Abstract
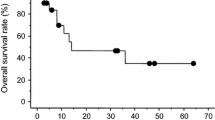
Purpose: Carboplatin has demonstrated significantly poorer response rates in non-seminomatous germ cell tumours. A phase II study of higher than standard doses of carboplatin was conducted because of suspicion that the poorer response might have been due to suboptimal dosing. Patients and methods : A group of 19 patients with advanced germ cell tumours (International Germ Cell Cancer Collaborative Group intermediate and poor prognosis) were treated with carboplatin at an AUC of 8 mg/ml·min (using Calvert's formula) on day 1, etoposide 120 mg/m2 days 1–3 and bleomycin 60,000 U over 2 days (EBCa). Treatment was repeated every 3 weeks and a maximum of four courses was given. Results: Of the 19 patients, 7 (37%) achieved complete remission, of whom 6 (32%) remained long-term progression-free. Post-chemotherapy surgery and further chemotherapy salvaged an additional 26%, leading to an overall disease-free survival rate of 58%. No relationship between outcome and degree of myelosuppression could be established. Conclusion: Dose-escalated carboplatin in combination, although feasible, did not improve the results and led to poorer results than those expected with cisplatin-based therapy. There is no evidence that the patients relapsing following this were easier to salvage. Further investigation of this regimen cannot be recommended.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Shamash, J., McLaren, B., LeVay, J. et al. Carboplatin AUC8 in combination with etoposide and bleomycin in the treatment of intermediate and poor-risk metastatic germ cell tumours: a phase II study. Cancer Chemother Pharmacol 47, 370–372 (2001). https://doi.org/10.1007/s002800000217
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s002800000217