Abstract
Purpose
To investigate the expression profiles of 86 miRNAs in paclitaxel-resistant prostate cancer cell lines and to identify the genes that have a role in the development of drug resistance.
Methods
Three prostate cancer cell lines, androgen-dependent VCaP, androgen-independent PC-3 and DU-145, were used to obtain paclitaxel-resistant cells by progressively increasing the concentration of paclitaxel in the culture medium. Viability assays with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium and sulforhodamine B were used to assess the cell resistance level and cytotoxic effects of paclitaxel treatment. Total RNA was isolated from both prostate cancer cell lines and their resistant versions, and cDNA samples were reverse transcribed from total RNA. Selected target genes of miRNAs that showed differences in expression and were estimated to be effective on drug resistance mechanism were analyzed with western blot analysis.
Results
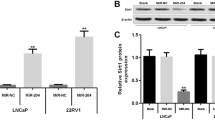
Expression study of 86 miRNAs by RT-PCR demonstrated that several of the miRNAs were expressed at different levels in paclitaxel-resistant cells compared to wild-type cells. Moreover, the expression profiles of these miRNAs varied among different prostate cancer cell line types, with 13 miRNAs being up-regulated in the resistant cells. Among these, miR-200b-3p, miR-34b-3p and miR-375 exhibited a marked up-regulation. Further, miR-100-5p showed a prominent increase in paclitaxel-resistant VCaP-R and DU145-R cells. Western blot and RT-PCR studies showed that only the LARP1 and CCND1 genes were over-expressed up to 2–5 times in all paclitaxel-resistant cell lines compared to the other investigated genes.
Conclusions
In this study, the three paclitaxel-resistant prostate cancer cell lines examined showed remarkably different miRNA expression profiles.



Similar content being viewed by others
References
Witte JS (2009) Prostate cancer genomics: towards a new understanding. Nat Rev Genet 10:77–82
Lee RC, Feinbaum RL, Ambros V (1993) The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75:843–854
Filipowicz W, Bhattacharyya SN, Sonenberg N (2008) Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 9:102–114
Kasinski AL, Slack FJ (2011) MicroRNAs en route to the clinic: progress in validating and targeting microRNAs for cancer therapy. Nat Rev Cancer 11:849–864
Schiff PB, Horwitz SB (1980) Taxol stabilizes microtubules in mouse fibroblast cells. Proc Natl Acad Sci USA 77:1561–1565
Li Y, Zeng Y, Mooney SM, Yin B, Mizokami A, Namiki M, Getzenberg RH (2011) Resistance to paclitaxel increases the sensitivity to other microenvironmental stresses in prostate cancer cells. J Cell Biochem 112(8):2125–2137
Cohen SM, Brennecke J, Stark A (2006) Denoising feedback loops by thresholding—a new role for microRNAs. Genes Dev 20:2769–2772
Fujita Y, Kojima K, Hamada N, Ohhashi R, Akao Y, Nozawa Y, Deguchi T, Ito M (2008) Effects of miR-34a on cell growth and chemoresistance in prostate cancer PC3 cells. Biochem Biophys Res Commun 377(1):114–119
Fujita Y, Kojima K, Ohhashi R, Hamada N, Nozawa Y, Kitamoto A, Sato A, Kondo S, Kojima T, Deguchi T, Ito M (2010) MiR-148a attenuates paclitaxel resistance of hormone-refractory, drug-resistant prostate cancer PC3 cells by regulating MSK1 expression. J Biol Chem 285(25):19076–19084
Puhr M, Hoefer J, Schäfer G, Erb HH, Oh SJ, Klocker H, Heidegger I, Neuwirt H, Culig Z (2012) Epithelial-to-mesenchymal transition leads to docetaxel resistance in prostate cancer and is mediated by reduced expression of miR-200c and miR-205. Am J Pathol 181(6):2188–2201
Fujita Y, Kojima T, Kawakami K, Mizutani K, Kato T, Deguchi T, Ito M (2015) miR-130a activates apoptotic signaling through activation of caspase-8 in taxane-resistant prostate cancer cells. Prostate 75(14):1568–1578
Shi G-H, Ye D-W, Yao X-D, Zhang S-L, Dai B, Zhang H-L, Shen Y-J, Zhu Y, Zhu Y-P, Xiao W-J, Ma C-G (2010) Involvement of microRNA-21 in mediating chemo-resistance to docetaxel in androgen-independent prostate cancer PC3 cells. Acta Pharmacol Sin 31(7):867–873
Lobert VH, Bruun J, Abrahamsen H, Lothe RA, Stenmark H, Kolberg M, Campsteijn C (2013) Antibody crossreactivity between the tumour suppressor PHLPP1 and the proto-oncogene β-catenin. EMBO Rep 14:10–11
Geney R, Ungureanu LM, Li D, Ojima I (2002) Overcoming multidrug resistance in taxane chemotherapy. Clin Chem Lab Med 40(9):918–925
Patel N, Chatterjee SK, Vrbanac V, Chung I, Chunyao JM, Olsen RR, Waghorne C, Zetter BR (2010) Rescue of paclitaxel sensitivity by repression of Prohibitin1 in drug-resistant cancer cells. Proc Natl Acad Sci USA 107(6):2503–2508
Leite KRM, Morais DR, Reis ST, Viana N, Moura C, Florez MG, Silva IA, Dip N, Srougi M (2013) MicroRNA 100: a context dependent miRNA in prostate cancer. Clinics 68(6):797–802
Costa-Pinheiro P, Ramalho-Carvalho J, Vieira FQ, Torres-Ferreira J, Oliveira J, Goncalves CS, Costa BM, Henrique R, Jeronimo C (2015) MicroRNA-375 plays a dual role in prostate carcinogenesis. Clin Epigenet 7(1):42–55
Majid S, Dar AA, Saini S, Shahryari V, Arora S, Zaman MS, Chang I, Yamamura S, Tanaka Y, Chiyomaru T, Deng G, Dahiya R (2013) miRNA-34b inhibits prostate cancer through demethylation, active chromatin modifications, and AKT pathways. Clin Cancer Res 19(1):73–84
Korpal M, Lee ES, Hu G, Kang Y (2008) The miR-200 family inhibits epithelial-mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem 283(22):14910–14914
Feng B, Wang R, Chen L-B (2012) Review of MiR-200b and cancer chemosensitivity. Biomed Pharmacother 66:397–402
Hayashita Y, Osada H, Tatematsu Y, Yamada H, Yanagisawa K, Tomida S, Yatabe Y, Kawahara K, Sekido Y, Takahashi T (2005) A polycistronic MicroRNA cluster, miR-17-92, Is overexpressed in human lung cancers and enhances cell proliferation. Cancer Res 65(21):9628–9632
Zhang J, Song Y, Zhang C, Zhi X, Fu H, Ma Y, Chen Y, Pan F, Wang K, Ni J, Jin W, He X, Su H, Cui D (2016) Circulating MiR-16-5p and MiR-19b-3p as two novel potential biomarkers to indicate progression of gastric cancer. Theranostics 5(7):733–745
Tanaka M, Oikawa K, Takanashi M, Kudo M, Ohyashiki J, Ohyashiki K, Kuroda M (2009) Down-regulation of miR-92 in human plasma is a novel marker for acute leukemia patients. PLoS One 4(5):e5532–e5535
Wang Y, Sun B, Sun H, Zhao X, Wang X, Zhao N, Zhang Y, Li Y, Gu Q, Liu F, Shao B, An J (2016) Regulation of proliferation, angiogenesis and apoptosis in hepatocellular carcinoma by miR-26b-5p. Tumor Biol 37(8):10965–10979
Liu Y, Cai Q, Bao PP, Su Y, Cai H, Wu J, Ye F, Guo X, Zheng W, Zheng Y, Shu XO (2015) Tumor tissue microRNA expression in association with triple-negative breast cancer outcomes. Breast Cancer Res Treat 152(1):183–191
Stavraka C, Blagden S (2015) The la-related proteins, a family with connections to cancer. Biomolecules 5(4):2701–2722
Mura M, Hopkins TG, Michael T, Abd-Latip N, Weir J, Aboagye E, Mauri F, Jameson C, Sturge J, Gabra H, Bushell M, Willis AE, Curry E, Blagden SP (2014) LARP1 post-transcriptionally regulates mTOR and contributes to cancer progression. Oncogene 34:5025–5036
Hopkins TG, Mura M, Al-Ashtal HA, Lahr RM, Abd-Latip N, Sweeney K, Lu H, Weir J, El-Bahrawy M, Steel JH, Ghaem-Maghami S, Aboagye EO, Berman AJ, Blagden SP (2016) The RNA-binding protein LARP1 is a post-transcriptional regulator of survival and tumorigenesis in ovarian cancer. Nucleic Acids Res 44(3):1227–1246
Tcherkezian J, Cargnello M, Romeo Y, Huttlin EL, Lavoie G, Gygi SP, Roux PP (2014) Proteomic analysis of cap-dependent translation identifies LARP1 as a key regulator of 5′TOP mRNA translation. Genes Dev 28(4):357–371
Kato M, Goto Y, Matsushita R, Kurozumi A, Fukumoto I, Nishikawa R, Sakamoto S, Enokida H, Nakagawa M, Ichikawa T, Seki N (2015) MicroRNA-26a/b directly regulate la-related protein 1 and inhibit cancer cell invasion in prostate cancer. Int J Oncol 47(2):710–718
Riley KJ, Rabinowitz GS, Yario TA, Luna JM, Darnell RB, Steitz JA (2012) EBV and human microRNAs co-target oncogenic and apoptotic viral and human genes during latency. EMBO J 31:2207–2221
Yin S, Xu L, Bandyopadhyay S, Sethi S, Reddy KB (2011) Cisplatin and TRAIL enhance breast cancer stem cell death. Int J Oncol 39(4):891–898
Xu W-S, Dang Y-Y, Chen X-P, Lu J-J, Wang Y-T (2013) Furanodiene presents synergistic anti-proliferative activity with paclitaxel via altering cell cycle and integrin signaling in 95-D lung cancer cells. Phytother Res 28(2):296–299
Jin H, Park M, Kim S (2015) 3,3′-Diindolylmethane potentiates paclitaxel-induced antitumor effects on gastric cancer cells through the Akt/FOXM1 signaling cascade. Oncol Rep 33:2031–2036
van Jaarsveld MTM, van Kuijk PF, Boersma AW, Helleman J, van IJcken WF, Mathijssen RH, Berns EM, Pothof J, Verweij J, Wiemer EA (2015) miR-634 restores drug sensitivity in resistant ovarian cancer cells by targeting the Ras-MAPK pathway. Mol Cancer 14:196–208
Acknowledgements
This study was supported by The Scientific and Technological Research Council of Turkey (TÜBİTAK), project number: 213S012.
Author information
Authors and Affiliations
Contributions
HS: protocol/project development. HS, BV, NA, DD, and SA: laboratory studies. HS, MS, AS, and FB: data analysis and interpreting results. HS and SA: manuscript writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Samli, H., Samli, M., Vatansever, B. et al. Paclitaxel resistance and the role of miRNAs in prostate cancer cell lines. World J Urol 37, 1117–1126 (2019). https://doi.org/10.1007/s00345-018-2501-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2501-6