Abstract
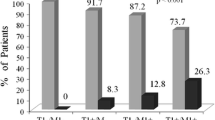
Glutathione S-transferases (GSTs) contribute to the metabolism of different xenobiotics and anticancer drugs and confer protection against oxidative stress thus may influence the treatment outcome of acute myeloid leukemia (AML). Studies regarding the association between GSTT1 and GSTM1 polymorphisms and treatment outcome in AML patients showed an inconsistent result. A systematic review and meta-analysis were performed to further explore this association. PubMed, Hartford User Group Exchange (HUGE), and China National Knowledge Infrastructure (CNKI) databases were searched for all related publications. Statistical analyses were analyzed by using RevMan 5.0 and Stata 9.0 softwares. A total of 1,837 patients in 11 studies were included. GSTT1 null genotype was found to be significantly associated with a reduced response after first course of induction chemotherapy (odds ratio (OR) = 0.894, 95 % confidence interval (CI) = 0.818–0.977, P = 0.013), progression-free survival (PFS; hazard ratio (HR) = 0.698, 95 % CI = 0.520–0.937, P = 0.017), and overall survival (OS; HR = 0.756, 95 % CI = 0.618–0.925, P = 0.007) in Asian population. GSTM1/GSTT1 double-null genotype was also identified to be significantly associated with response after the first course of induction chemotherapy (OR = 0.40, 95 % CI = 0.24–0.67, P = 0.0003). Our study suggested that GSTT1 null genotype and GSTT1/GSTM1 double-null genotype were associated with a worse treatment outcome for AML patients, especially in Asian population.



Similar content being viewed by others
Reference
Frohling S, Scholl C, Gilliland DG, Levine RL (2005) Genetics of myeloid malignancies: pathogenetic and clinical implications. J Clin Oncol 23(26):6285–6295. doi:10.1200/JCO.2005.05.010
Rubnitz JE, Gibson B, Smith FO (2010) Acute myeloid leukemia. Pediatr Clin North Am 24(1):35–63. doi:10.1016/j.hoc.2009.11.008
Jabbour EJ, Estey E, Kantarjian HM (2006) Adult acute myeloid leukemia. Mayo Clin Proc 81(2):247–260. doi:10.4065/81.2.247
Robak T, Wierzbowska A (2009) Current and emerging therapies for acute myeloid leukemia. Clin Ther 31(Pt 2):2349–2370. doi:10.1016/j.clinthera.2009.11.017
Byrd JC, Mrozek K, Dodge RK, Carroll AJ, Edwards CG, Arthur DC, Pettenati MJ, Patil SR, Rao KW, Watson MS, Koduru PR, Moore JO, Stone RM, Mayer RJ, Feldman EJ, Davey FR, Schiffer CA, Larson RA, Bloomfield CD (2002) Pretreatment cytogenetic abnormalities are predictive of induction success, cumulative incidence of relapse, and overall survival in adult patients with de novo acute myeloid leukemia: results from Cancer and Leukemia Group B (CALGB 8461). Blood 100(13):4325–4336. doi:10.1182/blood-2002-03-0772
Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH, Wheatley K, Harrison CJ, Burnett AK (2010) Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5,876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood 116(3):354–365. doi:10.1182/blood-2009-11-254441
Emadi A, Karp JE (2012) The clinically relevant pharmacogenomic changes in acute myelogenous leukemia. Pharmacogenomics 13(11):1257–1269. doi:10.2217/pgs.12.102
Szulawska A, Czyz M (2006) Molecular mechanisms of anthracyclines action. Postepy Hig Med Dosw 60:78–100
Minotti G, Menna P, Salvatorelli E, Cairo G, Gianni L (2004) Anthracyclines: molecular advances and pharmacologic developments in antitumor activity and cardiotoxicity. Pharmacol Rev 56(2):185–229. doi:10.1124/pr.56.2.6
Hayes JD, Flanagan JU, Jowsey IR (2005) Glutathione transferases. Annu Rev Pharmacol Toxicol 45:51–88. doi:10.1146/annurev.pharmtox.45.120403.095857
van Bladeren PJ (2000) Glutathione conjugation as a bioactivation reaction. Chem Biol Interact 129(1–2):61–76
Strange RC, Jones PW, Fryer AA (2000) Glutathione S-transferase: genetics and role in toxicology. Toxicol Lett 112–113:357–363
Rebbeck TR (1997) Molecular epidemiology of the human glutathione S-transferase genotypes GSTM1 and GSTT1 in cancer susceptibility. Cancer Epidemiol Biomarkers Prev 6(9):733–743
Mossallam GI, Abdel Hamid TM, Samra MA (2006) Glutathione S-transferase GSTM1 and GSTT1 polymorphisms in adult acute myeloid leukemia; its impact on toxicity and response to chemotherapy. J Egypt Natl Canc Inst 18(3):264–273
Hayes JD, Strange RC (2000) Glutathione S-transferase polymorphisms and their biological consequences. Pharmacology 61(3):154–166
Naoe T, Tagawa Y, Kiyoi H, Kodera Y, Miyawaki S, Asou N, Kuriyama K, Kusumoto S, Shimazaki C, Saito K, Akiyama H, Motoji T, Nishimura M, Shinagawa K, Ueda R, Saito H, Ohno R (2002) Prognostic significance of the null genotype of glutathione S-transferase-T1 in patients with acute myeloid leukemia: increased early death after chemotherapy. Leukemia 16(2):203–208. doi:10.1038/sj.leu.2402361
Xiao Z, Yang L, Xu Z, Zhang Y, Liu L, Nie L, Li L, Wang J, Hao Y (2008) Glutathione S-transferases (GSTT1 and GSTM1) genes polymorphisms and the treatment response and prognosis in Chinese patients with de novo acute myeloid leukemia. Leuk Res 32(8):1288–1291. doi:10.1016/j.leukres.2007.10.010
Muller P, Asher N, Heled M, Cohen SB, Risch A, Rund D (2008) Polymorphisms in transporter and phase II metabolism genes as potential modifiers of the predisposition to and treatment outcome of de novo acute myeloid leukemia in Israeli ethnic groups. Leuk Res 32(6):919–929. doi:10.1016/j.leukres.2007.10.011
Voso MT, D’Alo F, Putzulu R, Mele L, Scardocci A, Chiusolo P, Latagliata R, Lo-Coco F, Rutella S, Pagano L, Hohaus S, Leone G (2002) Negative prognostic value of glutathione S-transferase (GSTM1 and GSTT1) deletions in adult acute myeloid leukemia. Blood 100(8):2703–2707. doi:10.1182/blood.V100.8.2703
Zhang Y, Yang L, Li R, Zhang L, Zhang MR, Xiao ZJ (2006) The effects of glutathione S-transferase (GSTT1 and GSTM1) genes polymorphisms on treatment efficacy and prognosis of acute myeloid leukemia. Zhonghua Nei Ke Za Zhi [Chin J Intern Med] 45(3):213–216
Barragan E, Collado M, Cervera J, Martin G, Bolufer P, Roman J, Sanz MA (2007) The GST deletions and NQO1*2 polymorphism confers interindividual variability of response to treatment in patients with acute myeloid leukemia. Leuk Res 31(7):947–953. doi:10.1016/j.leukres.2006.10.002
Lee HS, Lee JH, Hur EH, Lee MJ, Lee JH, Kim DY, Kim SH, Seol M, Kang SI, Ryu SG, Kang YA, Lee YS, Kang MJ, Seo EJ, Kim YS, Chi HS, Park CJ, Jang S, Yun SC, Lee KH (2009) Clinical significance of GSTM1 and GSTT1 polymorphisms in younger patients with acute myeloid leukemia of intermediate-risk cytogenetics. Leuk Res 33(3):426–433. doi:10.1016/j.leukres.2008.07.021
Voso MT, Hohaus S, Guidi F, Fabiani E, D’Alo F, Groner S, Spath D, Doehner K, Leone G, Doehner H, Schlenk RF (2008) Prognostic role of glutathione S-transferase polymorphisms in acute myeloid leukemia. Leukemia 22(9):1685–1691. doi:10.1038/leu.2008.169
Weiss JR, Kopecky KJ, Godwin J, Anderson J, Willman CL, Moysich KB, Slovak ML, Hoque A, Ambrosone CB (2006) Glutathione S-transferase (GSTM1, GSTT1 and GSTA1) polymorphisms and outcomes after treatment for acute myeloid leukemia: pharmacogenetics in Southwest Oncology Group (SWOG) clinical trials. Leukemia 20(12):2169–2171. doi:10.1038/sj.leu.2404421
Autrup JL, Hokland P, Pedersen L, Autrup H (2002) Effect of glutathione S-transferases on the survival of patients with acute myeloid leukaemia. Eur J Pharmacol 438(1–2):15–18
Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR (2007) Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 8
Liang L, Lin Y, Yue Z, Zefeng X, Minhua Y, Jianxiang W (2008) Zhijian X (2008) Polymorphisms of RAD51G135C and XRCC3C241T genes and correlations thereof with prognosis and clinical outcomes of acute myeloid leukemia. Natl Med J China 88:378–382
Eyada TK, El Ghonemy EG, El Ghoroury EA, El Bassyouni SO, El Masry MK (2007) Study of genetic polymorphism of xenobiotic enzymes in acute leukemia. Blood Coagul Fibrinolysis 18(5):489–495
Donnelly JG (2004) Pharmacogenetics in cancer chemotherapy: balancing toxicity and response. Ther Drug Monit 26(2):231–235
Wei HB, Lu XS, Shang LH, Xu G, Hu J, Che DH, Liu F, Wu Y, Zhang GM, Yu Y (2011) Polymorphisms of ERCC1 C118T/C8092A and MDR1 C3435T predict outcome of platinum-based chemotherapies in advanced non-small cell lung cancer: a meta-analysis. Arch Med Res 42(5):412–420. doi:10.1016/j.arcmed.2011.07.008
Hayes JD, McLellan LI (1999) Glutathione and glutathione-dependent enzymes represent a co-ordinately regulated defence against oxidative stress. Free Radic Res 31(4):273–300
Strange RC, Spiteri MA, Ramachandran S, Fryer AA (2001) Glutathione-S-transferase family of enzymes. Mutat Res 482(1–2):21–26
Liu K, Zhang L, Lin X, Chen L, Shi H, Magaye R, Zou B, Zhao J (2013) Association of GST genetic polymorphisms with the susceptibility to hepatocellular carcinoma (HCC) in Chinese population evaluated by an updated systematic meta-analysis. PLoS One 8(2):e57043. doi:10.1371/journal.pone.0057043
Lai R, Crevier L, Thabane L (2005) Genetic polymorphisms of glutathione S-transferases and the risk of adult brain tumors: a meta-analysis. Cancer Epidemiol Biomarkers Prev 14(7):1784–1790. doi:10.1158/1055-9965.EPI-05-0105
Liu Y, Xu LZ (2012) Meta-analysis of association between GSTM1 gene polymorphism and cervical cancer. Asian Pac J Trop Med 5(6):480–484. doi:10.1016/S1995-7645(12)60083-2
Leme CV, Raposo LS, Ruiz MT, Biselli JM, Galbiatti AL, Maniglia JV, Pavarino-Bertelli EC, Goloni-Bertollo EM (2010) GSTM1 and GSTT1 genes analysis in head and neck cancer patients. Rev Assoc Med Bras 56(3):299–303
Das P, Shaik AP, Bammidi VK (2009) Meta-analysis study of glutathione-S-transferases (GSTM1, GSTP1, and GSTT1) gene polymorphisms and risk of acute myeloid leukemia. Leuk Lymphoma 50(8):1345–1351
Khedhaier A, Remadi S, Corbex M, Ahmed SB, Bouaouina N, Mestiri S, Azaiez R, Helal AN, Chouchane L (2003) Glutathione S-transferases (GSTT1 and GSTM1) gene deletions in Tunisians: susceptibility and prognostic implications in breast carcinoma. Br J Cancer 89(8):1502–1507. doi:10.1038/sj.bjc.6601292
Hohaus S, Massini G, D’Alo F, Guidi F, Putzulu R, Scardocci A, Rabi A, Di Febo AL, Voso MT, Leone G (2003) Association between glutathione S-transferase genotypes and Hodgkin’s lymphoma risk and prognosis. Clin Cancer Res 9(9):3435–3440
Ha YS, Yan C, Lym MS, Jeong P, Kim WT, Kim YJ, Yun SJ, Lee SC, Moon SK, Choi YH, Kim WJ (2010) GSTT1 as a prognosticator for recurrence and progression in patients with non-muscle-invasive bladder cancer. Dis Markers 29(2):81–87. doi:10.3233/dma-2010-0729
Zareifar S, Monabati A, Saeed A, Fakhraee F, Cohan N (2013) The association of glutathione S-transferase gene mutations (including GSTT1 and GSTM1) with the prognostic factors and relapse in acute lymphoblastic leukemia. Pediatr Hematol Oncol 30(6):568–573. doi:10.3109/08880018.2013.773474
Howells RE, Redman CW, Dhar KK, Sarhanis P, Musgrove C, Jones PW, Alldersea J, Fryer AA, Hoban PR, Strange RC (1998) Association of glutathione S-transferase GSTM1 and GSTT1 null genotypes with clinical outcome in epithelial ovarian cancer. Clin Cancer Res 4(10):2439–2445
Hengstler JG, Arand M, Herrero ME, Oesch F (1998) Polymorphisms of N-acetyltransferases, glutathione S-transferases, microsomal epoxide hydrolase and sulfotransferases: influence on cancer susceptibility. Recent Results Cancer Res Fortschr Krebsforschung Prog Rech Cancer 154:47–85
Kelsey KT, Wiencke JK, Ward J, Bechtold W, Fajen J (1995) Sister-chromatid exchanges, glutathione S-transferase theta deletion and cytogenetic sensitivity to diepoxybutane in lymphocytes from butadiene monomer production workers. Mutat Res 335(3):267–273
Norppa H, Hirvonen A, Jarventaus H, Uuskula M, Tasa G, Ojajarvi A, Sorsa M (1995) Role of GSTT1 and GSTM1 genotypes in determining individual sensitivity to sister chromatid exchange induction by diepoxybutane in cultured human lymphocytes. Carcinogenesis 16(6):1261–1264
Smyth MJ (1991) Glutathione modulates activation-dependent proliferation of human peripheral blood lymphocyte populations without regulating their activated function. J Immunol (Baltimore, M: 1950) 146(6):1921–1927
Macho A, Hirsch T, Marzo I, Marchetti P, Dallaporta B, Susin SA, Zamzami N, Kroemer G (1997) Glutathione depletion is an early and calcium elevation is a late event of thymocyte apoptosis. J Immunol (Baltimore, Md: 1950) 158(10):4612–4619
Shaikh RS, Amir M, Masood AI, Sohail A, Athar HU, Siraj S, Ali M, Anjam MS (2010) Frequency distribution of GSTM1 and GSTT1 null allele in Pakistani population and risk of disease incidence. Environ Toxicol Pharmacol 30(1):76–79. doi:10.1016/j.etap.2010.04.002
Moyer AM, Salavaggione OE, Hebbring SJ, Moon I, Hildebrandt MA, Eckloff BW, Schaid DJ, Wieben ED, Weinshilboum RM (2007) Glutathione S-transferase T1 and M1: gene sequence variation and functional genomics. Clin Cancer Res 13(23):7207–7216. doi:10.1158/1078-0432.CCR-07-0635
Agundez JA, Ladero JM (2008) Glutathione S-transferase GSTT1 and GSTM1 allozymes: beyond null alleles. Pharmacogenomics 9(3):359–363. doi:10.2217/14622416.9.3.359
Acknowledgments
The authors thank Dr. Prof. Deborah Rund (Department of Hematology, Hebrew University—Hadassah Medical Center) and Dr.ssa Maria Teresa Voso (Istituto di Ematologia Università Cattolica S. Cuore) and others for providing us with additional data or information from their studies. The study was supported by grants from the Guangxi Science Fund for Distinguished Young Scholars (2012GXNSFFA060009), Guangxi Natural Science Foundation (2010GXNSFA013181, 2010GXNSFB013064), and National Natural Science Foundation of China (81160072, 81060234). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(TIFF 149 kb)
Rights and permissions
About this article
Cite this article
Xiao, Q., Deng, D., Li, H. et al. GSTT1 and GSTM1 polymorphisms predict treatment outcome for acute myeloid leukemia: a systematic review and meta-analysis. Ann Hematol 93, 1381–1390 (2014). https://doi.org/10.1007/s00277-014-2050-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-014-2050-z