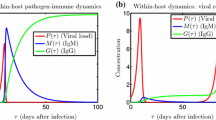
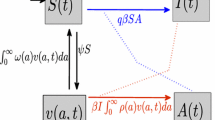
Abstract
This paper proposes and analyzes an immune-structured population model of tilapia subject to Tilapia Lake Virus (TiLV) disease. The model incorporates within-host dynamics, used to describe the interaction between the pathogen, the immune system and the waning of immunity. Individuals infected with a low dose acquire a low immunity level and those infected with a high dose acquire a high level of immunity. Since individuals’ immune status plays an important role in the spread of infectious diseases at the population level, the within-host dynamics are connected to the between-host dynamics in the population. We define an explicit formula for the reproductive number \({\mathcal {R}}_0\) and show that the disease-free equilibrium is locally asymptotically stable when \({\mathcal {R}}_0<1\), while it is unstable when \({\mathcal {R}}_0>1\). Furthermore, we prove that an endemic equilibrium exists. We also study the influence of the initial distribution of host resistance on the spread of the disease, and find that hosts’ initial resistance plays a crucial role in the disease dynamics. This suggests that the genetic selection aiming to improve hosts’ initial resistance to TiLV could help fight the disease. The results also point out the crucial role played by the inoculum size. We find that the higher the initial inoculum size, the faster the dynamics of infection. Moreover, if the initial inoculum size is below a certain threshold, it may not result in an outbreak at the between-host level. Finally, the model shows that there is a strong negative correlation between heterogeneity and the probability of pathogen invasion.






Similar content being viewed by others
References
Barbarossa MV, Röst G (2015) Immuno-epidemiology of a population structured by immune status: a mathematical study of waning immunity and immune system boosting. J Math Biol 71(6–7):1737–1770
Barría A, Trinh TQ, Mahmuddin M, Benzie JA, Chadag VM, Houston RD (2020) Genetic parameters for resistance to Tilapia Lake Virus (TiLV) in Nile Tilapia (Oreochromis niloticus). Aquaculture 522:735126
Behera B, Pradhan P, Swaminathan T, Sood N, Paria P, Das A, Verma D, Kumar R, Yadav M, Dev A, Parida P, Das B, Lal K, Jena J (2018) Emergence of Tilapia Lake Virus associated with mortalities of farmed Nile Tilapia Oreochromis niloticus (Linnaeus 1758) in India. Aquaculture 484:168–174
Best K, Barouch DH, Guedj J, Ribeiro RM, Perelson AS (2021) Zika virus dynamics: effects of inoculum dose, the innate immune response and viral interference. PLoS Comput Biol 17(1):e1008564
Brunner JL, Olson AD, Rice JG, Meiners SE, Le Sage MJ, Cundiff JA, Goldberg CS, Pessier AP (2019) Ranavirus infection dynamics and shedding in American bullfrogs: consequences for spread and detection in trade. Dis Aquat Org 135(2):135–150
Colijn C, Cohen T, Murray M (2007) Emergent heterogeneity in declining tuberculosis epidemics. J Theor Biol 247(4):765–774
Dong H, Siriroob S, Meemetta W, Santimanawong W, Gangnonngiw W, Pirarat N, Khunrae P, Rattanarojpong T, Vanichviriyakit R, Senapin S (2017) Emergence of Tilapia Lake Virus in Thailand and an alternative semi-nested rt-pcr for detection. Aquaculture 476:111–118
Dong H, Siriroob S, Meemetta W, Santimanawong W, Gangnonngiw W, Pirarat N, Khunrae P, Rattanarojpong T, Vanichviriyakit R, Senapin S (2017) Emergence of Tilapia Lake Virus in Thailand and an alternative semi-nested rt-pcr for detection. Aquaculture 476:111–118
Fathi M, Dickson C, Dickson M, Leschen W, Baily J, Muir F, Ulrich K, Weidmann M (2017) Identification of Tilapia Lake Virus in Egypt in Nile Tilapia affected by ‘summer mortality’ syndrome. Aquaculture 473:430–432
Firdaus-Nawi M, Zamri-Saad M (2016) Major components of fish immunity: a review. Pertanika J Trop Agric Sci 39(4):1
Gandolfi A, Pugliese A, Sinisgalli C (2015) Epidemic dynamics and host immune response: a nested approach. J Math Biol 70(3):399–435
Ganusov VV, Bergstrom CT, Antia R (2002) Within-host population dynamics and the evolution of microparasites in a heterogeneous host population. Evolution 56(2):213–223
Gilchrist MA, Sasaki A (2002) Modeling host-parasite coevolution: a nested approach based on mechanistic models. J Theor Biol 218(3):289–308
Gomes MGM (2019) On the mathematics of populations. bioRxiv 612366
Gulbudak H (2020) An immuno-epidemiological vector-host model with within-vector viral kinetics. J Biol Syst 28(02):233–275
Gulbudak H, Browne CJ (2020) Infection severity across scales in multi-strain immuno-epidemiological dengue model structured by host antibody level. J Math Biol 80:1803–1843
Gulbudak H, Cannataro VL, Tuncer N, Martcheva M (2017) Vector-borne pathogen and host evolution in a structured immuno-epidemiological system. Bull Math Biol 79(2):325–355
Hawley DM, Altizer SM (2011) Disease ecology meets ecological immunology: understanding the links between organismal immunity and infection dynamics in natural populations. Funct Ecol 25(1):48–60
Jansen M, Dong H, Mohan C (2019) Tilapia lake virus: a threat to the global tilapia industry? Rev Aquac 11(3):725–739
Kembou-Ringert JE, Steinhagen D, Readman J, Daly JM, Adamek M (2023) Tilapia lake virus vaccine development: a review on the recent advances. Vaccines 11(2):251
Kenne C, Dorville R, Mophou G, Zongo P (2021) An age-structured model for tilapia lake virus transmission in freshwater with vertical and horizontal transmission. Bull Math Biol 83(90):1–35
Kenne C, Mophou G, Dorville R, Zongo P (2022) A model for brucellosis disease incorporating age of infection and waning immunity. Mathematics 10(4):670
Kenne C, Zongo P, Dorville R (2022) A mathematical model for tilapia lake virus transmission with waning immunity. J Biol Dyn 16(1):98–116
Khan R (2012) Host-parasite interactions in some fish species. J Parasitol Res
Li B, Zheng S, Wang Y, Wang Q, Li Y, Yin J, Ren Y, Shi C, Zhao Z, Jiang Z et al (2022) Susceptibilities of ten fish cell lines to infection with Tilapia Lake Virus. Microbial Pathogenes 105510
Li X-Z, Yang J, Martcheva M (2020) Age structured epidemic modeling, vol 52. Springer, Berlin
Magal P, Noussair A, Pasquier J, Zongo P, Le Foll F (2017) A model for transfer of p-glycoproteins in mcf-7 breast cancer cell line with multiple transfer rules. Bull Math Biol 79(9):2049–2067
Magnadóttir B (2006) Innate immunity of fish (overview). Fish Shellfish Immunol 20(2):137–151
Mai TT, Kayansamruaj P, Soontara C, Kerddee P, Nguyen D-H, Senapin S, Costa JZ, Del-Pozo J, Thompson KD, Rodkhum C et al (2022) Immunization of Nile Tilapia (Oreochromis niloticus) Broodstock with Tilapia Lake Virus (TiLV) inactivated vaccines elicits protective antibody and passive maternal antibody transfer. Vaccines 10(2):167
Metcalf CJE, Farrar J, Cutts FT, Basta NE, Graham AL, Lessler J, Ferguson NM, Burke DS, Grenfell BT (2016) Use of serological surveys to generate key insights into the changing global landscape of infectious disease. The Lancet 388(10045):728–730
Mideo N, Alizon S, Day T (2008) Linking within-and between-host dynamics in the evolutionary epidemiology of infectious diseases. Trends Ecol Evol 23(9):511–517
Mihaljevic JR, Greer AL, Brunner JL (2019) Evaluating the within-host dynamics of ranavirus infection with mechanistic disease models and experimental data. Viruses 11(5):396
Mugimba KK, Lamkhannat M, Dubey S, Mutoloki S, Munangandu HM, Evensen O (2020) Tilapia Lake Virus downplays innate immune responses during early stage of infection in Nile Tilapia (Oreochromis niloticus). Sci Rep 10(1):1–12
Neri FM, Bates A, Füchtbauer WS, Pérez-Reche FJ, Taraskin SN, Otten W, Bailey DJ, Gilligan CA (2011) The effect of heterogeneity on invasion in spatial epidemics: from theory to experimental evidence in a model system. PLoS Comput Biol 7(9):e1002174
Sahoo S, Banu H, Prakash A, Tripathi G (2021) Immune system of fish: an evolutionary perspective. In: Antimicrobial immune response. IntechOpen
Sarder MRI, Thompson KD, Penman DJ, McAndrew BJ (2001) Immune responses of Nile Tilapia (Oreochromis niloticus L.) clones: I. Non-specific responses. Dev Comp Immunol 25(1):37–46
Tattiyapong P, Dechavichitlead W, Waltzek TB, Surachetpong W (2020) Tilapia develop protective immunity including a humoral response following exposure to tilapia lake virus. Fish Shellfish Immunol 106:666–674
Tuncer N, Gulbudak H, Cannataro VL, Martcheva M (2016) Structural and practical identifiability issues of immuno-epidemiological vector-host models with application to rift valley fever. Bull Math Biol 78(9):1796–1827
Uribe C, Folch H, Enríquez R, Moran G et al (2011) Innate and adaptive immunity in teleost fish: a review. Vet Med 56(10):486–503
Waiyamitra P, Piewbang C, Techangamsuwan S, Liew WC, Surachetpong W (2021) Infection of Tilapia Tilapinevirus in Mozambique Tilapia (Oreochromis mossambicus), a globally vulnerable fish species. Viruses 13(6):1104
Yang Y-F, Lu T-H, Lin H-C, Chen C-Y, Liao C (2018) Assessing the population transmission dynamics of tilapia lake virus in farmed tilapia. J Fish Dis 419:1439–1448
Acknowledgements
We would like to thank the editors and the anonymous referees for their helpful and very constructive comments, which greatly improved the presentation of this paper.
Funding
This work was funded by a grant from the African Institute for Mathematical Sciences, www.nexteinstein.org, with financial support from the Government of Canada, provided through Global Affairs Canada, www.international.gc.ca, and the International Development Research Centre, www.idrc.ca.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kenne, C., Mophou, G. & Zongo, P. A nested model with boosting and waning of immunity from Tilapia Lake Virus infection with distributed resistance to pathogens carrier-state. J. Math. Biol. 86, 67 (2023). https://doi.org/10.1007/s00285-023-01906-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00285-023-01906-8