Abstract
Background
Retinoids play a key role in lung development. Retinoid signaling pathway has been shown to be disrupted in the nitrofen model of congenital diaphragmatic hernia (CDH) but the exact mechanism is not clearly understood. Retinol-binding protein (RBP) and transthyretin (TTR) are transport proteins for delivery of retinol to the tissues via circulation. Previous studies have shown that pulmonary retinol levels are decreased during lung morphogenesis in the nitrofen CDH model. In human newborns with CDH, both retinol and RBP levels are decreased. It has been reported that maternal RBP does not cross the placenta and the fetus produces its own RBP by trophoblast. RBP and TTR synthesized in the fetus are essential for retinol transport to the developing organs including lung morphogenesis. We hypothesized that nitrofen interferes with the trophoblastic expression of RBP and TTR during lung morphogenesis and designed this study to examine the trophoblastic expression of RBP and TTR, and the total level of RBP and TTR in the lung in the nitrofen model of CDH.
Methods
Pregnant rats were exposed to either olive oil or nitrofen on day 9 of gestation (D9). Fetal lungs and placenta harvested on D21 and divided into two groups: control (n = 8) and nitrofen with CDH (n = 8). Total lung RBP and TTR levels using protein extraction were compared with enzyme linked immunoassay (ELISA). Immunohistochemistry was performed to evaluate trophoblastic RBP and TTR expression.
Results
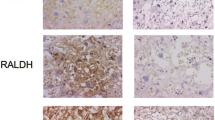
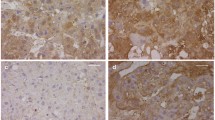
Total protein levels of lung RBP and TTR were significantly lower in CDH (0.26 ± 0.003 and 6.4 ± 0.5 μg/mL) compared with controls (0.4 ± 0.001 and 9.9 ± 1.6 μg/mL, p < 0.05). In the control group, immunohistochemical staining showed strong immunoreactivity of RBP and TTR in the trophoblast compared to CDH group.
Conclusions
Decreased trophoblast expression of retinol transport proteins suggest that nitrofen may interfere with the fetal retinol transport resulting in reduced pulmonary RBP and TTR levels and causing pulmonary hypoplasia in CDH.


Similar content being viewed by others
References
Colvin J, Bower C, Dickinson JE et al (2005) Outcomes of congenital diaphragmatic hernia: a population-based study in Western Australia. Pediatrics 116:e356–e363
Stege G, Fenton A, Jaffray B (2003) Nihilism in the 1990 s: the true mortality of congenital diaphragmatic hernia. Pediatrics 112:532–535
Gosche JR, Islam S, Boulanger SC (2005) Congenital diaphragmatic hernia: searching for answers. Am J Surg 190:324–332
Robinson PD, Fitzgerald DA (2007) Congenital diaphragmatic hernia. Paediatr Respir Rev 8:323–334 (quiz 334–325)
Montedonico S, Nakazawa N, Puri P (2008) Congenital diaphragmatic hernia and retinoids: searching for an etiology. Pediatr Surg Int 24:755–761
Noble BR, Babiuk RP, Clugston RD et al (2007) Mechanisms of action of the congenital diaphragmatic hernia-inducing teratogen nitrofen. Am J Physiol Lung Cell Mol Physiol 293:L1079–L1087
Clagett-Dame M, DeLuca HF (2002) The role of vitamin A in mammalian reproduction and embryonic development. Annu Rev Nutr 22:347–381
Beurskens LW, Tibboel D, Lindemans J et al (2010) Retinol status of newborn infants is associated with congenital diaphragmatic hernia. Pediatrics 126:712–720
Gallot D, Marceau G, Coste K et al (2005) Congenital diaphragmatic hernia: a retinoid-signaling pathway disruption during lung development? Birth Defects Res A Clin Mol Teratol 73:523–531
Nakazawa N, Montedonico S, Takayasu H et al (2007) Disturbance of retinol transportation causes nitrofen-induced hypoplastic lung. J Pediatr Surg 42:345–349
Ruttenstock E, Doi T, Dingemann J et al (2011) Prenatal administration of retinoic acid upregulates insulin-like growth factor receptors in the nitrofen-induced hypoplastic lung. Birth Defects Res B Dev Reprod Toxicol 92:148–151
Quadro L, Hamberger L, Gottesman ME et al (2005) Pathways of vitamin A delivery to the embryo: insights from a new tunable model of embryonic vitamin A deficiency. Endocrinology 146:4479–4490
Monaco HL, Rizzi M, Coda A (1995) Structure of a complex of two plasma proteins: transthyretin and retinol-binding protein. Science 268:1039–1041
Quadro L, Hamberger L, Colantuoni V et al (2003) Understanding the physiological role of retinol-binding protein in vitamin A metabolism using transgenic and knockout mouse models. Mol Aspects Med 24:421–430
Quadro L, Hamberger L, Gottesman ME et al (2004) Transplacental delivery of retinoid: the role of retinol-binding protein and lipoprotein retinyl ester. Am J Physiol Endocrinol Metab 286:E844–E851
Morriss-Kay GM, Ward SJ (1999) Retinoids and mammalian development. Int Rev Cytol 188:73–131
Sapin V, Ward SJ, Bronner S et al (1997) Differential expression of transcripts encoding retinoid binding proteins and retinoic acid receptors during placentation of the mouse. Dev Dyn 208:199–210
Sapin V, Chaib S, Blanchon L et al (2000) Esterification of vitamin A by the human placenta involves villous mesenchymal fibroblasts. Pediatr Res 48:565–572
Quadro L, Blaner WS, Salchow DJ et al (1999) Impaired retinal function and vitamin A availability in mice lacking retinol-binding protein. EMBO J 18:4633–4644
Episkopou V, Maeda S, Nishiguchi S et al (1993) Disruption of the transthyretin gene results in mice with depressed levels of plasma retinol and thyroid hormone. Proc Natl Acad Sci USA 90:2375–2379
Wei S, Episkopou V, Piantedosi R et al (1995) Studies on the metabolism of retinol and retinol-binding protein in transthyretin-deficient mice produced by homologous recombination. J Biol Chem 270:866–870
Kling DE, Cavicchio AJ, Sollinger CA et al (2010) Nitrofen induces apoptosis independently of retinaldehyde dehydrogenase (RALDH) inhibition. Birth Defects Res B Dev Reprod Toxicol 89:223–232
Quadro L, Blaner WS, Hamberger L et al (2004) The role of extrahepatic retinol binding protein in the mobilization of retinoid stores. J Lipid Res 45:1975–1982
van Bennekum AM, Wei S, Gamble MV et al (2001) Biochemical basis for depressed serum retinol levels in transthyretin-deficient mice. J Biol Chem 276:1107–1113
Montedonico S, Nakazawa N, Puri P (2006) Retinoic acid rescues lung hypoplasia in nitrofen-induced hypoplastic foetal rat lung explants. Pediatr Surg Int 22:2–8
Guilbert TW, Gebb SA, Shannon JM (2000) Lung hypoplasia in the nitrofen model of congenital diaphragmatic hernia occurs early in development. Am J Physiol Lung Cell Mol Physiol 279:L1159–L1171
Jay PY, Bielinska M, Erlich JM et al (2007) Impaired mesenchymal cell function in Gata4 mutant mice leads to diaphragmatic hernias and primary lung defects. Dev Biol 301:602–614
Doi T, Hajduk P, Puri P (2009) Upregulation of Slit-2 and Slit-3 gene expressions in the nitrofen-induced hypoplastic lung. J Pediatr Surg 44:2092–2095
Doi T, Puri P (2009) Up-regulation of Wnt5a gene expression in the nitrofen-induced hypoplastic lung. J Pediatr Surg 44:2302–2306
Tong QS, Zheng LD, Tang ST et al (2007) Nitrofen suppresses cell proliferation and promotes mitochondria-mediated apoptosis in type II pneumocytes. Acta Pharmacol Sin 28:672–684
Clugston RD, Zhang W, Greer JJ (2010) Early development of the primordial mammalian diaphragm and cellular mechanisms of nitrofen-induced congenital diaphragmatic hernia. Birth Defects Res A Clin Mol Teratol 88:15–24
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kutasy, B., Gosemann, J.H., Doi, T. et al. Nitrofen interferes with trophoblastic expression of retinol-binding protein and transthyretin during lung morphogenesis in the nitrofen-induced congenital diaphragmatic hernia model. Pediatr Surg Int 28, 143–148 (2012). https://doi.org/10.1007/s00383-011-2995-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-011-2995-0