Abstract
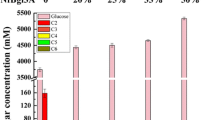
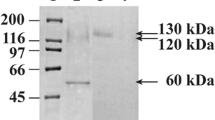
An intronless form of the bgl1 gene encoding an extracellular β-glucosidase from Trichoderma reesei was expressed in the yeast Saccharomyces cerevisiae under the control of the yeast GAL1 promoter. Transformation of a yeast strain with this vector resulted in transformants that produce and secrete active β-glucosidase into the growth medium. Additionally, active recombinant β-glucosidase protein was shown to be localized predominantly in the periplasmic space by using a p-nitrophenyl β-D-glycoside hydrolysis assay against fractionated yeast cells. The apparent size of the recombinant enzyme was 10–15 kDa larger than that of the native form. Treatment of the recombinant β-glucosidase with endoglycosidase-H indicated the apparent increase in size was due to N-linked glycosylation.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 10 September 1995
Rights and permissions
About this article
Cite this article
Cummings, C., Fowler, T. Secretion of Trichoderma reesei β-glucosidase by Saccharomyces cerevisiae. Curr Genet 29, 227–233 (1996). https://doi.org/10.1007/s002940050040
Issue Date:
DOI: https://doi.org/10.1007/s002940050040