Abstract
Objectives
Lung cancer is the most common cancer and the leading cause of cancer-related death worldwide. The optimal management of computed tomography (CT)-indeterminate pulmonary nodules is important. To optimize individualized follow-up strategies, we developed a radiomics nomogram for predicting 2-year growth in case of indeterminate small pulmonary nodules.
Methods
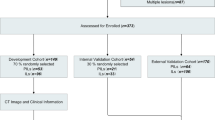
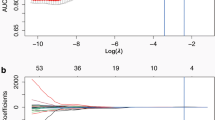
A total of 215 histopathology-confirmed small pulmonary nodules (21 benign and 194 malignant) in 205 patients with ultra-high-resolution CT (U-HRCT) were divided into growth and nongrowth nodules and were randomly allocated to the primary (n = 151) or validation (n = 64) group. The least absolute shrinkage and selection operator (LASSO) method was used for radiomics feature selection and radiomics signature determination. Multivariable logistic regression analysis was used to develop a radiomics nomogram that integrated the radiomics signature with significant clinical parameters (sex and nodule type). The area under the curve (AUC) was applied to assess the predictive performance of the radiomics nomogram. The net benefit of the radiomics nomogram was assessed using a clinical decision curve.
Results
The radiomics signature and nomogram yielded AUCs of 0.892 (95% confidence interval [CI]: 0.843–0.940) and 0.911 (95% CI: 0.867–0.955), respectively, in the primary group and 0.826 (95% CI: 0.727–0.926) and 0.843 (95% CI: 0.749–0.937), respectively, in the validation group. The clinical usefulness of the nomogram was demonstrated by decision curve analysis.
Conclusions
A radiomics nomogram was developed by integrating the radiomics signature with clinical parameters and was easily used for the individualized prediction of two-year growth in case of CT-indeterminate small pulmonary nodules.
Key Points
• A radiomics nomogram was developed for predicting the two-year growth of CT-indeterminate small pulmonary nodules.
• The nomogram integrated a CT-based radiomics signature with clinical parameters and was valuable in developing an individualized follow-up strategy for patients with indeterminate small pulmonary nodules.






Similar content being viewed by others
Abbreviations
- AIC:
-
Akaike information criterion
- CDC:
-
Clinical decision curve
- CI:
-
Confidence interval
- FOV:
-
Field of view
- GGN:
-
Ground-glass nodule
- LASSO:
-
Least absolute shrinkage and selection operator
- U-HRCT:
-
Ultra-high-resolution computed tomography
- VDT:
-
Volume-doubling time
- VOI:
-
Volume of interest
References
She J, Yang P, Hong QY, Bai CX (2013) Lung cancer in China challenges and interventions. Chest 143:1117–1126
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer 68:394–424
Gould MK, Tang T, Liu ILA et al (2015) Recent trends in the identification of incidental pulmonary nodules. Am J Respir Crit Care Med 192:1208–1214
Siegel RL, Miller KD, Jemal A (2020) Cancer statistics, 2020. CA Cancer J Clin 70:7–30
Gopal M, Abdullah SE, Grady JJ, Goodwin JS (2010) Screening for lung cancer with low-dose computed tomography: a systematic review and meta-analysis of the baseline findings of randomized controlled trials. J Thorac Oncol 5:1233–1239
Hawkins S, Wang H, Liu Y et al (2016) Predicting malignant nodules from screening CT scans. J Thorac Oncol 11:2120–2128
Shin KE, Lee KS, Yi CA, Chung MJ, Shin MH, Choi YH (2014) Subcentimeter lung nodules stable for 2 years at LDCT: Long-term follow-up using volumetry. Respirology 19:921–928
Kobayashi Y, Fukui T, Ito S et al (2013) How long should small lung lesions of ground-glass opacity be followed? J Thorac Oncol 8:309–314
Henschke CI, Yankelevitz DF, Mirtcheva R et al (2002) CT screening for lung cancer: frequency and significance of part-solid and nonsolid nodules. AJR Am J Roentgenol 178:1053–1057
Song YS, Park CM, Park SJ, Lee SM, Jeon YK, Goo JM (2014) Volume and mass doubling times of persistent pulmonary subsolid nodules detected in patients without known malignancy. Radiology 273:276–284
Fang WT, Xiang YW, Zhong CX, Chen QH (2014) The IASLC/ATS/ERS classification of lung adenocarcinoma-a surgical point of view. J Thorac Dis 6:S552–S560
Mets OM, de Jong PA, Chung K, Lammers JJ, van Ginneken B, Schaefer-Prokop CM (2016) Fleischner recommendations for the management of subsolid pulmonary nodules: high awareness but limited conformance - a survey study. Eur Radiol 26:3840–3849
Miyata T, Yanagawa M, Hata A et al (2020) Influence of field of view size on image quality: ultra-high-resolution CT vs. conventional high-resolution CT. Eur Radiol 30:3324–3333
Zhu HY, Zhang L, Wang YL et al (2017) Improved image quality and diagnostic potential using ultra-high-resolution computed tomography of the lung with small scan FOV: a prospective study. PLoS One 12:e0172688
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: Images are more than pictures, they are data. Radiology 278:563–577
Lambin P, Rios-Velazquez E, Leijenaar R et al (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441–446
Liu AL, Wang ZH, Yang YC et al (2020) Preoperative diagnosis of malignant pulmonary nodules in lung cancer screening with a radiomics nomogram. Cancer Commun (Lond) 40:16–24
Xue X, Yang Y, Huang Q et al (2018) Use of a radiomics model to predict tumor invasiveness of pulmonary adenocarcinomas appearing as pulmonary ground-glass nodules. Biomed Res Int 2018:6803971
Ferreira-Junior JR, Koenigkam-Santos M, MagalhãesTenório AP et al (2020) CT-based radiomics for prediction of histologic subtype and metastatic disease in primary malignant lung neoplasms. Int J Comput Assist Radiol Surg 15:163–172
Digumarthy SR, Padole AM, Rastogi S et al (2019) Predicting malignant potential of subsolid nodules: can radiomics preempt longitudinal follow up CT? Cancer Imaging 19:36
Sun Q, Huang Y, Wang J et al (2019) Applying CT texture analysis to determine the prognostic value of subsolid nodules detected during low-dose CT screening. Clin Radiol 74:59–66
Nair A, Baldwin DR, Field JK, Hansell DM, Devaraj A (2012) Measurement methods and algorithms for the management of solid nodules. J Thorac Imaging 27:230–239
Scholten ET, de Jong PA, de Hoop B et al (2015) Towards a close computed tomography monitoring approach for screen detected subsolid pulmonary nodules? Eur Resp J 45:765–773
Revel MP, Mannes I, Benzakoun J et al (2018) Subsolid lung nodule classification: a CT criterion for improving interobserver agreement. Radiology 286:316–325
Lindell RM, Hartman TE, Swensen SJ, Jet JR, Midthun DE, Mandrekar JN (2009) 5-Year lung cancer screening experience growth curves of 18 lung cancers compared to histologic type, CT attenuation, stage, survival, and size. Chest 136:1586–1595
Mets OM, Chung K, Zanen P et al (2018) In vivo growth of 60 non-screening detected lung cancers: a computed tomography study. Eur Resp J 51:1702183
de Margerie-Mellon C, Ngo LH, Gill RR et al (2020) The growth rate of subsolid lung adenocarcinoma nodules at chest CT. Radiology 297:189–198
Lee SW, Leem CS, Kim TJ et al (2013) The long-term course of ground-glass opacities detected on thin-section computed tomography. Respir Med 107:904–910
Hiramatsu M, Inagaki T, Inagaki T et al (2008) Pulmonary ground-glass opacity (GGO) lesions-large size and a history of lung cancer are risk factors for growth. J Thorac Oncol 3:1245–1250
Qiu TC, Ru XS, Yin K, Yu J, Song Y, Wu JL (2020) Two nomograms based on CT features to predict tumor invasiveness of pulmonary adenocarcinoma and growth in pure GGN: a retrospective analysis. Jpn J Radiol 38:761–770
Kakinuma R, Moriyama N, Muramatsu Y et al (2015) Ultra-high-resolution computed tomography of the lung: image quality of a prototype scanner. PLoS One 10:e0137165
Sheshadri A, Rodriguez A, Chen R et al (2015) Effect of reducing field of view on multidetector quantitative computed tomography parameters of airway wall thickness in asthma. J Comput Assist Tomogr 39:584–590
Funding
This work has received funding from Shanghai Chest Hospital (No. 2019YNJCQ02), Shanghai Municipal Health Commission (No. ZK2019B01), National Natural Science Foundation of China (No. 82071873, 81871353), and Science and Technology Commission of Shanghai Xuhui District Municipality (No. 2020–010).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Jinwei Qiang.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
Ying Li has significant statistical expertise.
Informed consent
Written informed consent was obtained from all patients in this study.
Ethical approval
Institutional Review Board approval was obtained by Shanghai Chest Hospital.
Study subjects or cohorts overlap
No study subjects or cohorts overlap.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xue, L.M., Li, Y., Zhang, Y. et al. A predictive nomogram for two-year growth of CT-indeterminate small pulmonary nodules. Eur Radiol 32, 2672–2682 (2022). https://doi.org/10.1007/s00330-021-08343-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-021-08343-5