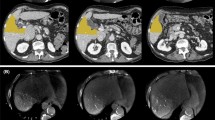
Abstract
Purpose
To compare the use of cone-beam computed tomography versus contrast-enhanced computed tomography (CT) and magnetic resonance imaging (MRI) in the calculation of liver volume and planned dose for yttrium-90 radioembolization.
Materials and Methods
The study retrospectively assessed 47 consecutive patients who underwent resin Y-90 radioembolization consecutively over a 2-year period at a single center. Volume calculation software was used to determine perfused lobar liver volumes from cone-beam CT (CBCT) images obtained during mapping angiography. CBCT-derived volumes were compared with perfused lobar volume derived from contrast-enhanced CT and MRI. Nominal activities as determined by the SIR-Spheres Microspheres Activity Calculator were similarly calculated and compared using both CBCT and conventionally acquired volumes.
Results
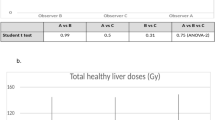
A total of 82 hepatic lobes were assessed in 47 patients. The mean percentage difference between combined CT-MRI- and CBCT-derived calculated lobar volumes was 25.3% (p = 0.994). The mean percentage difference in calculated dose between the two methods was 21.8 ± 24.6% (p = 0.42). Combined left and right lobar CT-derived dose difference was less than 10% in 22 lobes, between 10 and 25% in 20 lobes, between 25 and 50% in 13 lobes and greater than 50% in 5 lobes. Combined left and right lobar MRI-derived dose difference was less than 10% in 11 lobes, between 10 and 25% in 7 lobes, between 25 and 50% in 2 lobes and greater than 50% in 1 lobe.
Conclusions
Although volume measurements derived from CT/MRI did not differ significantly from those derived from CBCT, variability between the two methods led to large and unexpected differences in calculated dose.






Similar content being viewed by others
References
SIR-Spheres microspheres activity calculator North Sydney, Australia: SIRTeX Medical Ltd. [Internet]. https://apps01.sirtex.com/smac/. Accessed 1 March 2018.
Sangro B, Gil-Alzugaray B, Rodriguez J, Sola I, Martinez-Cuesta A, Viudez A, et al. Liver disease induced by radioembolization of liver tumors: description and possible risk factors. Cancer. 2008;112:1538–46.
Braat MNGJA, van Erpecum KJ, Zonnenberg BA, van den Bosch MAJ, Lam MGEH. Radioembolization-induced liver disease. Eur J Gastroenterol Hepatol. 2017;29:144–52.
Gil-Alzugaray B, Chopitea A, Iñarrairaegui M, Bilbao JI, Rodriguez-Fraile M, Rodriguez J, et al. Prognostic factors and prevention of radioembolization-induced liver disease. Hepatology. 2013;57:1078–87.
Kao YH, Hock Tan AE, Burgmans MC, Irani FG, Khoo LS, Gong Lo RH, et al. Image-guided personalized predictive dosimetry by artery-specific SPECT/CT partition modeling for safe and effective 90Y radioembolization. J Nucl Med. 2012;53:559–66.
Orth RC, Wallace MJ, Kuo MD. C-Arm cone-beam CT: general principles and technical considerations for use in interventional radiology. J Vasc Interv Radiol. 2008;19:814–20.
Louie JD, Kothary N, Kuo WT, Hwang GL, Hofmann LV, Goris ML, et al. Incorporating cone-beam CT into the treatment planning for yttrium-90 radioembolization. J Vasc Interv Radiol SIR. 2009;20:606–13.
van den Hoven AF, Prince JF, de Keizer B, Vonken EJPA, Bruijnen RCG, Verkooijen HM, et al. Use of C-Arm cone beam CT during hepatic radioembolization: protocol optimization for extrahepatic shunting and parenchymal enhancement. Cardiovasc Interv Radiol. 2016;39:64–73.
Kennedy A, Nag S, Salem R, Murthy R, McEwan AJ, Nutting C, et al. Recommendations for radioembolization of hepatic malignancies using yttrium-90 microsphere brachytherapy: a consensus panel report from the radioembolization brachytherapy oncology consortium. Int J Radiat Oncol Biol Phys. 2007;68:13–23.
Michels NA. Newer anatomy of the liver and its variant blood supply and collateral circulation. Am J Surg. 1966;112:337–47.
van den Hoven AF, van Leeuwen MS, Lam MGEH, van den Bosch MAAJ. Hepatic arterial configuration in relation to the segmental anatomy of the liver; observations on MDCT and DSA relevant to radioembolization treatment. Cardiovasc Interv Radiol. 2014;38:100–11.
Okuda K, Moriyama M, Yasumoto M, Jinnouchi S, Shimokawa Y. Roentgenologic demonstration of spontaneous reversal of portal blood flow in cirrhosis and primary carcinoma of the liver. Am J Roentgenol. 1973;119:419–28.
Yu J-S, Rofsky N. Magnetic resonance imaging of arterioportal shunts in the liver. Top Magn Reson Imaging. 2002;13:165–76.
Choi BI, Lee KH, Han JK, Lee JM. Hepatic arterioportal shunts: dynamic CT and MR features. Korean J Radiol. 2002;3:1–15.
Van Den Hoven AF, Prince JF, De Keizer B. Use of C-Arm cone beam CT during hepatic radioembolization: protocol optimization for extrahepatic shunting and parenchymal enhancement. Cardiovasc Interv Radiol. 2016;39:64–73.
Miyayama S, Yamashiro M, Hashimoto M, Hashimoto N, Ikuno M, Okumura K, et al. Identification of small hepatocellular carcinoma and tumor-feeding branches with cone-beam ct guidance technology during transcatheter arterial chemoembolization. J Vasc Interv Radiol. 2013;24:501–8.
Kaufmann S, Horger T, Oelker A, Beck S, Schulze M, Nikolaou K, et al. Volume perfusion computed tomography (VPCT)-based evaluation of response to TACE using two different sized drug eluting beads in patients with nonresectable hepatocellular carcinoma: impact on tumor and liver parenchymal vascularisation. Eur J Radiol. 2015;84:2548–54.
Pung L, Ahmad M, Mueller K, Rosenberg J, Stave C, Hwang GL, et al. The role of cone-beam ct in transcatheter arterial chemoembolization for hepatocellular carcinoma: a systematic review and meta-analysis. J Vasc Interv Radiol. 2016;28:334–41.
Kothary N, Abdelmaksoud MHK, Tognolini A, Fahrig R, Rosenberg J, Hovsepian DM, et al. Imaging guidance with C-Arm CT: prospective evaluation of its impact on patient radiation exposure during transhepatic arterial chemoembolization. J Vasc Interv Radiol. 2011;22:1535–43.
Garin E, Lenoir L, Rolland Y, Edeline J, Mesbah H, Laffont S, et al. Dosimetry based on 99mTc-macroaggregated albumin SPECT/CT accurately predicts tumor response and survival in hepatocellular carcinoma patients treated with 90Y-loaded glass microspheres: preliminary results. J Nucl Med. 2012;53:255–63.
Kallini JR, Gabr A, Salem R, Lewandowski RJ. Transarterial radioembolization with yttrium-90 for the treatment of hepatocellular carcinoma. Adv Ther. 2016;33:699–714.
Srinivas SM, Natarajan N, Kuroiwa J, Gallagher S, Nasr E, Shah SN, et al. Determination of radiation absorbed dose to primary liver tumors and normal liver tissue using post-radioembolization 90Y PET. Front Oncol. 2014;4:255.
Fowler KJ, Maughan NM, Laforest R, Saad NE, Sharma A, Olsen J, et al. PET/MRI of Hepatic 90Y microsphere deposition determines individual tumor response. Cardiovasc Interv Radiol. 2016;39:855–64.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All the authors declared that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ertreo, M., Choi, H., Field, D. et al. Comparison of Cone-Beam Tomography and Cross-Sectional Imaging for Volumetric and Dosimetric Calculations in Resin Yttrium-90 Radioembolization. Cardiovasc Intervent Radiol 41, 1857–1866 (2018). https://doi.org/10.1007/s00270-018-2030-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-018-2030-0