767>3.0.CO;2-R
Zhivotovsky B, Orrenius S (2006) Carcinogenesis and apoptosis: paradigms and paradoxes. Carcinogenesis 27:1939–1945. doi:10.1093/carcin/bgl035
Mita AC, Mita MM, Nawrocki ST, Giles FJ (2008) Survivin: key regulator of mitosis and apoptosis and novel target for cancer therapeutics. Clin Cancer Res 14:5000–5005. doi:10.1158/1078-0432.CCR-08-0746
Costa MJ, Guinee D Jr (2000) CD34 immunohistochemistry in female genital tract carcinosarcoma (malignant mixed mullerian tumors) supports a dominant role of the carcinomatous component. Appl Immunohistochem Mol Morphol 8:293–299
Acknowledgments
We thank Professor Kai-Xuan Yang and Professor Xian-yin Yao, Department of gynecological pathology, West China Second Hospital of Sichuan University for help in pathology.
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gan, XL., Li, JK., Yu, TH. et al. High expressions of bcl-2 and survivin, and decreased apoptosis in uterine cervical carcinosarcoma compared to cervical squamous cell carcinoma. Arch Gynecol Obstet 284, 175–181 (2011). https://doi.org/10.1007/s00404-010-1610-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-010-1610-2