Abstract
Purpose
To investigate radiopacity, size and size calibration, morphology, and vascular distribution of inherently radiopaque microspheres in vitro and in a pig embolization model.
Materials and Methods
We compared three types of microspheres: DCBead™ (size 100–300 µm) and Embozene™ (250 µm) as clinically established microspheres, and the prototype Visible (250 µm) that contains additional radiopaque material. Size and size calibration of microspheres were examined by laser diffraction. Pulmonary artery embolization was performed in 12 pigs, and radiopacity was examined by in vitro micro-computed tomography (CT), in vivo cone-beam CT, and ex vivo micro-CT after killing. Morphology and vascular distribution of microspheres were microscopically examined.
Results
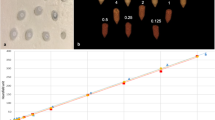
In in vitro and ex vivo micro-CT, radiopacity of Visible was higher than that of Embozene™, whereas DCBead™ showed no radiopacity. In in vivo cone-beam CT, radiopacity was observed with Visible but not with Embozene™ and DCBead™. Laser diffraction revealed that 7.0% (Visible), 6.5% (Embozene™), and 22.5% (DCBead™) of microspheres were smaller than 223.5 μm. Visible and Embozene™ microspheres were very often located in bronchiolus-associated arteries, but rarely in subsegmental and capillary arteries, whereas DCBead™ were very often and often detected in bronchiolus-associated arteries and capillary arteries, respectively (and rarely in subsegmental arteries).
Conclusion
After pulmonary artery embolization, Visible but not Embozene™ or DCBead™ provide in vivo radiopacity in cone-beam CT. In contrast to non-narrow-size-calibrated DCBead™, pulmonary artery embolization with narrow-size-calibrated Visible and Embozene™ result in a predictable arterial distribution without embolization-related hemorrhagic lung infarction.





Similar content being viewed by others
References
Sharma KV, Bascal Z, Kilpatrick H, Ashrafi K, Willis SL, Dreher MR, et al. Long-term biocompatibility, imaging appearance and tissue effects associated with delivery of a novel radiopaque embolization bead for image-guided therapy. Biomaterials. 2016;103:293–304. https://doi.org/10.1016/j.biomaterials.2016.06.064.
Ashrafi K, Tang Y, Britton H, Domenge O, Blino D, Bushby AJ, et al. Characterization of a novel intrinsically radiopaque Drug-eluting Bead for image-guided therapy: DC Bead LUMI. J Control Release. 2017;250:36–47. https://doi.org/10.1016/j.jconrel.2017.02.001.
Duran R, Sharma K, Dreher MR, Ashrafi K, Mirpour S, Lin M, et al. A novel inherently radiopaque bead for transarterial embolization to treat liver cancer—A pre-clinical study. Theranostics. 2016;6(1):28–39. https://doi.org/10.7150/thno.13137.
Golowa YS, Cynamon J, Reinus JF, Kinkhabwala M, Abrams M, Jagust M, et al. Value of noncontrast CT immediately after transarterial chemoembolization of hepatocellular carcinoma with drug-eluting beads. J Vasc Interv Radiol. 2012;23(8):1031–5. https://doi.org/10.1016/j.jvir.2012.04.020.
Loffroy R, Lin M, Yenokyan G, Rao PP, Bhagat N, Noordhoek N, et al. Intraprocedural C-arm dual-phase cone-beam CT: can it be used to predict short-term response to TACE with drug-eluting beads in patients with hepatocellular carcinoma? Radiology. 2013;266(2):636–48. https://doi.org/10.1148/radiol.12112316.
Tacher V, Duran R, Lin M, Sohn JH, Sharma KV, Wang Z, et al. Multimodality imaging of ethiodized oil-loaded radiopaque microspheres during transarterial embolization of rabbits with VX2 liver tumors. Radiology. 2016;279(3):741–53. https://doi.org/10.1148/radiol.2015141624.
Takayasu K, Muramatsu Y, Maeda T, Iwata R, Furukawa H, Muramatsu Y, et al. Targeted transarterial oily chemoembolization for small foci of hepatocellular carcinoma using a unified helical CT and angiography system: analysis of factors affecting local recurrence and survival rates. AJR Am J Roentgenol. 2001;176(3):681–8. https://doi.org/10.2214/ajr.176.3.1760681.
Sommer CM, Stampfl U, Bellemann N, Holzschuh M, Kueller A, Bluemmel J, et al. Multimodal visibility (radiography, computed tomography, and magnetic resonance imaging) of microspheres for transarterial embolization tested in porcine kidneys. Invest Radiol. 2013;48(4):213–22. https://doi.org/10.1097/RLI.0b013e31827f6598.
Stampfl U, Sommer CM, Bellemann N, Holzschuh M, Kueller A, Bluemmel J, et al. Multimodal visibility of a modified polyzene-F-coated spherical embolic agent for liver embolization: feasibility study in a porcine model. J Vasc Interv Radiol. 2012;23(9):1225–31. https://doi.org/10.1016/j.jvir.2012.06.008.
Carnevale FC, Iscaife A, Yoshinaga EM, Moreira AM, Antunes AA, Srougi M. Transurethral resection of the prostate (TURP) versus original and PErFecTED prostate artery embolization (PAE) due to benign prostatic hyperplasia (BPH): Preliminary results of a single center, prospective, urodynamic-controlled analysis. Cardiovasc Intervent Radiol. 2016;39(1):44–52. https://doi.org/10.1007/s00270-015-1202-4.
Uflacker A, Haskal ZJ, Bilhim T, Patrie J, Huber T, Pisco JM. Meta-analysis of prostatic artery embolization for benign prostatic hyperplasia. J Vasc Interv Radiol. 2016;27(11):1686–97. https://doi.org/10.1016/j.jvir.2016.08.004.
Gadaleta CD, Solbiati L, Mattioli V, Rubini G, Fazio V, Goffredo V, et al. Unresectable lung malignancy: combination therapy with segmental pulmonary arterial chemoembolization with drug-eluting microspheres and radiofrequency ablation in 17 patients. Radiology. 2013;267(2):627–37. https://doi.org/10.1148/radiol.12120198.
Sommer CM, Do TD, Schlett CL, Flechsig P, Gockner TL, Kuthning A, et al. In vivo characterization of a new type of biodegradable starch microsphere for transarterial embolization. J Biomater Appl. 2018;32(7):932–44. https://doi.org/10.1177/0885328217746674.
Heffernan BM, Heinson YW, Maughan JB, Chakrabarti A, Sorensen CM. Backscattering measurements of micron-sized spherical particles. Appl Opt. 2016;55(12):3214–8. https://doi.org/10.1364/AO.55.003214.
Aliberti C, Carandina R, Sarti D, Pizzirani E, Ramondo G, Cillo U, et al. Transarterial chemoembolization with DC Bead LUMI radiopaque beads for primary liver cancer treatment: preliminary experience. Future Oncol. 2017;13(25):2243–52. https://doi.org/10.2217/fon-2017-0364.
Stampfl S, Bellemann N, Stampfl U, Sommer CM, Thierjung H, Lopez-Benitez R, et al. Arterial distribution characteristics of Embozene particles and comparison with other spherical embolic agents in the porcine acute embolization model. J Vasc Interv Radiol. 2009;20(12):1597–607. https://doi.org/10.1016/j.jvir.2009.08.018.
Gnutzmann DM, Mechel J, Schmitz A, Kohler K, Krone D, Bellemann N, et al. Evaluation of the plasmatic and parenchymal elution kinetics of two different irinotecan-loaded drug-eluting embolics in a pig model. J Vasc Interv Radiol. 2015;26(5):746–54. https://doi.org/10.1016/j.jvir.2014.12.016.
Pereira PL, Plotkin S, Yu R, Sutter A, Wu Y, Sommer CM, et al. An in vitro evaluation of three types of drug-eluting microspheres loaded with irinotecan. Anticancer Drugs. 2016;27(9):873–8. https://doi.org/10.1097/CAD.0000000000000408.
Derdeyn CP, Graves VB, Salamat MS, Rappe A. Collagen-coated acrylic microspheres for embolotherapy: in vivo and in vitro characteristics. AJNR Am J Neuroradiol. 1997;18(4):647–53.
Dreher MR, Sharma KV, Woods DL, Reddy G, Tang Y, Pritchard WF, et al. Radiopaque drug-eluting beads for transcatheter embolotherapy: experimental study of drug penetration and coverage in swine. J Vasc Interv Radiol. 2012;23(2):257–64. https://doi.org/10.1016/j.jvir.2011.10.019.
Stampfl S, Stampfl U, Rehnitz C, Schnabel P, Satzl S, Christoph P, et al. Experimental evaluation of early and long-term effects of microparticle embolization in two different mini-pig models. Part II: liver. Cardiovasc Intervent Radiol. 2007;30(3):462–8. https://doi.org/10.1007/s00270-005-0350-3.
Funding
CeloNova BioSciences, San Antonio, USA supported technically this trial.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declared that they have conflict of interest.
Rights and permissions
About this article
Cite this article
Sommer, C.M., Harms, A., Do, T.D. et al. Inherently Radiopaque Narrow-Size-Calibrated Microspheres: Proof of Principle in a Pig Embolization Model. Cardiovasc Intervent Radiol 41, 1404–1411 (2018). https://doi.org/10.1007/s00270-018-1986-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-018-1986-0