Abstract
Objectives
To develop a multi-institutional prediction model to estimate the local response to oesophageal squamous cell carcinoma (ESCC) treated with definitive radiotherapy based on radiomics and dosiomics features.
Methods
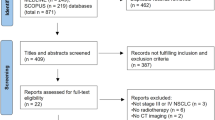
The local responses were categorised into two groups (incomplete and complete). An external validation model and a hybrid model that the patients from two institutions were mixed randomly were proposed. The ESCC patients at stages I–IV who underwent chemoradiotherapy from 2012 to 2017 and had follow-up duration of more than 5 years were included. The patients who received palliative or pre-operable radiotherapy and had no FDG PET images were excluded. The segmentations included the GTV, CTV, and PTV which are used in treatment planning. In addition, shrinkage, expansion, and shell regions were created. Radiomic and dosiomic features were extracted from CT, FDG PET images, and dose distribution. Machine learning-based prediction models were developed using decision tree, support vector machine, k-nearest neighbour (kNN) algorithm, and neural network (NN) classifiers.
Results
A total of 116 and 26 patients enrolled at Centre 1 and Centre 2, respectively. The external validation model exhibited the highest accuracy with 65.4% for CT-based radiomics, 77.9% for PET-based radiomics, and 72.1% for dosiomics based on the NN classifiers. The hybrid model exhibited the highest accuracy of 84.4% for CT-based radiomics based on the kNN classifier, 86.0% for PET-based radiomics, and 79.0% for dosiomics based on the NN classifiers.
Conclusion
The proposed hybrid model exhibited promising predictive performance for the local response to definitive radiotherapy in ESCC patients.
Clinical relevance statement
The prediction of the complete response for oesophageal cancer patients may contribute to improving overall survival. The hybrid model has the potential to improve prediction performance than the external validation model that was conventionally proposed.
Key Points
• Radiomics and dosiomics used to predict response in patients with oesophageal cancer receiving definitive radiotherapy.
• Hybrid model with neural network classifier of PET-based radiomics improved prediction accuracy by 8.1%.
• The hybrid model has the potential to improve prediction performance.





Similar content being viewed by others
Abbreviations
- AI:
-
Artificial intelligence
- CM:
-
Confusion matrix
- CRT:
-
Chemoradiotherapy
- CT:
-
Computed tomography
- CTV:
-
Clinical target volume
- DIR:
-
Deformable image registration
- ESCC:
-
Oesophageal squamous cell carcinoma
- FDG-PET:
-
Fluorodeoxyglucose-Positron Emission Tomography
- GTV:
-
Gross tumour volume
- KNN:
-
K-nearest neighbour
- LASSO:
-
Least absolute shrinkage and selection operator
- LN:
-
Lymph node
- NN:
-
Neural network
- pCR:
-
Pathological complete response
- PTV:
-
Planning target volume
- ROC:
-
Receiver operator characteristic
- ROI:
-
Regions of interest
- RT:
-
Radiotherapy
- SVM:
-
Support vector machine
- VIF:
-
Variance inflation factor
References
Zhang HZ, Jin GF, Shen HB (2012) Epidemiologic differences in esophageal cancer between Asian and Western populations. Chin J Cancer 31(6):281–286
van Hagen P, Hulshof MC, van Lanschot JJ et al (2012) Preoperative chemoradiotherapy for esophageal or junctional cancer. N Engl J Med 366:2074–2084
van Heijl M, van Lanschot JJ, Koppert LB et al (2008) Neoadjuvant chemoradiation followed by surgery versus surgery alone for patients with adenocarcinoma or squamous cell carcinoma of the esophagus (CROSS). BMC Surg 8:21
National Comprehensive Cancer Network (2017) NCCN Guidelines for Treatment of Cancer by Site: Esophageal Cancer [online]. Version 1.2017. http://www.nccn.org/professionals/default.aspx. Accessed 5 April 2022
Nomura M, Kato K, Ando N et al (2017) Comparison between neoadjuvant chemotherapy followed by surgery and definitive chemoradiotherapy for overall survival in patients with clinical stage II/III esophageal squamous cell carcinoma (JCOG 1406-A). Jpn J Clin Oncol 47(6):480–6
Yan MH, Hou XB, Cai BN et al (2020) Neoadjuvant chemoradiotherapy plus surgery in the treatment of potentially resectable thoracic esophageal squamous cell carcinoma. World J Clin Cases 8(24):6315–6321
Rizzo S, Botta F, Raimondi S et al (2018) Radiomics: the facts and the challenges of image analysis. Eur Radiol Exp 2(1):36
Parekh V, Jacobs MA (2016) Radiomics: a new application from established techniques. Expert Rev Precis Med Drug Dev 1(2):207–226
Xie CY, Pang CL, Chan B et al (2021) Machine Learning and Radiomics Applications in Esophageal Cancers Using Non-Invasive Imaging Methods-A Critical Review of Literature. Cancers (Basel) 13(10):2469. https://doi.org/10.3390/cancers13102469
Hu Y, Xie C, Yang H et al (2020) Assessment of Intratumoral and Peritumoral Computed Tomography Radiomics for Predicting Pathological Complete Response to Neoadjuvant Chemoradiation in Patients With Esophageal Squamous Cell Carcinoma. JAMA Netw Open 3:e2015927
Yang Z, He B, Zhuang X et al (2019) CT-based radiomic signatures for prediction of pathologic complete response in esophageal squamous cell carcinoma after neoadjuvant chemoradiotherapy. J Radiat Res 60:538–545
Tixier F, Le Rest CC, Hatt M et al (2011) Intratumor heterogeneity characterized by textural features on baseline 18F-FDG PET images predicts response to concomitant radiochemotherapy in esophageal cancer. J Nucl Med 52(3):369–378
Desbordes P, Ruan S, Modzelewski R et al (2017) Predictive value of initial FDG-PET features for treatment response and survival in esophageal cancer patients treated with chemo-radiation therapy using a random forest classifier. PLoS One 12:e0173208
Murakami Y, Kawahara D, Tani S et al (2021) Predicting the Local Response of Esophageal Squamous Cell Carcinoma to Neoadjuvant Chemoradiotherapy by Radiomics with a Machine Learning Method Using 18F-FDG PET Images. Diagnostics (Basel) 11(6):1049
Hou Z, Li S, Ren W et al (2018) Radiomic analysis in T2W and SPAIR T2W MRI: Predict treatment response to chemoradiotherapy in esophageal squamous cell carcinoma. J Thorac Dis 10:2256–2267
Cao Q, Li Y, Li Z et al (2020) Development and validation of a radiomics signature on differentially expressed features of 18F-FDG PET to predict treatment response of concurrent chemoradiotherapy in thoracic esophagus squamous cell carcinoma. Radiother Oncol 146:9–15
Fulmer JD, Roberts WC, von Gal ER et al (1979) Morphologic-physiologic correlates of the severity of fibrosis and degree of cellularity in idiopathic pulmonary fibrosis. J Clin Invest 63(4):665–676
van Griethuysen JJM, Fedorov A, Parmar C et al (2017) Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res 77:e104–e107
Tibshirani R (1997) The lasso method for variable selection in the Cox model. Stat Med 16:385–395
Zhang JX, Song W, Chen ZH et al (2013) Prognostic and predictive value of a microRNA signature in stage II colon cancer: a microRNA expression analysis. Lancet Oncol 14:1295–1306
Breiman L (2001) Random Forests. Machine Learn 45:5–32
Cortes C, Vapnik V (1995) Support-vector networks. Mach Learn 20:273–297
Zhang Z (2016) Introduction to machine learning: k-nearest neighbors. Ann Transl Med 4:218
Ho TK (1998) The random subspace method for constructing decision forests. IEEE Trans Pattern Anal Mach Intell 20:832–844
Hou Z, Ren W, Li S et al (2017) Radiomic analysis in contrast-enhanced CT: Predict treatment response to chemoradiotherapy in esophageal carcinoma. Oncotarget 8:104444–104454
van Rossum PS, Fried DV, Zhang L et al (2016) The Incremental Value of Subjective and Quantitative Assessment of 18F-FDG PET for the Prediction of Pathologic Complete Response to Preoperative Chemoradiotherapy in Esophageal Cancer. J Nucl Med 57(5):691–700
Tan S, Kligerman S, Chen W et al (2013) Spatial-temporal [18F] FDG-PET features for predicting pathologic response of esophageal cancer to neoadjuvant chemoradiation therapy. Int J Radiat Oncol Biol Phys 85(5):1375–1382
Tan S, Zhang H, Zhang Y et al (2013) Predicting pathologic tumor response to chemoradiotherapy with histogram distances characterizing longitudinal changes in 18F-FDG uptake patterns. Med Phys 40(10):101707
Hu Y, Xie C, Yang H et al (2021) Computed tomography-based deep-learning prediction of neoadjuvant chemoradiotherapy treatment response in esophageal squamous cell carcinoma. Radiother Oncol 154:6–13
Bleker J, Yakar D, van Noort B et al (2021) Single-center versus multi-center biparametric MRI radiomics approach for clinically significant peripheral zone prostate cancer. Insights Imaging 12(1):150
Pati S, Baid U, Edwards B et al (2022) Nat Commun 13(1):7346
Braman NM, Etesami M, Prasanna P et al (2017) Intratumoral and peritumoral radiomics for the pretreatment prediction of pathological complete response to neoadjuvant chemotherapy based on breast DCE-MRI. Breast Cancer Res 19(1):57
Sun C, Tian X, Liu Z et al (2019) Radiomic analysis for pretreatment prediction of response to neoadjuvant chemotherapy in locally advanced cervical cancer: A multicentre study. EBioMedicine 46:160–169
Beig N, Khorrami M, Alilou M et al (2019) Perinodular and intranodular radiomic features on lung CT images distinguish adenocarcinomas from granulomas. Radiology 290(3):783–792
Wu Q, Wang S, Chen X et al (2019) Radiomics analysis of magnetic resonance imaging improves diagnostic performance of lymph node metastasis in patients with cervical cancer. Radiother Oncol 138:141–148
Acknowledgements
We would like to thank Mathworks, Inc. for providing technical support.
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Yasushi Nagata.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Study subjects or cohorts overlap
None.
Methodology
• retrospective
• cross sectional study
• multicentre study
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kawahara, D., Murakami, Y., Awane, S. et al. Radiomics and dosiomics for predicting complete response to definitive chemoradiotherapy patients with oesophageal squamous cell cancer using the hybrid institution model. Eur Radiol 34, 1200–1209 (2024). https://doi.org/10.1007/s00330-023-10020-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-023-10020-8