Abstract
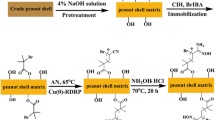
Bromoactylated apple seeds (BAS) was used as new source for the synthesis of environmental adsorbent. Apple seeds were modified by bromoacetyl bromide to obtain a macroinitiator for single electron transfer-living radical polymerization (SET-LRP) of acrylonitrile (AN). The amidoxime (AO) adsorbent was prepared by the reaction of the graft copolymer bromoactylated apple seeds (BAS)/polyacrylonitrile (BAS-g-PAN) with hydroxylamine. Amidoxime BAS-g-PAN (AO BAS-g-PAN) was used to remove Pb(II) and the maximum adsorption for Pb(II) was 1.58 mmol/g. AO BAS-g-PAN could also selectively adsorb Pb(II) from binary ion systems in the existence of Ag(I), Ni(II) and Zn(II). This efficient modifying approach provides a new powerful mean toward filling the gap on apple waste as a potential source for controlling heavy metal pollution.













Similar content being viewed by others
References
Demirbas A (2008) Heavy metal adsorption onto agro-based waste materials: a review. J Hazard Mater 157:220–229
Verma R, Gupta BD (2015) Detection of heavy metal ions in contaminated water by surface plasmon resonance based optical fibre sensor using conducting polymer and chitosan. Food Chem 166:568–575
Mishra B, Haack EA, Maurice PA, Bunker BA (2010) A spectroscopic study of the effects of a microbial siderophore on Pb adsorption to kaolinite. J Chem Geol 275:199–207
Wu X, Ma HW, Yang J, Wang FJ, Li Z (2012) Adsorption of Pb(II) from aqueous solution by a poly-elemental mesoporous adsorbent. J Appl Surf Sci 258:5516–5521
Rahman N, Haseen U (2014) Equilibrium modeling, kinetic, and thermodynamic studies on adsorption of Pb(II) by a hybrid inorganic–organic material: polyacrylamide zirconium(IV) iodate. Ind Eng Chem Res 53(19):8198–8207
Shi J, Zhao Z, Liang Z (2016) Adsorption characteristics of Pb(II) from aqueous solutions onto a natural biosorbent, fallen arborvitae leaves. Water Sci Technol 73(10):2422–2429
Niu Y, Qu R, Liu X (2014) Thiol-functionalized polysilsesquioxane as efficient adsorbent for adsorption of Hg(II) and Mn(II) from aqueous solution. Mater Res Bull 52(2):134–142
Bai L, Wang D, Chen H (2015) Synthesis of peanut shell/polyacrylonitrile copolymer via Cu(0)-mediated RDRP and its adsorption behavior after modification. Polym Bull 72(10):2455–2469
Xu J, Zhou CW, Wang RZ (2012) Lipase-coupling esterification of starch with octenyl succinic anhydride. Carbohydr Polym 87(3):2137–2144
Huijbrechts AML, Desse M, Budtova T (2008) Physicochemical properties of etherified maize starch. Carbohydr Polym 74(2):170–184
Liu Z, Yan D, Men H (2012) Post-crosslinking modification of thermoplastic starch/PVA blend films by using sodium hexametaphosphate. Carbohydr Polym 89(2):473–477
Masuda H, Horie T, Hubacz R (2013) Process intensification of continuous starch hydrolysis with a Couette–Taylor flow reactor. Chem Eng Res Des 91(11):2259–2264
Ma Z, Qian L, Yue Q (2011) Synthesis and characterization of a novel super-absorbent based on wheat straw. Bioresour Technol 102(3):2853–2858
Ratcliffe LPD, Blanazs A, Williams CN et al (2014) RAFT polymerization of hydroxy-functional methacrylic monomers under heterogeneous conditions: effect of varying the core-forming block. Polym Chem 5(11):3643–3655
Moad G, Rizzardo E, Thang SH (2008) Radical addition–fragmentation chemistry in polymer synthesis. Polymer 49(5):1079–1131
Barner Kowollik C, Perrier S (2008) The future of reversible addition fragmentation chain transfer polymerization. J Polym Sci Part A Polym Chem 46(17):5715–5723
Nguyen NH, Percec V (2010) Dramatic acceleration of SET-LRP of methyl acrylate during catalysis with activated Cu(0) wire. J Polym Sci Part A Polym Chem 48(22):5109–5119
Potisek SL, Davis DA, Sottos NR (2007) Mechanophore-linked addition polymers. J Am Chem Soc 129(45):13808–13809
Xuan J, Fleischmann S, Nguyen NH (2009) Cooperative and synergistic solvent effects in SET-LRP of MA. J Polym Sci Part A Polym Chem 47(21):5591–5605
Xin L, Hou C, Wang C (2010) Synthesis of porous acrylonitrile/methyl acrylate copolymer beads by suspended emulsion polymerization and their adsorption properties after amidoximation. J Hazard Mater 175(1–3):1014–1021
Guangxi Z, Hou C, Rongjun Q (2011) Synthesis of polyacrylonitrile-grafted cross-linked N-chlorosulfonamidated polystyrene via surface-initiated ARGET ATRP, and use of the resin in mercury removal after modification. J Hazard Mater 186(1):614–621
Zhihai Hao, Jiang Zhang, Hou Chen (2013) Preparation of polyacrylonitrile via SET-LRP catalyzed by lanthanum powder in the presence of VC. J Polym Sci Part A Polym Chem 51(19):4088–4094
Delong Liu, Hou Chen, Naiyi Ji (2013) Living radical polymerization of acrylonitrile catalyzed by copper with a high concentration of radical initiator and its application in removal of Ag(I) after modification. J Polym Sci Part A Polym Chem 51(2):340–346
Colella MB, Siggia S, Barnes RM (1980) Synthesis and characterization of a poly(acrylamidoxime) metal chelating resin. Anal Chem 52(6):967–972
Shaaban AF, Fadel DA, Mahmoud AA et al (2014) Synthesis of a new chelating resin bearing amidoxime group for adsorption of Cu(II), Ni(II) and Pb(II) by batch and fixed-bed column methods. J Environ Chem Eng 2(1):632–641
Saito K, Yamaguchi T, Uezu K (1990) Optimum preparation conditions of amidoxime hollow fiber synthesized by radiation-induced grafting. J Appl Polym Sci 39(10):2153–2163
Seebach D, Schaeffer L, Gessier F (2003) Enantioselective preparation of 2-aminomethyl carboxylic acid derivatives: solving the β2-amino acid problem with the chiral auxiliary 4-isopropyl-5,5-diphenyloxazolidin-2-one. Helv Chim Acta 86(6):1852–1861
Najafi Moghaddam P, Fareghi AR, Entezami AA (2013) Synthesis of biodegradable thermoplastic copolymers based on starch by atom transfer radical polymerization (ATRP): monolayer chain growth on starch. Starch/Staerke 65:210–218
El-Khouly AS, Takahashi Y, Saafan AA, Kenawy E, Hafiz YA (2011) Study of heavy metal ion absorbance by amidoxime group introduced to cellulose-graft-polyacrylonitrile. J Appl Polym Sci 120(2):866–873
Carro L, Barriada JL, Herrero R (2011) Adsorptive behaviour of mercury on algal biomass: competition with divalent cations and organic compounds. J Hazard Mater 192(1):284–291
Liu C, Bai R, Quan SL (2008) Selective removal of copper and lead ions by diethylenetriamine-functionalized adsorbent: behaviors and mechanisms. Water Res 42(6–7):1511–1522
Monier M, Abdel-Latif DA (2012) Preparation of cross-linked magnetic chitosan-phenylthiourea resin for adsorption of Hg(II), Cd(II) and Zn(II) ions from aqueous solutions. J Hazard Mater 209–210(1):240–249
Niu Y, Qu R, Sun C (2013) Adsorption of Pb(II) from aqueous solution by silica-gel supported hyperbranched polyamidoamine dendrimers. J Hazard Mater 244–245(2):276–286
Wen D, Shen-Yong Z, Jun-Tao L (2013) Preparation and adsorption properties of dialdehyde 8-aminoquinoline starch. Water Sci Technol 67(2):306–310
Wang DJ, Chen H, Xu H, Sun JM, Xu YY (2014) Preparation of wheat straw matrix-g-polyacrylonitrile-based adsorbent by set-lrp and its applications for heavy metal ion removal. ACS Sustain Chem Eng 2(7):1843–1848
Hao Z, Wang D, Chen H, Sun J, Xu Y (2014) Sweet potato starch residue as starting material to prepare polyacrylonitrile adsorbent via SI-SET-LRP. J Agric Food Chem 62(8):1765–1770
Long H, Zhao Z, Chai Y, Li X, Hua Z, Xiao Y, Yang Y (2015) Binding mechanism of the amidoxime functional group on chelating resins toward gallium(III) in Bayer liquor. Ind Eng Chem Res 54(33):8025–8030
Acknowledgements
The authors are grateful for the financial support by the National Natural Scientific Foundation of China (No. 51573075), the Program for New Century Excellent Talents in University (No. NCET-11-1028), the Natural Science Foundation for Distinguished Young Scholars of Shandong province (No. JQ201203), and the Program for Scientific Research Innovation Team in Colleges and universities of Shandong Province.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, D., Yu, L., Chen, H. et al. Use of apple seeds as new source for synthesis of polyacrylonitrile-based adsorbent to remove Pb(II). Polym. Bull. 74, 5231–5247 (2017). https://doi.org/10.1007/s00289-017-2012-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-017-2012-4