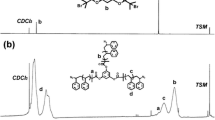
Abstract
Block copolymers consisting of blocks of poly(amino acid)s (PAAs) derived from naturally occurring l-amino acids and biocompatibilizing poly(ethylene glycol) (PEG) are of interest for biomedical applications because of their compatibility, partial degradability, and, depending on their molecular architecture, unique self-assembly behavior. PEGylated PAAs were synthesized by α-methoxy-ω-amino PEG-initiated ring-opening polymerization, ROP, of the N-carboxyanhydrides, NCAs, of l-leucine and l-glutamic acid γ-benzyl ester. The molecular weights of the resulting di- and triblock copolymers were well controlled with narrow polydispersities, and the block copolymers were obtained in high yields (>90 %). PEGylated PAA diblock copolymers with a copolymer block of PAAs were obtained when the amino acid NCAs were polymerized as a mixture and triblock copolymers with two PAA hompolymer blocks resulted from the successive addition of the respective NCAs. Dynamic light scattering indicated that these block copolymers self-assemble when exposed to a selective solvent into micelles with hydrodynamic radii between 7 and 10 nm, which aggregate into larger structures with radii ranging between 27 and 40 nm. The coexistence and sizes of the two types of particles were corroborated by small-angle neutron scattering analyses. A detailed analysis of the micelle core structures by transmission electron microscopy suggests that the cores of the micelles do not form perfect spheres and thereby enable their continued aggregation, which is further aided by the immiscibility and subsequent phase separation of poly(l-glutamic acid γ-benzyl ester) and poly(l-leucine) blocks that constitute the PAA block.





Similar content being viewed by others
References
Antonietti M, Förster S (2003) Vesicles and liposomes: a self-assembly principle beyond lipids. Adv Mater 15:1323–1333
Ahmed F, Pakunlu RI, Srinivas G, Brannan A, Bates F, Klein MI, Minko T, Discher DE (2006) Shrinkage of a rapidly growing tumor by drug-loaded polymersomes: pH-triggered release through copolymer degradation. Mol Pharm 3:340–350
Akagi T, Baba M, Akashi M (2007) Preparation nanoparticles by the self-organization of polymers consisting of hydrophobic and hydrophilic segments: potential applications. Polymer 48:6729–6747
Putnam D, Gentry CA, Pack DW, Langer R (2001) Polymer-based gene delivery with low cytotoxicity by a unique balance of side-chain termini. PNAS 98:1200–1205
Harada A, Kataoka K (2006) Supramolecular assemblies of block copolymers in aqueous media as nanocarriers relevant to biological applications. Prog Polym Sci 31:949–982
Blanazs A, Armes SP, Ryan AJ (2009) Self-assembled block copolymer aggregates: from micelles to vesicles and their biological applications. Macromol Rapid Commun 30:267–277
Loverde SM, Pantano DA, Christian DA, Mahmud A, Klein ML, Discher DE (2011) Curvature, rigidity, and pattern formation in functional polymer micelles and vesicles—from dynamic visualization to molecular simulation. Curr Opin Solid State Mater Sci 15:277–284
Rajagopal K, Mahmud A, Christian DA, Pajerowski JD, Brown AEX, Loverde SM, Discher DE (2010) Curvature-couples hydration of semicrystalline polymer amphiphiles yields flexible worm micelles but favors rigid vesicles: polycaprolactone-based block copolymers. Macromolecules 43:9736–9746
Israelachvili JN (1991) Intermolecular and surface forces, 2nd edn. Academic Press London, San Diego
Discher BM, Won Y-Y, Ege DS, Lee JC-M, Bates FS, Discher DE, Hammer DA (1999) Polymersomes: tough vesicles made from diblock copolymers. Science 284:1143–1146
Choucair A, Eisenberg A (2003) Control of amphiphilic block copolymer morphologies using solution conditions. Eur Phys J E 10:37–44
Kwon GS, Kataoka K (1995) Block copolymer micelles as long-circulating drug vehicles. Adv Drug Deliv Rev 16:295–309
Barz M, Duro Castano A, Vicent MJ (2013) A versatile post-polymerization modification method for polyglutamic acid: synthesis of orthogonal reactive polyglutamates and their use in click chemistry. Polym Chem 4:2989–2994
Lee HJ, Bae Y (2011) Cross-linked nanoassemblies from poly(ethylene glycol)-poly(aspartate) block copolymers as stable supramolecular templates for particulate drug delivery. Biomacromolecules 12:2686–2696
Giammona G, Cavallaro G, Pitarresi G, Pedone E (2000) Novel polyaminoacidic copolymers as nonviral gene vectors. Colloid Polym Sci 278:69–73
Byrne M, Thornton PD, Cryan SA, Heise A (2012) Star polypeptides by NCA polymerisation from dendritic initiators: synthesis and enzyme controlled payload release. Polym Chem 3:2825–2831
Rosu C, Selcuk S, Soto-Cantu E, Russo PS (2014) Progress in silica polypeptide composite colloidal hybrids: from silica cores to fuzzy shells. Colloid Polym Sci 292:1009–1040
Jeong SY, Moon HJ, Park MH, Joo MK, Jeong B (2012) Molecular captain: a light-sensitive linker molecule in poly(ethylene glycol)-poly(L-alanine-poly(ethylene glycol) triblock copolymer directs molecular nano-assembly, conformation and sol-gel transition. J Polym Sci 50:3184–3191
Song B, Song J, Zhang S, Anderson MA, Ao Y, Yang CY, Deming TJ, Sofroniew MV (2012) Sustained local delivery of bioactive nerve growth factor in the central nervous system via tunable diblock copolypeptide hydrogel depots. Biomaterials 33:9105–9116
Yang C-Y, Song B, Ao Y, Nowak AP, Abelowitz RB, Korsak RA, Havton LA, Deming TJ, Sofroniew MV (2009) Biocompatibility of amphiphilic diblock copolypeptide hydrogels in the central nervous system. Biomaterials 30:2881–2898
Hehir S, Cameron NR (2014) Recent advances in drug delivery systems based on polypeptides prepared from N-carboxyanhydrides. Polym Int 63:943–954
Ding J, Shi F, Xiao C, Lin L, Chen L, He C, Zhuang X, Chen X (2011) One-step preparation of reduction-responsive poly(ethylene glycol)-poly(amino acid)s nanogels as efficient intracellular drug delivery platforms. Polym Chem 2:2857
Breedveld V, Nowak AP, Sato J, Deming TJ, Pine DJ (2004) Rheology of block copolypeptide solutions: hydrogels with tunable properties. Macromolecules 37:3943–3953
Sgouras D, Duncan R (1990) Methods for the evaluation of biocompatibility of soluble synthetic polymers which have potential for biomedical use. 1. Use of the tretazolium based colorimetric assay (MTT) as a preliminary screen for evaluation of in vitro cytotoxicity. J Mater Sci 1:61–68
Harris, MJ (1992) Poly(ethylene glycol) chemistry, biotechnical and biomedical applications. In: Harris JM (ed) Topics in applied chemistry, Plenum Press, New York, pp 1–14
Abuchowski A, van Es T, Palczuk NC, Davis FF (1977) Alteration of immunological properties of bovine serum albumin by covalent attachment of polyethylene glycol. J Biol Chem 252:3578–3581
Abuchowski A, McCoy R, Palczuk NC, van Es T, Davis FF (1977) Effect of covalent attachment of polyethylene glycol on immunogenicity and circulating life of bovine liver catalase. J Biol Chem 252:3582–3586
Cabral H, Kataoka K (2014) Progress of drug-loaded polymeric micelles into clinical studies. J Control Release 190:465–476
Knop K, Hoogenboom R, Fisher D, Schubert US (2010) Poly(ethylene glycol) in drug delivery: pros and cons as well as potential alternatives. Angew Chem Int Ed 49:6288–6308
Greenwald RB (2001) PEG drugs: an overview. J Control Release 74:159–171
Gon S, Fang B, Santore MM (2011) Interaction of cationic proteins and polypeptides with biocompatible cationically-anchored PEG brushes. Macromolecules 44:8161–8168
Moghimi S, Hunter A, Murray J (2001) Long-circulating and target-specific nanoparticles: theory to practice. Pharmacol Rev 53:283–318
Obeid R, Armstrong T, Peng X, Busse K, Kressler J, Scholz C (2014) The behavior of poly(amino acids) containing L-cysteine and their block copolymers with poly(ethylene glycol) on gold surfaces. J Polym Sci A1(52):248–257
Obeid R, Scholz C (2011) Synthesis and self-assembly of well-defined Poly(amino acid) end capped Poly(ethylene glycol) and Poly(2-methyl-2-oxazoline). Biomacromolecules 12:3797–3804
Yokohama M, Kwon GS, Okano T, Sakurai Y, Sato T, Kataoka K (1992) Preparation of micelle-forming polymer-drug conjugates. Bioconjug Chem 3:295–301
Kataoka K, Kwon GS, Yokohama M, Okano T, Sakurai Y (1993) Block copolymer micelles as vehicles for drug delivery. J Control Release 24:119–132
Deng J, Gao N, Wang Y, Yi H, Fang S, Ma Y, Cai L (2012) Self-assembled cationic micelles based on PEG-PLL-PLLeu hybrid polypeptides as highly effective gene vectors. Biomacromolecules 13:3795–3804
Guenther M, Gerlach G, Wallmersperger T, Avula MN, Cho SH, Xie X, Devener BV, Solzbacher F, Tathireddy P, Magda JJ, Scholz C, Obeid R, Armstrong T (2013) Smart hydrogel-based biochemical microsensor array for medical diagnostics. Adv Sci Technol 85:47–52
Armstrong T, Scholz C (2012) Investigation of the secondary structure of oligomeric poly(amino acid)s. Polym Prepr 53(1):600–601
Lee S, Spencer ND (2008) Adsorption properties of poly(L-lysine)-graft-poly(ethylene glycol) (PLL-g-PEG) at a hydrophobic interface: influence of tribological stress, pH, salt concentration, and polymer molecular weight. Langmuir 24:9479–9488
Scholz C (2007) Perspectives on: material aspects of retinal prostheses. J Bioact Compat Polym 22:539–568
Hell SW, Wichmann J (1994) Breaking the diffraction resolution limit by stimulated emission: stimulated-emission-depletion fluorescence microscopy. Opt Lett 19:780–782
Horkay F, Hammounda B (2008) Small angle neutron scatttering form typical synthetic and biopolymer solutions. Colloid Polym Sci 286:611–620
Daly WH, Poche D (1988) The preparation of N-carboxyanhydrides of α-amino acids using bis(trichloromethyl)carbonate. Tetrahedron Lett 29:5859–5862
Kricheldorf HR (2006) Polypeptides and 100 years of chemistry of a-amino acid N-carboxyanhydrides. Angew Chem Int Ed 45:5752–5784
Zou J, Fan J, He X, Zhang S, Wang H, Wooley KL (2013) A facile glovebox-free strategy to significantly accelerate the syntheses of well-defined polypeptides by N‑carboxyanhydride (NCA) ring-opening polymerizations. Macromolecules 46:4223–4226
Kricheldorf HR (1987) In: α-Amino acid-N-Carboxy-Anhydrides and related heterocycles, Springer, Berlin
Ulkoski D, Armstrong T, Scholz C (2013) Investigating the secondary structure of oligomeric poly(amino acid)s. In: Tailored polymer architectures for pharmaceutical and biomedical applications (ed.: C. Scholz, J. Kressler). ACS Symp Ser 1135:69–85
Ainavarapu SRK, Brujić J, Huang HH, Wiita AP, Lu H, Li L, Walther KA, Carrion-Vazquez M, Li H, Fernandez JM (2007) Contour length and refolding rate of a small protein controlled by engineered disulfide bonds. Biophys J 92:225–233
Lee H, Venable RM, MacKerell AD Jr, Pastor RW (2008) Molecular dynamics studies of polyethylene oxide and polyethylene glycol: hydrodynamic radius and shape anisotropy. Biophys J 95:1590–1599
Lee H, deVries A, Marrink SJ, Pastor RW (2009) A coarse-grained model for polyethylene oxide and polyethylene glycol: conformation and hydrodynamics. J Phys Chem B 113:13186–13194
Devanand K, Selser JC (1991) Asymptotic behavior and long-range interactions in aqueous solutions of poly(ethylene oxide). Macromolecules 24:5943–5947
Kyeremateng SO, Busse K, Kohlbrecher J, Kressler J (2011) Synthesis and self-organization of poly(propylene oxide)-based amphiphilic and triphilic block copolymers. Macromolecules 44:583–593
Gu PF, Xu H, Sui BW, Gou JX, Meng LK, Sun F, Wang XJ, Qi N, Zhang Y, He HB, Tang X (2012) Polymeric micelles based on poly(ethylene glycol) block poly(racemic amino acids) hybrid polypeptides: conformation-facilitated drug-loading behavior and potential application as effective anticancer drug carriers. Int J Nanomed 7:109–122
Trent JS (1984) Ruthenium tetraoxide staining of polymers: new preparative methods for electron microscopy. Macromolecules 17:2930–2931
Letchford K, Burt H (2007) A review of the formation and classification of amphiphilic block copolymer nanoparticulate structures: micelles, nanospheres, nanocapsules and polymersomes. Eur J Pharm Biopharm 65:259–269
Asada T, Muramatsu H, Watanabe R, Onogi S (1980) Rheooptical studies of racemic poly(γ-benzyl glutamate) liquid crystals. Macromolecules 13:867–871
Acknowledgments
The authors would like to thank Dr. Donghui Zhang and her research group at LSU Baton Rouge for assistance with the GPC measurements. The authors also acknowledge the Oakridge Associated Universities program, which allowed for access to the Spallation Neutron Source, at Oakridge National Laboratory. JK thanks the Deutsche Forschungsgemeinschaft for financial support (FOR 1145).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 575 kb)
Rights and permissions
About this article
Cite this article
Ulkoski, D., Meister, A., Busse, K. et al. Synthesis and structure formation of block copolymers of poly(ethylene glycol) with homopolymers and copolymers of l-glutamic acid γ-benzyl ester and l-leucine in water. Colloid Polym Sci 293, 2147–2155 (2015). https://doi.org/10.1007/s00396-015-3632-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00396-015-3632-6