Abstract
Key message
A novel rice constitutive promoter (P OsCon1 ) was isolated. The molecular mechanism of the promoter activity was investigated. P OsCon1 could be used as an alternative constitutive promoter for crop transgenic engineering.
Abstract
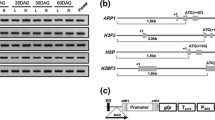
Monocot constitutive promoter is an important resource for crop transgenic engineering. In this report, we isolated a novel promoter, Oscon1 promoter (P OsCon1 ), from the 5′ upstream region of a constitutively expressed rice gene OsDHAR1. In P OsCon1 ::GUS transgenic rice, we showed that P OsCon1 had a broad expression spectrum in all tested tissues. The expression of the promoter was further analyzed in comparison with the previously characterized strong constitutive promoters. P OsCon1 exhibited comparable activity to OsCc1, OsAct1 or ZmUbi promoters in most tissues, and more active than 35S promoter in roots, seeds, and calli. Further quantitative assays indicated that P OsCon1 activity was not affected by developmental stages or by environmental factors. Further, 5′-deletions analysis indicated that the distinct regions might contribute to the strong expression of P OsCon1 in different tissues. Overall, our results suggest that P OsCon1 is a novel constitutive promoter, which could potentially use in transgenic crop development.







Similar content being viewed by others
Abbreviations
- P OsCon1 :
-
Rice constitutive promoter 1
- P OsAct1 :
-
Rice Actin1 promoter
- P 35S :
-
Cauliflower mosaic virus 35S promoter
- P ZmUbi :
-
Maize ubiquitin promoter
- P OsCc1 :
-
Rice OsCc1 promoter
- DHAR:
-
Dehydroascorbate reductase
- GUS:
-
β-Glucuronidase
- qRT-PCR:
-
Quantitative reverse transcription polymerase chain reaction
- MS:
-
Murashige and Skoog
- X-Gluc:
-
5-Bromo-4-chloro-3-indolyl-beta-glucuronide
- 4-MU:
-
4-Methylumbelliferone
- DAG:
-
Days after germination
- ASF1:
-
Activating sequence factor 1
- ASF2:
-
Activating sequence factor 2
- EEC:
-
Enhancer element consensus
- CGF1:
-
Chlorophyll a/b-binding protein gene GATA-box factor 1
- DHA:
-
Dehydroascorbate
- cytDHAR:
-
Cytosolic DHAR
- AsA:
-
Ascorbate acid
- ROS:
-
Reactive oxygen species
- SD:
-
Standard deviation
- GAP:
-
Glyceraldydyde-3-phosphate dehydrogenase
- CAB:
-
Chlorophyll A/B binding protein
- RBCS:
-
Ribulose bisphosphate carboxylase
References
Argüello-Astorga G, Herrera-Estrella L (1998) Evolution of light-regulated plant promoters. Annu Rev Plant Biol 49(1):525–555
Benfey PN, Ren L, Chua N-H (1990) Tissue-specific expression from CaMV 35S enhancer subdomains in early stages of plant development. EMBO J 9(6):1677
Bhattacharyya J, Chowdhury AH, Ray S, Jha JK, Das S, Gayen S, Sen SK (2012) Native polyubiquitin promoter of rice provides increased constitutive expression in stable transgenic rice plants. Plant Cell Rep 31(2):271–279. doi:10.1007/s00299-011-1161-4
Casal JJ, Yanovsky MJ (2005) Regulation of gene expression by light. Int J Dev Biol 49(5/6):501
Chen Q-J, Xie M, Ma X-X, Dong L, Chen J, Wang X-C (2010) MISSA is a highly efficient in vivo DNA assembly method for plant multiple-gene transformation. Plant Physiol 153(1):41–51. doi:10.1104/pp.109.152249
Christensen AH, Sharrock RA, Quail PH (1992) Maize polyubiquitin genes: structure, thermal perturbation of expression and transcript splicing, and promoter activity following transfer to protoplasts by electroporation. Plant Mol Biol 18(4):675–689. doi:10.1007/BF00020010
Cornejo M-J, Luth D, Blankenship KM, Anderson OD, Blechl AE (1993) Activity of a maize ubiquitin promoter in transgenic rice. Plant Mol Biol 23(3):567–581
Duan Y, Zhai C, Li H, Li J, Mei W, Gui H, Ni D, Song F, Li L, Zhang W, Yang J (2012) An efficient and high-throughput protocol for Agrobacterium-mediated transformation based on phosphomannose isomerase positive selection in Japonica rice (Oryza sativa L.). Plant Cell Reports 31(9):1611–1624. doi:10.1007/s00299-012-1275-3
Gallie DR (2013a) L-Ascorbic acid: a multifunctional molecule supporting plant growth and development. Scientifica 2013:24. doi:10.1155/2013/795964
Gallie DR (2013b) The role of L-ascorbic acid recycling in responding to environmental stress and in promoting plant growth. J Exp Bot 64(2):433–443. doi:10.1093/jxb/ers330
Holme IB, Wendt T, Holm PB (2013) Intragenesis and cisgenesis as alternatives to transgenic crop development. Plant Biotechnol J 11(4):395–407. doi:10.1111/pbi.12055
Jang IC, Choi WB, Lee KH, Song SI, Nahm BH, Kim JK (2002) High-level and ubiquitous expression of the rice cytochrome c gene OsCc1 and its promoter activity in transgenic plants provides a useful promoter for transgenesis of monocots. Plant Physiol 129(4):1473–1481. doi:10.1104/pp.002261
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6(13):3901
Jeong M-J, Shih M-C (2003) Interaction of a GATA factor with cis-acting elements involved in light regulation of nuclear genes encoding chloroplast glyceraldehyde-3-phosphate dehydrogenase in Arabidopsis. Biochem Biophys Res Commun 300(2):555–562
Karthikeyan A, Shilpha J, Karutha PS, Ramesh M (2012) Agrobacterium-mediated transformation of indica rice cv. ADT 43. Plant Cell Tissue Organ Culture (PCTOC) 109(1):153–165. doi:10.1007/s11240-011-0083-8
Kato Y, Urano J, Maki Y, Ushimaru T (1997) Purification and characterization of dehydroascorbate reductase from rice. Plant Cell Physiol 38(2):173–178
Kehoe DM, Degenhardt J, Winicov I, Tobin EM (1994) Two 10-bp regions are critical for phytochrome regulation of a Lemna gibba Lhcb gene promoter. Plant Cell Online 6(8):1123–1134
Kucho K, Yoshioka S, Taniguchi F, Ohyama K, Fukuzawa H (2003) Cis-acting elements and DNA-binding proteins involved in CO2-responsive transcriptional activation of Cah1 encoding a periplasmic carbonic anhydrase in Chlamydomonas reinhardtii. Plant Physiol 133(2):783–793
Kumar D, Patro S, Ghosh J, Das A, Maiti IB, Dey N (2012) Development of a salicylic acid inducible minimal sub-genomic transcript promoter from Figwort mosaic virus with enhanced root-and leaf-activity using TGACG motif rearrangement. Gene 503(1):36–47
Lam E, Benfey PN, Gilmartin PM, Fang R-X, Chua N-H (1989) Site-specific mutations alter in vitro factor binding and change promoter expression pattern in transgenic plants. Proc Natl Acad Sci 86(20):7890–7894
Li G, Peng X, Wei L, Kang G (2013) Salicylic acid increases the contents of glutathione and ascorbate and temporally regulates the related gene expression in salt-stressed wheat seedlings. Gene 529(2):321–325. doi:10.1016/j.gene.2013.07.093
Liu YJ, Yuan Y, Liu YY, Liu Y, Fu JJ, Zheng J, Wang GY (2012) Gene families of maize glutathione–ascorbate redox cycle respond differently to abiotic stresses. J Plant Physiol 169(2):183–192. doi:10.1016/j.jplph.2011.08.018
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25(4):402–408. doi:10.1006/meth.2001.1262
Lu J, Sivamani E, Li X, Qu R (2008) Activity of the 5′ regulatory regions of the rice polyubiquitin rubi3 gene in transgenic rice plants as analyzed by both GUS and GFP reporter genes. Plant Cell Rep 27(10):1587–1600. doi:10.1007/s00299-008-0577-y
Luo Q, Li Y, Gu H, Zhao L, Gu X, Li W (2013) The promoter of soybean photoreceptor GmPLP1 gene enhances gene expression under plant growth regulator and light stresses. Plant Cell Tissue Organ Culture (PCTOC) 114(1):109–119
Ma Q, Dai X, Xu Y, Guo J, Liu Y, Chen N, Chong K (2009) Enhanced tolerance to chilling stress in OsMYB3R-2 transgenic rice is mediated by alteration in cell cycle and ectopic expression of stress genes. Plant Physiol 150(1):244–256. doi:10.1104/pp.108.133454
McElroy D, Blowers AD, Jenes B, Wu R (1991) Construction of expression vectors based on the rice actin 1 (Act1) 5′ region for use in monocot transformation. Mol Gen Genet MGG 231(1):150–160. doi:10.1007/bf00293832
Meng L, Bregitzer P, Zhang S, Lemaux PG (2003) Methylation of the exon/intron region in the Ubi1 promoter complex correlates with transgene silencing in barley. Plant Mol Biol 53(3):327–340. doi:10.1023/B:PLAN.0000006942.00464.e3
Mochida K, Yoshida T, Sakurai T, Yamaguchi-Shinozaki K, Shinozaki K, Tran L-S (2010) LegumeTFDB: an integrative database of Glycine max, Lotus japonicus and Medicago truncatula transcription factors. Bioinformatics 26(2):290–291
Odell JT, Nagy F, Chua N-H (1985) Identification of DNA sequences required for activity of the cauliflower mosaic virus 35S promoter. Nature 313(6005):810–812
Park SH, Yi N, Kim YS, Jeong MH, Bang SW, Choi YD, Kim JK (2010) Analysis of five novel putative constitutive gene promoters in transgenic rice plants. J Exp Bot 61(9):2459–2467. doi:10.1093/jxb/erq076
Reyes JC, Muro-Pastor MI, Florencio FJ (2004) The GATA family of transcription factors in Arabidopsis and rice. Plant Physiol 134(4):1718–1732
Schledzewski K, Mendel RR (1994) Quantitative transient gene expression: comparison of the promoters for maize polyubiquitin1, rice actin1, maize-derivedEmu andCaMV 35S in cells of barley, maize and tobacco. Transgenic Res 3(4):249–255. doi:10.1007/BF02336778
Schnurr JA, Guerra DJ (2000) The CaMV-35S promoter is sensitive to shortened photoperiod in transgenic tobacco. Plant Cell Rep 19(3):279–282
Soitamo A, Piippo M, Allahverdiyeva Y, Battchikova N, Aro E-M (2008) Light has a specific role in modulating Arabidopsis gene expression at low temperature. BMC Plant Biol 8(1):13
Takeda T, Suwa Y, Suzuki M, Kitano H, Ueguchi-Tanaka M, Ashikari M, Matsuoka M, Ueguchi C (2003) The OsTB1 gene negatively regulates lateral branching in rice. Plant J 33(3):513–520
Tang W, Page M (2013) Overexpression of the Arabidopsis AtEm6 gene enhances salt tolerance in transgenic rice cell lines. Plant Cell Tissue Organ Culture (PCTOC) 114(3):339–350. doi:10.1007/s11240-013-0329-8
Teakle GR, Manfield IW, Graham JF, Gilmartin PM (2002) Arabidopsis thaliana GATA factors: organisation, expression and DNA-binding characteristics. Plant Mol Biol 50(1):43–56
Urano J, Nakagawa T, Maki Y, Masumura T, Tanaka K, Murata N, Ushimaru T (2000) Molecular cloning and characterization of a rice dehydroascorbate reductase. FEBS Lett 466(1):107–111. doi:10.1016/S0014-5793(99)01768-8
Verdaguer B, de Kochko A, Fux CI, Beachy RN, Fauquet C (1998) Functional organization of the cassava vein mosaic virus (CsVMV) promoter. Plant Mol Biol 37(6):1055–1067
Wang J, Oard JH (2003) Rice ubiquitin promoters: deletion analysis and potential usefulness in plant transformation systems. Plant Cell Rep 22(2):129–134
Weeks JT, Anderson OD, Blechl AE (1993) Rapid production of multiple independent lines of fertile transgenic wheat (Triticum aestivum). Plant Physiol 102(4):1077–1084. doi:10.1104/pp.102.4.1077
Yang L, Ding J, Zhang C, Jia J, Weng H, Liu W, Zhang D (2005) Estimating the copy number of transgenes in transformed rice by real-time quantitative PCR. Plant Cell Rep 23(10–11):759–763. doi:10.1007/s00299-004-0881-0
Yoshida S, Tamaoki M, Shikano T, Nakajima N, Ogawa D, Motohide I, Hikaru S (2006) Cytosolic dehydroascorbate reductase is important for ozone tolerance in Arabidopsis thaliana. Plant Cell Physiol 47(2):304–308. doi:10.1093/pcp/pci246
Zhang W, McElroy D, Wu R (1991) Analysis of rice Act1 5′ region activity in transgenic rice plants. Plant Cell Online 3(11):1155–1165. doi:10.1105/tpc.3.11.1155
Zimmermann P, Laule O, Schmitz J, Hruz T, Bleuler S, Gruissem W (2008) Genevestigator transcriptome meta-analysis and biomarker search using rice and barley gene expression databases. Mol Plant 1(5):851–857
Acknowledgments
We thank Dr. Changlin Wang (Washington State University) for help with the manuscript. This research was supported by the National Natural Science Foundation of China (NSFC Grant No. 31100216) and by the Creative Foundation of the Anhui Agricultural Academy of Sciences (13C0101 & 14B0113).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Huw D. Jones.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, J., Xu, RF., Qin, RY. et al. Isolation and functional characterization of a novel rice constitutive promoter. Plant Cell Rep 33, 1651–1660 (2014). https://doi.org/10.1007/s00299-014-1644-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-014-1644-1