Abstract
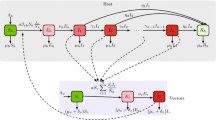
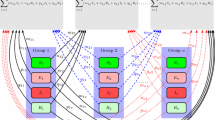
We develop a multi-group and multi-patch model to study the effects of population dispersal on the spatial spread of vector-borne diseases across a heterogeneous environment. The movement of host and/or vector is described by Lagrangian approach in which the origin or identity of each individual stays unchanged regardless of movement. The basic reproduction number \(\mathcal {R}_0\) of the model is defined and the strong connectivity of the host-vector network is succinctly characterized by the residence times matrices of hosts and vectors. Furthermore, the definition and criterion of the strong connectivity of general infectious disease networks are given and applied to establish the global stability of the disease-free equilibrium. The global dynamics of the model system are shown to be entirely determined by its basic reproduction number. We then obtain several biologically meaningful upper and lower bounds on the basic reproduction number which are independent or dependent of the residence times matrices. In particular, the heterogeneous mixing of hosts and vectors in a homogeneous environment always increases the basic reproduction number. There is a substantial difference on the upper bound of \(\mathcal {R}_0\) between Lagrangian and Eulerian modeling approaches. When only host movement between two patches is concerned, the subdivision of hosts (more host groups) can lead to a larger basic reproduction number. In addition, we numerically investigate the dependence of the basic reproduction number and the total number of infected hosts on the residence times matrix of hosts, and compare the impact of different vector control strategies on disease transmission.





Similar content being viewed by others
References
Allen LJS, Bolker BM, Lou Y, Nevai AL (2007) Asymptotic profiles of the steady states for an SIS epidemic patch model. SIAM J. Appl. Math. 67(5):1283–1309
Arino J (2009) Diseases in metapopulations. In: Ma Z, Zhou Y, Wu J (eds) Modeling and dynamics of infectious diseases, Ser. Contemp. Appl. Math. World Scientific, Singapore, pp 64–122
Auger P, Kouokam E, Sallet G, Tchuente M, Tsanou B (2008) The Ross-Macdonald model in a patchy environment. Math. Biosci. 216:123–131
Berman A, Plemmons RJ (1994) Nonnegative matrices in the mathematical sciences. SIAM, Philadelphia
Bichara D, Castillo-Chavez C (2016) Vector-borne diseases models with residence times—a Lagrangian perspective. Math. Biosci. 281:128–138
Bichara D, Holechek SA, Velázquez-Castro J, Murillo AL, Castillo-Chavez C (2016) On the dynamics of dengue virus type 2 with residence times and vertical transmission. Lett. Biomath. 3(1):140–160
Bichara D, Iggidr A (2018) Multi-patch and multi-group epidemic models: a new framework. J. Math. Biol. 77:107–134
Bichara D, Iggidr A, Yacheur S (2021) Effects of heterogeneity and global dynamics of weakly connected subpopulations. Math. Model. Nat. Phenom. 16:44
Bichara D, Kang Y, Castillo-Chavez C, Horan R, Perrings C (2015) SIS and SIR epidemic models under virtual dispersal. Bull. Math. Biol. 77(11):2004–2034
Castillo-Chavez C, Bichara D, Morin BR (2016) Perspectives on the role of mobility, behavior, and time scales in the spread of diseases. Proc. Natl. Acad. Sci. U. S. A. 113(51):14582–14588
Castillo-Chavez C, Thieme HR (1995) Asymptotically autonomous epidemic models. In: Arino O, Axelrod DE, Kimmel M, Langlais M (eds) Mathematical population dynamics: analysis of heterogeneity. Wuerz, Winnipeg, pp 33–50
Chen X, Gao D (2020) Effects of travel frequency on the persistence of mosquito-borne diseases. Discrete Contin. Dyn. Syst. Ser. B 25(12):4677–4701
Chinazzi M, Davis JT, Ajelli M, Gioannini C, Litvinova M, Merler S, Piontti AP, Mu K, Rossi L, Sun K, Viboud C, Xiong X, Yu H, Halloran ME, Longini IM Jr, Vespignani A (2020) The effect of travel restrictions on the spread of the 2019 novel coronavirus (COVID-19) outbreak. Science 368(6489):395–400
Citron DT, Guerra CA, Dolgert AJ, Wu SL, Henry JM, Sánchez HM, Smith DL (2021) Comparing metapopulation dynamics of infectious diseases under different models of human movement. Proc. Natl. Acad. Sci. U. S. A. 118(18):e2007488118
Cosner C, Beier JC, Cantrell RS, Impoinvil D, Kapitanski L, Potts MD, Troyo A, Ruan S (2009) The effects of human movement on the persistence of vector-borne diseases. J. Theor. Biol. 258(4):550–560
Diekmann O, Heesterbeek JAP, Metz JAJ (1990) On the definition and the computation of the basic reproduction ratio \(R_0\) in models for infectious diseases in heterogeneous populations. J. Math. Biol. 28(4):365–382
Diekmann O, Heesterbeek JAP, Roberts MG (2010) The construction of next-generation matrices for compartmental epidemic models. J. R. Soc. Interface 7:873–885
Ducrot A, Sirima SB, Somé B, Zongo P (2009) A mathematical model for malaria involving differential susceptibility, exposedness and infectivity of human host. J. Biol. Dyn. 3(6):574–598
Dye C, Hasibeder G (1986) Population dynamics of mosquito-borne disease: effects of flies which bite some people more frequently than others. Trans. R. Soc. Trop. Med. Hyg. 80(1):69–77
Gao D (2019) Travel frequency and infectious diseases. SIAM J. Appl. Math. 79(4):1581–1606
Gao D (2020) How does dispersal affect the infection size? SIAM J. Appl. Math. 80(5):2144–2169
Gao D, Cosner C, Cantrell RS, Beier JC, Ruan S (2013) Modeling the spatial spread of Rift Valley fever in Egypt. Bull. Math. Biol. 75:523–542
Gao D, Dong C-P (2020) Fast diffusion inhibits disease outbreaks. Proc. Am. Math. Soc. 148(4):1709–1722
Gao D, Lou Y, Ruan S (2014) A periodic Ross-Macdonald model in a patchy environment. Discrete Contin. Dyn. Syst. Ser. B 19(10):3133–3145
Gao D, Ruan S (2011) An SIS patch model with variable transmission coefficients. Math. Biosci. 232(2):110–115
Gao D, van den Driessche P, Cosner C (2019) Habitat fragmentation promotes malaria persistence. J. Math. Biol. 79(6):2255–2280
Gao D, Yuan X (2023) A hybrid Eulerian–Lagrangian model for vector-borne diseases, under review
Gatto M, Bertuzzo E, Mari L, Miccoli S, Carraro L, Casagrandi R, Rinaldo A (2020) Spread and dynamics of the COVID-19 epidemic in Italy: effects of emergency containment measures. Proc. Natl. Acad. Sci. U. S. A. 117(19):10484–10491
Gösgens M, Hendriks T, Boon M, Steenbakkers W, Heesterbeek H, van der Hofstad R, Litvak N (2021) Trade-offs between mobility restrictions and transmission of SARS-CoV-2. J. R. Soc. Interface 18(175):20200936
Hasibeder G, Dye C (1988) Population dynamics of mosquito-borne disease: persistence in a completely heterogeneous environment. Theor. Popul. Biol. 33(1):31–53
Horn RA, Johnson CR (2013) Matrix analysis, 2nd edn. Cambridge University Press, New York
Hsieh Y-H, van den Driessche P, Wang L (2007) Impact of travel between patches for spatial spread of disease. Bull. Math. Biol. 69(4):1355–1375
Iggidr A, Sallet G, Souza MO (2016) On the dynamics of a class of multi-group models for vector-borne diseases. J. Math. Anal. Appl. 441(2):723–743
Ikejezie J, Shapiro CN, Kim J, Chiu M, Almiron M, Ugarte C, Espinal MA, Aldighieri S (2017) Zika virus transmission-Region of the Americas, May 15, 2015-December 15, 2016. MMWR Morb. Mortal. Wkly. Rep. 66(12):329–334
Lajmanovich A, Yorke JA (1976) A deterministic model for gonorrhea in a nonhomogeneous population. Math. Biosci. 28:221–236
Lasalle JP (1976) The stability of dynamical systems. SIAM, Philadelphia
Li C-K, Schneider H (2002) Applications of Perron-Frobenius theory to population dynamics. J. Math. Biol. 44:450–462
Liu M, Fu X, Zhao D (2021) Dynamical analysis of an SIS epidemic model with migration and residence time. Int. J. Biomath. 14:2150023
Lou Y, Wu J (2017) Modeling Lyme disease transmission. Infect. Dis. Model. 2(2):229–243
Macdonald G (1957) The epidemiology and control of Malaria. Oxford University Press, London
Manore CA, Hickmann KS, Xu S, Wearing HJ, Hyman JM (2014) Comparing dengue and chikungunya emergence and endemic transmission in A. aegypti and A. albopictus. J. Theor. Biol. 356:174–191
Mead PS (2015) Epidemiology of Lyme disease. Infect. Dis. Clin. North Am. 29(2):187–210
Misra AK, Sharma A, Shukla JB (2011) Modeling and analysis of effects of awareness programs by media on the spread of infectious diseases. Math. Comput. Model. 53:1221–1228
Moreno V, Espinoza B, Barley K, Paredes M, Bichara D, Mubayi A, Castillo-Chavez C (2017) The role of mobility and health disparities on the transmission dynamics of Tuberculosis. Theor. Biol. Med. Model. 14:3
Moreno V, Espinoza B, Bichara D, Holechek SA, Castillo-Chavez C (2017) Role of short-term dispersal on the dynamics of Zika virus in an extreme idealized environment. Infect. Dis. Model. 2(1):21–34
Pérez-Molina JA, Molina I (2018) Chagas disease. Lancet 391(10115):82–94
Reisen WK, Wheeler SS (2019) Overwintering of West Nile Virus in the United States. J. Med. Entomol. 56(6):1498–1507
Rodríguez DJ, Torres-Sorando L (2001) Models of infectious diseases in spatially heterogeneous environments. Bull. Math. Biol. 63(3):547–571
Ross R (1911) The Prevention of Malaria. John Murray, London
Ruan S, Wu J (2009) Modeling spatial spread of communicable diseases involving animal hosts. In: Cantrell S, Cosner C, Ruan S (eds) Spatial ecology. Chapman and Hall/CRC, Boca Raton, pp 293–316
Ruan S, Xiao D, Beier JC (2006) On the delayed Ross-Macdonald model for malaria transmission. Bull. Math. Biol. 70(4):1098–1114
Ruktanonchai NW, Smith DL, De Leenheer P (2016) Parasite sources and sinks in a patched Ross-Macdonald malaria model with human and mosquito movement: Implications for control. Math. Biosci. 279:90–101
Salmani M, van den Driessche P (2006) A model for disease transmission in a patchy environment. Discrete Contin. Dyn. Syst. Ser. B 6(1):185–202
Schneider H (1984) Theorems on \(M\)-splittings of a singular \(M\)-matrix which depend on graph structure. Linear Algebra Appl. 58:407–424
Shuai Z, van den Driessche P (2013) Global stability of infectious disease models using Lyapunov functions. SIAM J. Appl. Math. 73:1513–1532
Smith HL (1995) Monotone dynamical systems: an introduction to the theory of competitive and cooperative systems. American Mathematical Society, Providence
Tang B, Wang X, Li Q, Bragazzi NL, Tang S, Xiao Y, Wu J (2020) Estimation of the transmission risk of the 2019-nCoV and its implication for public health interventions. J. Clin. Med. 9(2):462
Torres-Sorando L, Rodríguez DJ (1997) Models of spatio-temporal dynamics in malaria. Ecol. Model. 104:231–240
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 180:29–48
Verdonschot PFM, Besse-Lototskaya AA (2014) Flight distance of mosquitoes (Culicidae): a metadata analysis to support the management of barrier zones around rewetted and newly constructed wetlands. Limnologica 45:69–79
Wang X, Liu S, Wang L, Zhang W (2015) An epidemic patchy model with entry-exit screening. Bull. Math. Biol. 77(7):1237–1255
Wang W, Mulone G (2003) Threshold of disease transmission in a patch environment. J. Math. Anal. Appl. 285(1):321–335
Wang W, Zhao X-Q (2004) An epidemic model in a patchy environment. Math. Biosci. 190(1):97–112
World Health Organization (2020a) Dengue and Severe Dengue, https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue
World Health Organization (2020b) Vector-borne Disease, https://www.who.int/news-room/fact-sheets/detail/vector-borne-diseases
World Health Organization (2020c) World Malaria Report 2020, https://www.who.int/teams/global-malaria-programme/reports/world-malaria-report-2020
Wu JT, Leung K, Leung GM (2020) Nowcasting and forecasting the potential domestic and international spread of the 2019-nCoV outbreak originating in Wuhan, China: A modelling study. Lancet 395(10225):689–697
Zhang J, Cosner C, Zhu H (2018) Two-patch model for the spread of West Nile virus. Bull. Math. Biol. 80(4):840–863
Zhao X-Q (2017) Dynamical systems in population biology, 2nd edn. Springer-Verlag, New York
Zhao X-Q, Jing Z (1996) Global asymptotic behavior in some cooperative systems of functional differential equations. Can. Appl. Math. Q. 4(4):421–444
Acknowledgements
We sincerely thank the handling editor Dr. Andrea Pugliese and anonymous reviewers for their careful reading and constructive comments, and Drs. Derdei Bichara, Yijun Lou and Lei Zhang for helpful discussions. This work was supported by the National Natural Science Foundation of China (12071300), the Natural Science Foundation of Shanghai (20ZR1440600 and 20JC1413800), and the CSU Office of Research through a startup grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Professor Chris Cosner on his 70th birthday.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix A: Variable and notation index
Symbol | Meaning | Page |
|---|---|---|
\(H_i\) | Total host population of group i | 5 |
\(S_i^h\) | Number of susceptible hosts in group i | 5 |
\(I_i^h\) | Number of infected hosts in group i | 5 |
\(N_j^v\) | Total vector population of patch j | 5 |
\(S_j^v\) | Number of susceptible vectors in group j | 5 |
\(I_j^v\) | Number of infected vectors in group j | 5 |
\(I_{ik}^h\) | Number of infected hosts of group i in patch k | 18 |
\(I_{jk}^v\) | Number of infected vectors of patch j in patch k | 18 |
\(I_{i}^{hf}\) | Number of infected hosts of group i that are frequent travelers | 35 |
\(I_{i}^{hu}\) | Number of infected hosts of group i that are unfrequent travelers | 35 |
\(\Omega _h\) | Set of host groups | 5 |
\(\Omega _v\) | Set of vector patches | 5 |
\(\Omega _v^0\) | Set of vector patches that are visited by at least one host group | 5 |
\(\Omega _v^j\) | Set of vector patches that are visited by host group j | 22 |
\(\Gamma \) | Positively invariant region for model (2.2) | 9 |
\(E_0\) | Disease-free equilibrium of model (2.2) | 9 |
\(E^*\) | Endemic equilibrium of model (2.2) | 17 |
F | New infection matrix for model (2.2) | 9 |
V | Transition matrix for model (2.2) | 9 |
\(\rho \) | Spectral radius of a square matrix | 9 |
\({\mathscr {A}}\) | Block of matrix F at position (1,2) | 9 |
\({\mathscr {B}}\) | Block of matrix F at position (2,1) | 9 |
\({\mathscr {C}}\) | Block of matrix V at position (1,1) | 9 |
\({\mathscr {D}}\) | Block of matrix V at position (2,2) | 9 |
\(\alpha _{ij}\) | The (i, j)-entry of matrix \({\mathscr {A}}{\mathscr {D}}^{-1}{\mathscr {B}}{\mathscr {C}}^{-1}\) | 10 |
\(\beta _{ij}\) | The (i, j)-entry of matrix \({\mathscr {B}}{\mathscr {C}}^{-1}{\mathscr {A}}{\mathscr {D}}^{-1}\) | 10 |
M | A Sign equivalent form of \(FV^{-1}\) | 11 |
W | Host-vector contact matrix in terms of residence times matrices | 11 |
\({\mathcal {G}}\) | Infectious disease network | 13 |
\(\mathcal {R}_0\) | Basic reproduction number of model (2.2) | 9 |
\(\mathcal {R}_0^{(k)}\) | Basic reproduction number of patch k in disconnection | 18 |
\(\mathcal {R}_0^{[k]}\) | Basic reproduction number of patch k in isolation | 19 |
\({\hat{\mathcal {R}}}_{ijskr}\) | Basic reproduction number that involves host groups i, j and | 20 |
vector patches s, k, r, and depends on the ratio of overall | ||
population sizes of vectors and hosts, i.e., V/H | ||
\({\hat{\mathcal {R}}}_{jjskr}\) | \({\hat{\mathcal {R}}}_{ijskr}\) in the case of \(i=j\) | 23 |
\({\hat{\mathcal {R}}}_{ijk}\) | \({\hat{\mathcal {R}}}_{ijskr}\) in the case of \(s=r=k\) (no vector movement) | 31 |
\({\hat{\mathcal {R}}}_{jjk}\) | \({\hat{\mathcal {R}}}_{ijk}\) in the case of \(i=j\) | 31 |
\(\mathcal {R}_0(m/n)\) | \(\mathcal {R}_0\) of model (2.2) containing m host groups and n vector patches | 23 |
\(\mathcal {R}_{ijskr}\) | Basic reproduction number that involves host groups i, j and vector | 26 |
patches s, k, r, and depends on the ratio of population sizes | ||
of vector patch s and host group j, i.e., \(V_s/H_j\) | ||
\(\mathcal {R}_{jjskr}\) | \(\mathcal {R}_{ijskr}\) in the case of \(i=j\) | 28 |
\(\mathcal {R}_{ijk}\) | \(\mathcal {R}_{ijskr}\) in the case of \(s=r=k\) (no vector movement) | 31 |
\(\mathcal {R}_{jjk}\) | \(\mathcal {R}_{ijk}\) in the case of \(i=j\) | 31 |
\({\tilde{\mathcal {R}}}_{ijk}\) | Basic reproduction number that involves host group k and vector | 32 |
patches i, j, depends on the ratio of population sizes of vector patch |
Symbol | Meaning | Page |
|---|---|---|
i and host group k, i.e., \(V_i/H_k\), and has no vector movement | ||
\({\tilde{\mathcal {R}}}_0^{(k)}\) | Basic reproduction number of patch k in disconnection with only | 33 |
vector movement before disconnection | ||
\(h_i\) | Proportion of hosts in group i, i.e., \(H_i/H\) | 20 |
\(v_j\) | Proportion of vectors of patch j, i.e., \(V_j/V\) | 20 |
\(p_k\) | Proportion of hosts from all host groups that commute to patch k | 23 |
\(q_k\) | Proportion of vectors from all vector patches that commute to patch k | 23 |
\({\hat{L}}\) | \({\mathscr {A}}{\mathscr {D}}^{-1}{\mathscr {B}}{\mathscr {C}}^{-1}\) in the case of all \({\hat{\mathcal {R}}}_{ijskr}\equiv 1\) | 21 |
L | \({\mathscr {A}}{\mathscr {D}}^{-1}{\mathscr {B}}{\mathscr {C}}^{-1}\) in the case of all \(\mathcal {R}_{ijskr}\equiv 1\) | 27 |
\({\tilde{A}}\) | A spectral radius equivalent form of \({\mathscr {A}}{\mathscr {D}}^{-1}{\mathscr {B}}{\mathscr {C}}^{-1}\) | 51 |
\({\tilde{L}}\) | \({\tilde{A}}\) in the case of all \({\hat{\mathcal {R}}}_{jjskr}\equiv 1\) | 51 |
\({\check{L}}\) | \({\mathscr {B}}{\mathscr {C}}^{-1}{\mathscr {A}}{\mathscr {D}}^{-1}\) in the case of all \({\tilde{\mathcal {R}}}_{ijk}\equiv 1\) (no vector movement) | 53 |
Appendix B: Derivation of matrix W
-
(1)
The vector-borne disease considered here cannot be directly transmitted from host to host, so the (1, 1) block in Table 2 is \(0_{m\times m}\).
-
(2)
Suppose the hosts of group \(i~(1\le i\le m)\) spend part of time in patch \(j~(1\le j\le k)\). If an infected host of group i is introduced, then this infected host can visit patch j and infect the susceptible vectors of patch j. On the other hand, if an infected vector of patch j is introduced, then this infected vector can infect the susceptible hosts of group i who visit patch j. So, the (1, 2) and (2, 1) blocks in Table 2 are \(P_{1}\) and \(P_{1}^{\textrm{T}}\), respectively.
-
(3)
Since no host visits the last \(n-k\) patches, the disease transmission from the infected hosts of group \(i~(1\le i\le m)\) to the susceptible vectors of patch \(j~(k+1\le j\le n)\) in patch j cannot occur. Similarly, the disease transmission from the infected vectors of patch j to the susceptible hosts of group i in patch j is impossible. So, the (1, 3) and (3, 1) blocks in Table 2 are \(0_{m\times (n-k)}\) and \(0_{(n-k)\times m}\), respectively.
-
(4)
Suppose the vectors of patch \(j~(1\le j\le k)\) spend part of time in patch \(l~(1\le l\le k)\). If an infected vector of patch j is introduced, then this infected vector can visit patch l and infect the susceptible hosts in patch l (the first k patches have host visitors). The infected hosts in patch l can then infect the susceptible vectors of patch l. On the other hand, if an infected vector of patch l is introduced, then this infected vector can infect the susceptible hosts in patch l. The infected hosts in patch l can infect the susceptible vectors of patch j who visit patch l. In other words, the visit of vectors of patch j to patch l has the same transmission impact as the visit of vectors of patch l to patch j. So, the (2, 2) block in Table 2 is \(Q_1+Q_1^{\textrm{T}}\).
-
(5)
Suppose the vectors of patch \(l~(k+1\le l\le n)\) spend part of time in patch \(j~(1\le j\le k)\). If an infected vector of patch l is introduced, then this infected vector can visit patch j and infect the susceptible hosts in patch j (the first k patches have host visitors). The infected hosts in patch j can then infect the susceptible vectors of patch j. On the other hand, if an infected vector of patch j is introduced, then this infected vector can infect the susceptible hosts in patch j. The infected hosts in patch j can infect the susceptible vectors of patch l who visit patch j. Since the last \(n-k\) patches are host-free, the visit of the vectors of the first k patches to the last \(n-k\) patches does not cause disease transmission. So, the (3, 2) and (2, 3) blocks in Table 2 are \(Q_2\) and \(Q_2^{\textrm{T}}\), respectively.
-
(6)
The last \(n-k\) patches are host-free, so the movement of the vectors of the last \(n-k\) patches between the last \(n-k\) patches produces no infection. So, the (3, 3) block in Table 2 is \(0_{(n-k)\times (n-k)}\).
Appendix C: Proof of Theorem 3.2
Proof
Necessity. It follows from the nonnegativity of matrices \(P_1\), \(Q_1\), and \(Q_2\) that
Thus, the irreducibility of the matrix M implies the irreducibility of the matrix \(W^2\). Hence W is also irreducible. Suppose not, i.e., W is reducible, then there exists a permutation matrix S such that
and hence
Therefore \(W^2\) is reducible, a contradiction.
Sufficiency. Suppose that the matrix W is irreducible, then the directed graph generated by W, denoted by \({\mathcal {G}}(W)\), is strongly connected (Horn and Johnson 2013). We write the sets of nodes of the directed graphs \({\mathcal {G}}(W)\) and \({\mathcal {G}}(M)\) as
respectively. Denote \(W=(w_{ij})_{(m+n)\times (m+n)}\) and \(M=(m_{ij})_{(m+n)\times (m+n)}\), where
and
Based on the symmetry of matrices W and M, and the number of nonzero blocks in matrix W, we have three parts to show that: if there is a directed path between a given pair of nodes of graph \({\mathcal {G}}(W)\), then there is also a directed path between the corresponding pair of nodes of graph \({\mathcal {G}}(M)\). Therefore, the strong connectivity of \({\mathcal {G}}(W)\) implies the strong connectivity of \({\mathcal {G}}(M)\).
-
(1)
Consider the (1, 2) block (or called submatrix) of the block (or called partitioned) matrix W. Suppose \(p_{lj}>0\) for some \(l\in \{1,\dots ,m\}\) and \(j\in \{1,\dots ,k\}\). It follows from (C.1) and the symmetry of W that
$$\begin{aligned} w_{l,m+j}=w_{m+j,l}=p_{lj}>0. \end{aligned}$$So, there is a bidirectional edge connecting nodes \(W_{l}\) and \(W_{m+j}\) of graph \({\mathcal {G}}(W)\). Meanwhile, it follows from (C.2), the symmetry of M and
$$\begin{aligned} p_{lj}>0\ \text{ and } \ q_{jj}>0, \end{aligned}$$that
$$\begin{aligned} m_{l,m+j}=m_{m+j,l}=\sum \limits _{r=1}^{k}p_{lr}q_{jr}\ge p_{lj}q_{jj}>0. \end{aligned}$$Hence, there is also a bidirectional edge connecting nodes \(M_{l}\) and \(M_{m+j}\) of graph \({\mathcal {G}}(M)\).
-
(2)
Consider the (2, 2) block of W. Suppose \(q_{sj}>0\) for some \(s,\,j\in \{1,\dots ,k\}\), then
$$\begin{aligned} w_{m+s,m+j}=w_{m+j,m+s}=q_{sj}+q_{js}\ge q_{sj}>0. \end{aligned}$$So, there is a bidirectional edge connecting nodes \(W_{m+s}\) and \(W_{m+j}\) of \({\mathcal {G}}(W)\). Since at least one host group visits patch j, there exists some \(i\in \{1,\dots ,m\}\) such that \(p_{ij}>0\), which means that
$$\begin{aligned} m_{i,m+s}=m_{m+s,i}=\sum \limits _{r=1}^{k}p_{ir}q_{sr}\ge p_{ij}q_{sj}>0. \end{aligned}$$So, the nodes \(M_{i}\) and \(M_{m+s}\) of \({\mathcal {G}}(M)\) are connected by a bidirectional edge. Moreover, it follows from (C.2), the symmetry of M and
$$\begin{aligned} p_{ij}>0\;\; \text{ and } \;\; q_{jj}>0, \end{aligned}$$that
$$\begin{aligned} m_{i,m+j}=m_{m+j,i}=\sum \limits _{r=1}^{k}p_{ir}q_{jr}\ge p_{ij}q_{jj}>0. \end{aligned}$$So, the nodes \(M_{i}\) and \(M_{m+j}\) of \({\mathcal {G}}(M)\) are also connected by a bidirectional edge. Thus, there is a bidirectional path passing nodes \(M_{m+s}\) and \(M_{m+j}\) of \({\mathcal {G}}(M)\).
-
(3)
Consider the (3, 2) block of W. Suppose \(q_{cj}>0\) for some \(c\in \{k+1,\dots ,n\}\) and \(j\in \{1,\dots ,k\}\), then
$$\begin{aligned} w_{m+c,m+j}=w_{m+j,m+c}=q_{cj}>0. \end{aligned}$$So, there is a bidirectional edge connecting nodes \(W_{m+c}\) and \(W_{m+j}\) of graph \({\mathcal {G}}(W)\). Similarly, there exists some \(i\in \{1,\dots ,m\}\) such that \(p_{ij}>0\), which implies that
$$\begin{aligned} m_{i,m+c}=m_{m+c,i}=\sum \limits _{r=1}^{k}p_{ir}q_{cr}\ge p_{ij}q_{cj}>0. \end{aligned}$$So, the nodes \(M_{i}\) and \(M_{m+c}\) of graph \({\mathcal {G}}(M)\) are connected by a bidirectional edge. Moreover, it again follows from (C.2), the symmetry of M and
$$\begin{aligned} p_{ij}>0\ \text{ and } \ q_{jj}>0, \end{aligned}$$that
$$\begin{aligned} m_{i,m+j}=m_{m+j,i}=\sum \limits _{r=1}^{k}p_{ir}q_{jr}\ge p_{ij}q_{jj}>0. \end{aligned}$$There is a bidirectional edge connecting nodes \(M_{i}\) and \(M_{m+j}\) of graph \({\mathcal {G}}(M)\). Thus, the nodes \(M_{m+c}\) and \(M_{m+j}\) of graph \({\mathcal {G}}(M)\) are connected by a bidirectional path.
In conclusion, if the directed graph \({\mathcal {G}}(W)\) is strongly connected, then the directed graph \({\mathcal {G}}(M)\) is also strongly connected. So, the matrix M is irreducible. \(\square \)
Appendix D: Proof of Theorem 3.13
Proof
Let \({\varvec{f}}=(f_{1},\dots ,f_{m+n})\) be the vector field generated by (2.2) and \(\phi _t\) the associated semiflow. We rewrite the model system (2.2) as \({\varvec{x}}'={\varvec{f}}({\varvec{x}})\), where
By Proposition 2.2, it suffices to consider system (2.2) in the positively invariant set \(\Gamma \). We complete the proof by verifying the conditions of Corollary 3.2 in Zhao and Jing (1996).
-
(1)
Direct calculation yields the Jacobian matrix of system (2.2) at \({\varvec{x}}\), denoted by
$$\begin{aligned} D{\varvec{f}}({\varvec{x}})=\left( \frac{\partial f_{s}}{\partial x_{r}}\right) _{(m+n)\times (m+n)}, \end{aligned}$$where
$$\begin{aligned} \frac{\partial f_{s}}{\partial x_{r}}= {\left\{ \begin{array}{ll} -b_{s}\sum \limits _{k\in \Omega _{v}^{0}}a_{k}\dfrac{\sum _{j\in \Omega _{v}}q_{jk}I_{j}^{v}}{\sum _{l\in \Omega _{h}}p_{lk}H_{l}}\,p_{sk}- \left( \gamma _{s}+\mu _{s}^{h}\right) , &{} 1\le s=r\le m, \\ b_{s}\left( H_{s}-I_{s}^{h}\right) \sum \limits _{k\in \Omega _{v}^{0}}a_{k}\dfrac{p_{sk}q_{r-m,k}}{\sum _{l\in \Omega _{h}}p_{lk}H_{l}}, &{} 1\le s\le m,\;m+1\le r\le m+n, \\ c_{r}\left( V_{s-m}-I_{s-m}^{v}\right) \sum \limits _{k\in \Omega _{v}^{0}}a_{k}\dfrac{q_{s-m,k}p_{rk}}{\sum _{l\in \Omega _{h}}p_{lk}H_{l}}, &{} m+1\le s\le m+n,\;1\le r\le m, \\ -\sum \limits _{k\in \Omega _{v}^{0}}a_{k}\dfrac{\sum _{i\in \Omega _{h}}c_{i}p_{ik}I_{i}^{h}}{\sum _{l\in \Omega _{h}}p_{lk}H_{l}}\,q_{s-m,k}-\mu _{s-m}^{v}, &{} m+1\le s=r\le m+n,\\ 0, &{} \text{ otherwise }. \end{array}\right. } \end{aligned}$$Clearly, the matrix \(D\varvec{f}(\varvec{x})\) is quasi-positive (or called essentially nonnegative) on \(\Gamma \). So, system (2.2) is cooperative on \(\Gamma \). The strong connectivity of the host-vector network implies that \(D\varvec{f}(\varvec{0})=F-V\) is irreducible. It follows that \(D\varvec{f}(\varvec{x})\) is irreducible in \(\mathring{\Gamma }\), the interior of \(\Gamma \). Moreover, any nonzero solution starting at the boundary of \(\Gamma \) will immediately enter and stay in \(\mathring{\Gamma }\).
-
(2)
Clearly, \({\varvec{f}}({\varvec{0}})={\varvec{0}}\), and \(f_{i}({\varvec{x}})\ge 0\) for all \({\varvec{x}}\in \Gamma \) with \(x_i=0\), \(i=1,\dots ,m+n\). Meanwhile, \(f_{i}(\varvec{x})=-(\gamma _i+\mu _i^h)H_i<0\) for all \({\varvec{x}}\in \Gamma \) with \(x_i=H_i\), \(i=1,\dots ,m\), and \(f_{i}(\varvec{x})=-\mu _{i-m}^vV_{i-m}<0\) for all \({\varvec{x}}\in \Gamma \) with \(x_i=V_{i-m}\), \(i=m+1,\dots ,m+n\).
-
(3)
For any \(\xi \in (0,1)\) and \({\varvec{x}}\in \Gamma \) with \({\varvec{x}}\gg \varvec{0}\), we have
$$\begin{aligned} \begin{aligned} f_{i}(\xi {\varvec{x}})-\xi f_{i}({\varvec{x}}) =\xi \,(1-\xi )\,b_{i}\sum _{k\in \Omega _{v}^{0}}a_{k}\frac{\sum _{j\in \Omega _{v}}q_{jk}I_{j}^{v}}{\sum _{l\in \Omega _{h}}p_{lk}H_{l}}\,p_{ik}\,I_{i}^{h}>0, \quad i\in \Omega _{h}, \end{aligned} \end{aligned}$$and
$$\begin{aligned} \begin{aligned} f_{m+j}(\xi {\varvec{x}})-\xi f_{m+j}({\varvec{x}}) =\xi \,(1-\xi )\, \sum _{k\in \Omega _{v}^{0}}a_{k}\frac{\sum _{i\in \Omega _{h}}c_{i}p_{ik}I_{i}^{h}}{\sum _{l\in \Omega _{h}}p_{lk}H_{l}}\,q_{jk}\, I_{j}^{v}>0, \quad j\in \Omega _{v}. \end{aligned} \end{aligned}$$Hence, \({\varvec{f}}\) is strictly sublinear on \(\Gamma \).
It follows from Theorem 2 in van den Driessche and Watmough (2002) or Theorem A.1 in Diekmann et al. (2010) that
By Corollary 3.2 in Zhao and Jing (1996), the global asymptotic stability of system (2.2) is proved. Moreover, the strong monotonicity of \(\phi _t\) implies that the unique endemic equilibrium satisfies \(\varvec{0}\ll E^*\ll (H_1,\dots ,H_m,V_1,\dots ,V_n)\). \(\square \)
Appendix E: Proof of Theorem 4.2
Proof
According to (3.1) and (3.2a), we have
Define
Clearly, the matrices \({\tilde{A}}\) and \({\mathscr {A}}{\mathscr {D}}^{-1}{\mathscr {B}}{\mathscr {C}}^{-1}\) have the same spectral radius. Denote
So,
which implies that
Notice that \({\hat{L}}^{\textrm{T}}={\tilde{L}}\), so it follows from the estimates of \(\rho ({\tilde{L}})\) in the proof of Theorem 4.1 that this theorem is proved. \(\square \)
Appendix F: Proof of Theorem 4.9
Proof
The proof of Theorem 4.2 indicates that \(\mathcal {R}_0=\sqrt{\rho ({\tilde{A}})}\) where
Denote
then
Define \({\hat{S}}=\mathop {\textrm{diag}}\{H_1,\ldots ,H_m\}\). A similarity transformation finds \({\hat{S}}U{\hat{S}}^{-1}=L\) and hence \(\rho (U)=\rho (L)\). The proof is complete by using the estimates on \(\rho (L)\) in Theorem 4.7. \(\square \)
Appendix G: Proof of Theorem 4.12
Proof
It follows from (4.11b) that \({\mathscr {B}}{\mathscr {C}}^{-1}{\mathscr {A}}{\mathscr {D}}^{-1} =(\beta _{ij})_{n\times n}\) where
Define
Clearly,
It follows from Corollary 8.1.29 in Horn and Johnson (2013) that
implies that \(\rho ({\check{L}})\le n\). So the upper bound of \(\mathcal {R}_0\) is proved.
On the other hand, since \({\check{L}}\) is a real symmetric matrix, the Rayleigh quotient theorem (Horn and Johnson 2013) gives
Choosing \({\check{x}}_{i}=1/\sqrt{n}\) for all \(i\in \Omega _{v}\) and applying the Cauchy–Schwarz inequality yield
So, we have \(\rho \left( {\check{L}}\right) \ge n/m\) and the proof is complete. \(\square \)
Appendix H: Proof of Theorem 4.16
Proof
Using the next generation matrix method (Diekmann et al. 1990; van den Driessche and Watmough 2002), the basic reproduction numbers of models (4.13) and (4.14) are
respectively, where
Clearly,
where \(C_i\) and \(D_i\) are the trace and determinant of matrix \({\mathscr {B}}_{i}{\mathscr {C}}_{i}^{-1}{\mathscr {A}}_{i}{\mathscr {D}}_{i}^{-1}\), respectively.
The inequality \(\mathcal {R}_{1}\ge \mathcal {R}_{2}\) is equivalent to
According to Theorem 3.9 in Gao (2019) or Theorem 3.3 in Chen and Gao (2020), it suffices to show that
A straightforward but tedious algebraic calculation gives
with equality if and only if \(P^f=P^u\), and
with equality if and only if \(K_1=0\) or \(K_2=0\), i.e., \(\mathcal {R}_1^{(1)}=\mathcal {R}_1^{(2)}\) or \(P^f=P^u\), where
Consequently, \(\mathcal {R}_{1}\ge \mathcal {R}_{2}\) with equality if and only if \(\mathcal {R}_1^{(1)}=\mathcal {R}_1^{(2)}\) or \(P^f=P^u\). \(\square \)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, D., Cao, L. Vector-borne disease models with Lagrangian approach. J. Math. Biol. 88, 22 (2024). https://doi.org/10.1007/s00285-023-02044-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00285-023-02044-x
Keywords
- Vector-borne disease
- Lagrangian approach
- Basic reproduction number
- Infectious disease network
- Population movement
- Global dynamics