Abstract
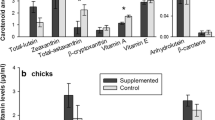
Carotenoids, as pigments with antioxidant and immunoregulatory properties, play a crucial role in developing chicks. Carotenoids must be acquired through diet and are relatively scarce, suggesting that their availability is a limiting factor leading to a trade-off between colour displays and physiological functions. However, potential differences in this trade-off between male and female chicks have been little studied. We manipulated carotenoid availability in 9 days old common tern Sterna hirundo chicks by supplementing their fish diet with four carotenoids during 9 days. Our aim was to examine sex-specific responses to the experimental increase of dietary carotenoids on plasma circulation, physiological and condition variables and successful fledging. Furthermore, to explore the functional and evolutionary basis of the trade-off, we studied the relationships among carotenoid concentration, mediated immune response and foot colouration. After treatment, control chicks showed decreasing plasma levels for most carotenoid types, whereas supplemented chicks had strong increases. Colour luminosity and saturation increased in both treatment groups, while hue only changed significantly towards redder feet in supplemented females. Supplemented chicks presented neither different T-cell-mediated immunity nor other differences compared to control chicks. Nevertheless, supplemented females showed tendencies towards decreased immune responses and increased δ15N signatures, and supplemented males towards greater body mass. Our results indicate colouration may have, in females, a signalling function as to compensate for immunological costs. In males, additional availability of carotenoids may contribute to improve the body condition. This study suggests that trade-off responses to carotenoid availability are sex-specific in tern chicks. Thus, parental carotenoid supply to chicks may be an unrecognised component in sex allocation.




Similar content being viewed by others
References
Alonso-Álvarez C, Bertrand S, Devevey G, Gaillard M, Prost J, Faivre B, Sorci G (2004) An experimental test of the dose dependent effect of carotenoids and immune activation on sexual signals and antioxidant activity. Am Nat 164:651–659
Badyaev AV, Seaman DA, Navara KJ, Hill GE, Mendonça MT (2006) Evolution of sex-biased maternal effects in birds. III. Adjustment of ovulation order can enable sex-specific allocation of hormones, carotenoids, and vitamins. J Evol Biol 19:1044–1057
Becker PH (1998) Langzeittrends des Bruterfolgs der Flußseeschwalbe Sterna hirundo und seiner Einflußgrößen im Wattenmeer. Vogelwelt 119:223–234
Becker PH, Ludwigs JD (2004) Sterna hirundo Common Tern. In: Parkin D (ed) BWP update vol 6 nos 1/2. Oxford University Press, London, pp 93–139
Becker PH, Wendeln H (1997) A new application for transponders in population ecology of the Common Tern. Condor 99:534–538
Becker PH, Wink M (2003) Influences of sex, sex composition of brood and hatching order on mass growth in Common Terns Sterna hirundo. Behav Ecol Sociobiol 54:136–146
Becker PH, Wendeln H, González-Solís J (2001) Population dynamics, recruitment, individual quality and reproductive strategies in common terns Sterna hirundo marked with transponders. Ardea 89(Special issue):241–252
Berthouly A, Helfenstein F, Tanner M, Richner H (2008) Sex-related effects of maternal egg investment on offspring in relation to carotenoid availability in the great tit. J Anim Ecol 77:74–82
Blas J, Pérez-Rodriguez L, Bortolotti GR, Vinuela J, Marchant TA (2006) Testosterone increases bioavailability of carotenoids: insights into the honesty of sexual signaling. Proc Natl Acad Sci USA 103:18633–18637
Blount JD, Surai PF, Nager RG, Houston DC, Møller AP, Trewby ML, Kennedy MW (2002) Carotenoids and egg quality in the lesser black-backed gull (Larus fuscus): a supplemental feeding study of maternal effects. Proc R Soc Lond B 269:29–36
Cabana G, Rasmussen JB (1994) Modelling food chain structure and contaminant bioaccumulation using stable nitrogen isotopes. Nature 372:255–257
Casagrande S, Costantini D, Fanfani A, Tagliavini J, Dell’Omo G (2007) Patterns of serum carotenoid accumulation and skin color variation in kestrel nestlings in relation to breeding conditions and different terms of carotenoid supplementation. J Comp Physiol B 177:237–245
Costantini D, Møller AP (2008) Carotenoids are minor antioxidants for birds. Funct Ecol 22:367–370
Costantini D, Fanfani A, Dell’Omo G (2007) Carotenoid availability does not limit the capability of nestling kestrels (Falco tinnunculus) to cope with oxidative stress. J Exp Biol 210:1238–1244
Cucco M, Guasco B, Malacarne G, Ottonelli R (2006) Effects of β-carotene supplementation on chick growth, immune status and behaviour in the grey partridge, Perdix perdix. Behav Process 73:325–332
de Ayala RM, Martinelli R, Saino N (2006) Vitamin E supplementation enhances growth and condition of nestling barn swallows (Hirundo rustica). Behav Ecol Sociobiol 60:619–630
de Ayala RM, Saino N, Møller AP, Anselmi C (2007) Mouth coloration of nestlings covaries with offspring quality and influences parental feeding behavior. Behav Ecol 18:526–534
Forero MG, Tella JL, Hobson KA, Bertellotti M, Blanco G (2002) Conspecific food competition explains variability in colony size: a test in Magellanic Penguins. Ecology 83:3466–3475
Frank D (1992) The influence of feeding conditions on food provisioning of chicks in Common Terns Sterna hirundo nesting in the German Wadden Sea. Ardea 80:45–55
Goodwin TW (1984) The biochemistry of the carotenoids. Volume II. Animals. Chapman & Hall, London
Goodwin TW (1986) Metabolism, nutrition, and function of carotenoids. Annu Rev Nutr 6:273–297
Grether GF, Hudon J, Millie DF (1999) Carotenoid limitation of sexual coloration along an environmental gradient in guppies. Proc R Soc Lond B 266:1317–1322
Griggio M, Morosinotto C, Pilastro A (2009) Nestlings’ carotenoid feather ornament affects parental allocation strategy and reduces maternal survival. J Evol Biol 22:2077–2085
Hill GE (1991) Plumage coloration is a sexually selected indicator of male quality. Nature 350:337–339
Hill GE (1992) Proximate basis of variation in carotenoid pigmentation in male house finches. Auk 109:1–12
Hobson KA, Clark RG (1992) Assessing avian diets using stable isotopes. II. Factors influencing diet-tissue fractionation. Condor 94:187–195
Hobson KA, Clark RG (1993) Turnover of 13C in cellular and plasma fractions of blood: implications for nondestructive sampling in avian dietary studies. Auk 110:638–641
Hudon J, Brush AH (1990) Identification of carotenoid pigments in birds. Methods Enzymol 213:312–321
Kilner RM (2006) Function and evolution of color in young birds. In: Hill GE, McGraw KJ (eds) Bird coloration. Volume II. Function and evolution. Harvard University Press, London, pp 201–232
Kristiansen KO, Bustnes JO, Folstad I, Helberg M (2006) Carotenoid coloration in great black-backed gull Larus marinus reflects individual quality. J Avian Biol 37:6–12
Lessells CM, Boag PT (1987) Unrepeatable repeatabilities: a common mistake. Auk 104:116–121
McGraw KJ, Gregory AJ, Parker RS, Adkins-Regan E (2003) Diet, plasma carotenoids, and sexual coloration in the zebra finch (Taeniopygia guttata). Auk 120:400–410
McGraw KJ, Adkins-Regan E, Parker RS (2005) Maternally derived carotenoid pigments affect offspring survival, sex ratio, and sexual attractiveness in a colorful songbird. Naturwissenschaften 92:375–380
McGraw KJ, Tourville EA, Butler MW (2008) A quantitative comparison of the commonly used methods for extracting carotenoids from avian plasma. Behav Ecol Sociobiol 62:1991–2002
Møller AP, Biard C, Blount JD, Houston DC, Ninni P, Saino N, Surai PF (2000) Carotenoid-dependent signals: indicators of foraging efficiency, immunocompetence or detoxification ability? Avian Poult Biol Rev 11:137–159
Møller AP, Flensted-Jensen E, Mardal W (2007) Black beak tip coloration as a signal of phenotypic quality in a migratory seabird. Behav Ecol Sociobiol 61:1561–1571
Neubauer W (1978) Experimentelle Untersuchungen zur akustischen und visuellen Kommunikation an der Flußseeschwalbe (Sterna hirundo L.) unter besonderer Berücksichtigung der Jungenaufzucht. Dissertation, Humboldt-Universität, Berlin
Olson VA, Owens IPF (2005) Interspecific variation in the use of carotenoid-based coloration in birds: diet, life history and phylogeny. J Evol Biol 18:1534–1546
Romano M, Caprioli M, Ambrosini R, Rubolini D, Fasola M, Saino N (2008) Maternal allocation strategies and differential effects of yolk carotenoids on the phenotype and viability of yellow-legged gull (Larus michahellis) chicks in relation to sex and laying order. J Evol Biol 21:1626–1640
Saino N, Ninni P, Calza S, Martinelli R, de Bernardi F, Møller AP (2000) Better red than dead: carotenoid-based mouth coloration reveals infection in barn swallow nestlings. Proc R Soc Lond B Biol Sci 267:57–61
Saino N, Romano M, Ferrari RP, Martinelli R, Møller AP (2003) Maternal antibodies but not carotenoids in barn swallow eggs covary with embryo sex. J Evol Biol 16:516–522
Saino N, Bertacche V, Bonisoli Alquati A, Romano M, Rubolini D (2008) Phenotypic correlates of yolk and plasma carotenoid concentration in yellow-legged gull chicks. Physiol Biochem Zool 81:211–225
Smits JE, Bortolotti GR, Tella JL (1999) Simplifying the phytohaemagglutinin skin-testing technique in studies of avian immunocompetence. Funct Ecol 13:567–572
Stradi R (1998) The colour of flight: carotenoids in bird plumage. Solei Gruppo Editoriale Informatico, Milan
Surai PF (2002) Natural antioxidants in avian nutrition and reproduction. Nottingham University Press, Nottingham
Surai PF, Speake BK (1998) Distribution of carotenoids from the yolk to the tissues of the chick embryo. J Nutr Biochem 9:645–651
Surai PF, Speake BK, Wood NAR, Blount JD, Bortolotti GR, Sparks NHC (2001) Carotenoid discrimination by the avian embryo: a lesson from wild birds. Comp Biochem Physiol B 128:743–750
Tschirren B, Fitze PS, Richner H (2005) Carotenoid-based nestling colouration and parental favouritism in the great tit. Oecologia 143:477–482
Velando A, Beamonte-Barrientos R, Torres R (2006) Pigment-based skin colour in the blue-footed booby: an honest signal of current condition used by females to adjust reproductive investment. Oecologia 149:535–542
Verboven N, Evans NP, D’Alba L, Nager RG, Blount JD, Surai PF, Monaghan P (2005) Intra-specific interactions influence egg composition in the lesser-black gull (Larus fuscus). Behav Ecol Sociobiol 57:357–365
Wagener M (1998) Praktische Hinweise für brutbiologische Untersuchungen an der Flußseeschwalbe Sterna hirundo. Vogelwelt 119:279–286
Zuk M, McKean KA (1996) Sex differences in parasite infections: patterns and processes. Int J Parasitol 26:1009–1023
Acknowledgments
We thank the “Banter See team” in 2006, especially J. Sprenger. We are also grateful to J.M. Vilanova (Kemin Iberica) and R. Tena (DSM Nutritional Products) for kindly supplying dietary carotenoid supplements free of charges; to the Research Centre TERRAMARE, U. Pijanowska, and F. Bairlein for providing logistical and technical support, and to B. Metzger for invaluable help during the whole process. M.M.B. was supported by a postgraduate grant of the Generalitat de Catalunya (Spain) and J.G-S. was supported by a contract of the Program Ramon y Cajal funded by the Ministerio de Educación y Ciencia and Fondos FEDER. This project was done under licence of LAVES, Oldenburg, of the Stadt Wilhelmshaven and financially supported by the Deutsche Forschungsgemeinschaft (BE 916/8-2).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Benito, M.M., González-Solís, J. & Becker, P.H. Carotenoid supplementation and sex-specific trade-offs between colouration and condition in common tern chicks. J Comp Physiol B 181, 539–549 (2011). https://doi.org/10.1007/s00360-010-0537-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-010-0537-z