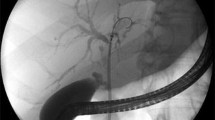
Abstract
Objectives
Benign strictures of the bile duct may be difficult to treat endoscopically due to altered bowel anatomy. Furthermore, recurrence of stenosis and symptoms remains high. The aim of the Spanish Prospective Registry BiELLA study was to investigate the safety and efficacy of absorbable stents in the treatment of benign biliary strictures and their outcomes on the medium and long-term follow-up.
Methods
A prospective, multicenter, observational, non-randomized study (the BiELLA study) was conducted from January 2014 to September 2018. One hundred fifty-nine patients with benign biliary strictures, mostly postsurgical, were enrolled for implantation of absorbable biliary stents in the 11 participating Spanish tertiary hospitals. The average patient follow-up was 45.4 ± 15.9 months (range, 12–60 months). The follow-up data included symptoms, biochemical parameters, and ultrasound images at 1, 6, and 12 months and then yearly for up to 60 months.
Results
The immediate technical and clinical success rates were 100%. In all patients, stent placement resulted in improvement of clinical symptoms and biochemical parameters. The primary mean patency for stent was 86.7, 79.6, and 78.9% at 12, 36, and 60 months, respectively (95% CI). Biliary restenosis and occlusion occurred in 40 (26.6%) patients. Of the 40 patients, 18 (12%) patients were treated with a second stent and 22 (14.6%) patients had operative repair of the recurrent strictures. There were no major complications associated with stent implantation.
Conclusions
Implantation of an absorbable polydioxanone biliary stent is safe and effective for treatment of benign biliary strictures refractory to balloon dilatation or other biliary intervention.
Key Points
• Percutaneous implantation of biodegradable prostheses for the treatment of benign postsurgical biliary strictures is a safe and effective procedure.
• More than 75% of the patients presented patency of the stented biliary tree at 5 years follow-up.
• Absorbable stents improved clinical symptoms and signs (jaundice, itching, fever), and laboratory parameters in a few days after stent placement.






Similar content being viewed by others
Abbreviations
- ALP:
-
Alkaline phosphatase
- ASA:
-
American Society of Anesthesiologist
- BBDS:
-
Benign bile duct stenosis
- BBS:
-
Biodegradable biliary stents
- BiELLA:
-
Name of the Spanish prospective registry of biodegradable biliary stents
- CEICA:
-
Ethical Committee of Investigation of Aragon Community
- CI:
-
Confidence interval
- ECOG:
-
Eastern Cooperative Oncology Group
- ERCP:
-
Endoscopic retrograde cholangiopancreatography
- Fr:
-
French
- GGT:
-
Gamma-glutamyl transferase
- mGy:
-
Milligray
- PPDX:
-
Polydioxanone
- PTA:
-
Percutaneous transluminal angioplasty
- PTHC:
-
Percutaneous transhepatic cholangiogram
- SERVEI:
-
Spanish Society of Vascular and Interventional Radiology
References
Dimou FM, Adhikari D, Mehta HB, Olino K, Riall TS, Brown KM (2016) Incidence of hepaticojejunostomy stricture following hepaticojejunostomy. Surgery 160:691–698
Ahrendt SA, Pitt HA (2001) Surgical therapy of iatrogenic lesions of biliary tract. World J Surg 25:1360–1365
Strasberg SM, Picus DD, Drebin JA (2001) Results of a new strategy for reconstruction of biliary injuries having an isolated right-sided component. J Gastrointest Surg 5:266–274
Lillemoe KD, Petrofski JA, Choti MA, Venbrux AC, Cameron JL (2000) Isolated right segmental hepatic injury: a diagnostic and therapeutic challenge. J Gastrointest Surg 2:168–177
Mercado MA, Chan C, Orozco H, Tielve M, Hinojosa CA (2003) Acute bile duct injury. Surg Endosc 17:1351–1355
Mercado MA, Orozco H, De la Garza L, López-Martínez LM, Contreras A , Guillén-Navarro E (1999) Biliary duct injury: partial segment IV resection for intrahepatic reconstruction of biliary lesions. Arch Surg 134:1008–1010
Lillemoe KD, Pitt HA, Cameron JL (1992) Current management of benign bile duct strictures. Adv Surg 25:119–174
Lillemoe KD (1997) Benign postoperative bile duct strictures. Baillieres Clin Gastroenterol 11:749–779
Lillemoe KD (2003) To err is human, but should we expect more from a surgeon? Ann Surg 237:470–471
Kapoor BS, Mauri G, Lorenz JM (2018) Management of biliary strictures: state-of-the-art review. Radiology 289:590–603
Baron TH, Davee T (2012) Endoscopic management of benign bile duct strictures. Gastrointest Endosc Clin N Am 23:295–311
Kim JH, Gwon D II, Ko G-Y et al (2011) Temporary placement of retrievable fully covered metallic stents versus percutaneous balloon dilation in the treatment of benign biliary strictures. J Vasc Interv Radiol 22:893–899
Irani S, Baron TH, Akbar A et al (2014) Endoscopic treatment of benign biliary strictures using covered self-expandable metal stents (CSEMS). Dig Dis Sci 59:152–160
Ludwig JM, Webber GR, Knechtle SJ, Spivey JR, Xing M, Kim HS (2016) Percutaneous management of benign biliary strictures with large-bore catheters: comparison between patients with and without orthotopic liver transplantation. J Vasc Interv Radiol 27:219–225
Gwon D II, Ko G-Y, Ko HK, Yoon H-K, Sung K-B (2013) Percutaneous transhepatic treatment using retrievable covered stents in patients with benign biliary strictures: mid-term outcomes in 68 patients. Dig Dis Sci 58:3270–3279
Moraca RJ, Lee F, Ryan JA Jr, Traverso LW (2002) Long term biliary function after reconstruction of major bile duct injuries with hepaticoduodenostomy or hepaticojejunostomy. Arch Surg 137:889–893
Melton GB, Lillemoe KD (2002) The current management of postoperative bile duct strictures. Adv Surg 36:193–221
Kalinowski M, Alfke H, Kleb B, Dürfeld F, Joachim Wagner H (2002) Paclitaxel inhibits proliferation of cell lines responsible for metal stent obstruction: possible topical application in malignant bile duct obstructions. Invest Radiol 37:399–404
Mauri G, Michelozzi C, Melchiorre F et al (2013) Biodegradable biliary stent implantation in the treatment of benign bilioplastic-refractory biliary strictures: preliminary experience. Eur Radiol 23:3304–3310
Mauri G, Michelozzi C, Melchiorre F et al (2016) Benign biliary strictures refractory to standard bilioplasty treated using polydoxanone biodegradable biliary stents: retrospective multicentric data analysis on 107 patients. Eur Radiol 26:4057–4063 13
Petrtýl J, Brůha R, Horák L, Zádorová Z, Dosedel J, Laasch HU (2010) Management of benign intrahepatic bile duct strictures: initial experience with polydioxanone biodegradable stents. Endoscopy 42:E89–E90
Dopazo C, Diez I, Quintero J et al (2018) Role of biodegradable stents as part of treatment of biliary strictures after pediatric and adult liver transplantation: an observational single-center study. J Vasc Interv Radiol 29:899–904
Vondrys D, Elliott MJ, McLaren CA, Noctor C, Roebuck DJ (2011) First experience with biodegradable airway stents in children. Ann Thorac Surg 92:1870–1874 24
Janík V, Horák L, Hnaníček J, Málek J, Laasch HU (2011) Biodegradable polydioxanone stents: a new option for therapy resistant anastomotic strictures of the colon. Eur Radiol 21:1956–1961
Stivaros SM, Williams LR, Senger C, Wilbraham L, Laasch HU (2010) Woven polydioxanone biodegradable stents: a new treatment option for benign and malignant oesophageal strictures. Eur Radiol 20:1069–1072
Shi J, Lv Y, Yu L et al (2013) Interest of a new biodegradable stent coated with paclitaxel on anastomotic wound healing after biliary reconstruction. Eur J Gastroenterol Hepatol 25:1415–1423
Yamamoto K, Yoshioka T, Furuichi K et al (2011) Experimental study of poly- L-lactic acid biodegradable stents in normal canine bile ducts. Cardiovasc Intervent Radiol 34:601–608
Mauri G, Criado E (2016) Percutaneous management of benign biliary strictures: is it time to focus on reducing procedure invasiveness? J Vasc Interv Radiol 27:934–936
von Elm E, Altman DG, Egger M et al (2008) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 61:344–349
Khan M, Baron T, Kamal F et al (2017) Efficacy of self-expandable metal stents in management of benign biliary strictures and comparison with multiple plastic stents: a meta-analysis. Endoscopy 49:682–694
Shimada H, Endo I, Shimada K, Matsuyama R, Kobayashi N, Kubota K (2012) The current diagnosis and treatment of benign biliary stricture. Surg Today 42:1143–1153
Judah JR, Draganov PV (2007) Endoscopic therapy of benign biliary strictures. World J Gastroenterol 13:3531–3539
Zepeda-Gómez S, Baron TH (2011) Benign biliary strictures: current endoscopic management. Nat Rev Gastroenterol Hepatol 8:573–581
Garcia-Cano J (2012) Use of fully covered self-expanding metal stents in benign biliary diseases. World J Gastrointest Endosc 4:142–147
van Boeckel PGA, Vleggaar FP, Siersema PD (2009) Plastic or metal stents for benign extrahepatic biliary strictures: a systematic review. BMC Gastroenterol 9:96
Cozzaglio L, Cimino M, Mauri G et al (2011) Percutaneous transhepatic biliary drainage and occlusion balloon in the management of duodenal stump fistula. J Gastrointest Surg 15:1977–1981
Pedicini V, Poretti D, Mauri G et al (2010) Management of post-surgical biliary leakage with percutaneous transhepatic biliary drainage (PTBD) and occlusion balloon (OB) in patients without dilatation of the biliary tree: preliminary results. Eur Radiol 20:1061–1068
Yano T, Yoda Y, Nomura S et al (2017) Prospective trial of biodegradable stents for refractory benign esophageal strictures after curative treatment of esophageal cancer. Gastrointest Endosc 86:492–499
Sigounas DE, Siddhi S, Plevris JN (2016) Biodegradable esophageal stents in benign and malignant strictures - a single center experience. Endosc Int Open 4:E618–E623
Köcher M, Cerná M, Havlík R, Král V, Gryga A, Duda M (2007) Percutaneous treatment of benign bile duct strictures. Eur J Radiol 62:170–174
Atar E, Bachar GN, Bartal G et al (2005) Use of peripheral cutting balloon in the management of resistant benign ureteral and biliary strictures. J Vasc Interv Radiol 16:241–245
Dhondt E, Vanlangenhove P, Van Vlierberghe H et al (2019) Benign anastomotic biliary strictures untreatable by ERCP: a novel percutaneous balloon dilatation technique avoiding indwelling catheters. Eur Radiol 29:636–644
Grolich T, Crha M, Novotný L et al (2015) Self-expandable biodegradable biliary stents in porcine model. J Surg Res 193:606–612
ASGE Technology Committee, Tokar JL, Banerjee S et al (2011) Drugeluting/ biodegradable stents. Gastrointest Endosc 74:954–958
Siiki A, Rinta-Kiikka I, Sand J, Laukkarinen J (2018) A pilot study of endoscopically inserted biodegradable biliary stents in the treatment of benign biliary strictures and cystic duct leaks. Gastrointest Endosc 87:1132–1137
Siiki A, Sand J, Laukkarinen J (2018) A systematic review of biodegradable biliary stents: promising biocompatibility without stent removal. Eur J Gastroenterol Hepatol 30:813–818
Lorenzo-Zúñiga V, Moreno-de-Vega V, Marín I, Boix J (2014) Biodegradable stents in gastrointestinal endoscopy. World J Gastroenterol 20:2212–2217
Funding
The authors state that this work has not received any funding.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Miguel Ángel de Gregorio Ariza.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Study subjects or cohorts overlap
Some study subjects or cohorts have been previously reported in Treatment of benign lesions of the bile duct via Biodegradable stents. Long-term follow-up. Prospective Multicentric Spanish BIELLA Registry. Poster CIRSE 2019. By E. Criado Paredes.
Methodology
• Prospective
• Observational
• Multicenter study
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
De Gregorio, M.A., Criado, E., Guirola, J.A. et al. Absorbable stents for treatment of benign biliary strictures: long-term follow-up in the prospective Spanish registry. Eur Radiol 30, 4486–4495 (2020). https://doi.org/10.1007/s00330-020-06797-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-06797-7