Abstract
Purpose
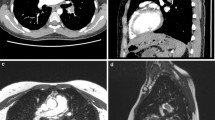
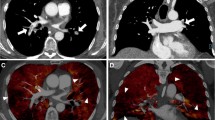
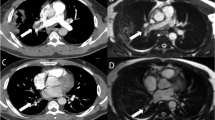
Truncation artefact (Gibbs ringing) causes central signal drop within vessels in pulmonary magnetic resonance angiography (MRA) that can be mistaken for emboli, reducing diagnostic accuracy for pulmonary embolism (PE). We propose a quantitative approach to differentiate truncation artefact from PE.
Methods
Twenty-eight patients who underwent pulmonary computed tomography angiography (CTA) for suspected PE were recruited for pulmonary MRA. Signal intensity drops within pulmonary arteries that persisted on both arterial-phase and delayed-phase MRA were identified. The percent signal loss between the vessel lumen and central drop was measured. CTA served as the reference standard for presence of pulmonary emboli.
Results
A total of 65 signal intensity drops were identified on MRA. Of these, 48 (74 %) were artefacts and 17 (26 %) were PE, as confirmed by CTA. Truncation artefacts had a significantly lower median signal drop than PE on both arterial-phase (26 % [range 12–58 %] vs. 85 % [range 53–91 %]) and delayed-phase MRA (26 % [range 11–55 %] vs. 77 % [range 47–89 %]), p < 0.0001 for both. Receiver operating characteristic (ROC) analyses revealed a threshold value of 51 % (arterial phase) and 47 % signal drop (delayed phase) to differentiate between truncation artefact and PE with 100 % sensitivity and greater than 90 % specificity.
Conclusion
Quantitative signal drop is an objective tool to help differentiate truncation artefact and pulmonary embolism in pulmonary MRA.
Key points
• Inexperienced readers may mistake truncation artefacts for emboli on pulmonary MRA
• Pulmonary emboli have non-uniform signal drop
• 51 % (arterial phase) and 47 % (delayed phase) cut-off differentiates truncation artefact from PE
• Quantitative signal drop measurement enables more accurate pulmonary embolism diagnosis with MRA






Similar content being viewed by others
References
Schiebler ML, Nagle SK, François CJ et al (2013) Effectiveness of MR angiography for the primary diagnosis of acute pulmonary embolism: clinical outcomes at 3 months and 1 year. J Magn Reson Imaging 38:914–925
François CJ, Hartung MP, Reeder SB et al (2013) MRI for acute chest pain: current state of the Art. J Magn Reson Imaging 37:1290–1300
Kalb B, Sharma P, Tigges S et al (2012) MR imaging of pulmonary embolism: diagnostic accuracy of contrast-enhanced 3D MR pulmonary angiography, contrast-enhanced low-flip angle 3D GRE, and nonenhanced free-induction FISP sequences. Radiology 263:271–278
Lum DP, Busse RF, François CJ et al (2009) Increased volume of coverage for abdominal contrast-enhanced MR angiography with two-dimensional autocalibrating parallel imaging: initial experience at 3.0 Tesla. J Magn Reson Imaging 30:1093–1100
Londy FJ, Lowe S, Stein PD et al (2012) Comparison of 1.5 and 3.0 T for contrast-enhanced pulmonary magnetic resonance angiography. Clin Appl Thromb Hemost 18:134–139
Meaney JF, Weg JG, Chenevert TL et al (1997) Diagnosis of pulmonary embolism with magnetic resonance angiography. N Engl J Med 336:1422–1427
Stein PD, Fowler SE, Goodman LR et al (2006) Multidetector computed tomography for acute pulmonary embolism. N Engl J Med 354:2317–2327
Morelli JN, Runge VM, Ai F et al (2011) An image-based approach to understanding the physics of MR artifacts. Radiographics 31:849–866
Stein PD (2010) Gadolinium-enhanced magnetic resonance angiography for pulmonary embolism: a multicenter prospective study (PIOPED III). Ann Intern Med 152:434–443
Revel MP, Sanchez O, Couchon S et al (2012) Diagnostic accuracy of magnetic resonance imaging for an acute pulmonary embolism: results of the “IRM-EP” study. J Thromb Haemost 10:743–750
Nagle SK, Schiebler ML, François CJ et al (2010) Common artifacts of pulmonary artery MRA and potential solutions. Proc Intl Soc Mag Reson Med 18:1378
Kramer H, Nagle SK, François CJ et al (2013) Efficacy of 3D whole-lung single breath-hold contrast enhanced pulmonary MRA for detection of pulmonary embolism: comparison to CTA. Proc Intl Soc Mag Reson Med 21:1501
Harris FJ (1978) On the use of windows for harmonic analysis with the discrete Fourier transform. Proc IEEE 66:51–83
Gottlieb D, Shu C-W (1997) On the Gibbs phenomenon and its resolution. SIAM Rev 39:644–668
Di Bella EVR, Parker DL, Sinusas AJ (2005) On the dark rim artifact in dynamic contrast-enhanced MRI myocardial perfusion studies. Magn Reson Med 54:1295–1299
Sharif B, Dharmakumar R, LaBounty T et al (2013) Towards elimination of the dark-rim artifact in first-pass myocardial perfusion MRI: removing Gibbs ringing effects using optimized radial imaging. Magn Reson Med. doi:10.1002/mrm.24913
Rakow-Penner R, Gold G, Daniel B et al (2008) Reduction of truncation artifacts in rapid 3D articular cartilage imaging. J Magn Reson Imaging 27:860–865
Erickson SJ, Waldschmidt JG, Czervionke LF, Prost RW (1996) Hyaline cartilage: truncation artifact as a cause of trilaminar appearance with fat-suppressed three-dimensional spoiled gradient-recalled sequences. Radiology 201:260–264
Brau ACS, Beatty PJ, Skare S, Bammer R (2008) Comparison of reconstruction accuracy and efficiency among autocalibrating data-driven parallel imaging methods. Magn Reson Med 59:382–395
Maki JH, Prince MR, Londy FJ, Chenevert TL (1996) The effects of time varying intravascular signal intensity and k-space acquisition order on three-dimensional MR angiography image quality. J Magn Reson Imaging 6:642–651
Wittram C, Yoo AJ (2007) Transient interruption of contrast on CT pulmonary angiography: proof of mechanism. J Thorac Imaging 22:125–129
Fink C, Ley S, Risse F et al (2005) Effect of inspiratory and expiratory breathhold on pulmonary perfusion: assessment by pulmonary perfusion magnetic resonance imaging. Investig Radiol 40:72–79
Acknowledgments
The authors wish to thank Alejandro Munoz del Rio, PhD for his statistical advice at all stages of this project. The authors also wish to thank Bracco Diagnostic for their support through an unrestricted research grant. The scientific guarantor of this publication is Scott K Nagle. The authors of this manuscript declare relationships with the following companies: Bracco and GE. This study has received funding by the Department of Radiology Research and Development Committee. Institutional review board approval was obtained. Written informed consent was obtained from all subjects (patients) in this study. Methodology: prospective, cross-sectional study, performed at one institution.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bannas, P., Schiebler, M.L., Motosugi, U. et al. Pulmonary MRA: Differentiation of pulmonary embolism from truncation artefact. Eur Radiol 24, 1942–1949 (2014). https://doi.org/10.1007/s00330-014-3219-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-014-3219-5