Abstract
Objective
In this study, we investigate the biological characteristics of ADSCs from the liposuction area in patients with hemifacial atrophy in vitro.
Methods
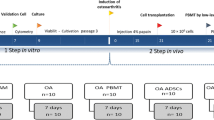
ADSCs were respectively extracted from the donor site of patients with hemifacial atrophy and healthy ones. ADSCs of two groups were respectively tested for proliferation ability, phenotype, multipotency, migration ability, self-repair ability, apoptosis, and autophagy. Exosomes extracted from the supernatant of two groups were detected by NTA particle size, electron microscopy (TEM), and WB for CD63 and TSG10, respectively.
Results
CCK-8 showed a statistically less increase in cell proliferation in PHA-ADSCs after the sixth day. ADSCs in both groups had typical phenotypes and multidirectional abilities. PHA-ADSCs exhibited weaker droplet formation. The cell migration ability in PHA-ADSCs was weaker tested by Transwell assay. The live/dead proportion calculated by ImageJ following calcein-AM/PI double staining revealed live cells in PHA-ADSCs was 46.11% compared with 54.21% in NORM-ADSCs after OGD treatment. A significant down-regulation of ATG7 and ATG12 and a higher percentage of apoptosis were found in PHA-ADSCs. A significant up-regulation of BAX occurred in PHA-ADSCs.ARPC5 expression in the PHA group was extremely distinct down-regulated.CDKN1A and CDKN2A expression in the PHA group was significantly up-regulated.WB analyses confirmed that both groups' ADSCs-Exosomes surface markers CD63 and TSG101 were positively expressed but varied significantly.
Conclusions
PHA-ADSCs exhibited a poorer proliferation ability, higher apoptosis percentage, weaker lipid droplets formation, weaker cell migration, poorer intolerance to OGD, aging earlier, and weaker self-renewal and repairability.PHA-ADSCs-Exosomes showed low expressions of CD63 and TSG101.This study provides strong evidence that the addition of exosomes with specific cytokines can improve the fat survival rate after fat filling in patients with hemifacial atrophy.
No Level Assigned
This journal requires that authors 42 assign a level of evidence to each submission to which 43 Evidence-Based Medicine rankings are applicable. This 44 excludes Review Articles, Book Reviews, and manuscripts 45 that concern Basic Science, Animal Studies, Cadaver 46 Studies, and Experimental Studies. For a full description of 47 these Evidence-Based Medicine ratings, please refer to the 48 Table of Contents or the online Instructions to Authors 49 https://www.springer.com/00266.






Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
References
Schultz KP, Dong E, Truong TA, Maricevich RS (2019) Parry Romberg syndrome. Clin Plast Surg 46(2):231–237. https://doi.org/10.1016/j.cps.2018.11.007
Deshingkar SA, Barpande SR, Bhavthankar JD et al (2012) Progressive hemifacial atrophy (Parry–Romberg syndrome). Contemp Clin Dent. 3(Suppl 1):S78-81. https://doi.org/10.4103/0976-237X.95111.
Tolkachjov SN, Patel NG, Tollefson MM (2015) Progressive hemifacial atrophy: a review. Orphanet J Rare Dis 1(10):39. https://doi.org/10.1186/s13023-015-0250-9.
Rodby KA, Kaptein YE, Roring J et al (2016) Evaluating autologous lipofilling for Parry–Romberg syndrome-associated defects: a systematic literature review and case report. Cleft Palate Craniofac J 53(3):339–350. https://doi.org/10.1597/14-232
Denadai R, Buzzo CL, Raposo-Amaral CA et al (2019) Facial contour symmetry outcomes after site-specific facial fat compartment augmentation with fat grafting in facial deformities. Plast Reconstr Surg 143(2):544–556. https://doi.org/10.1097/PRS.0000000000005220
Zheng H, Yu Z, Deng M et al (2019) Fat extract improves fat graft survival via proangiogenic, anti-apoptotic and pro-proliferative activities. Stem Cell Res Ther 10(1):174. https://doi.org/10.1186/s13287-019-1290-1.
Chen Y, Xue K, Zhang X et al (2018) Exosomes derived from mature chondrocytes facilitate subcutaneous stable ectopic chondrogenesis of cartilage progenitor cells. Stem Cell Res Ther 9(1):318
Liu H, Zhang M, Shi M et al (2021) Adipose-derived mesenchymal stromal cell-derived exosomes promote tendon healing by activating both SMAD1/5/9 and SMAD2/3. Stem Cell Res Ther 12(1):338. https://doi.org/10.1186/s13287-021-02410-w.
Chieregato K, Castegnaro S, Madeo D et al (2011) Epidermal growth factor, basic fibroblast growth factor and platelet-derived growth factor-bb can substitute for fetal bovine serum and compete with human platelet-rich plasma in the ex vivo expansion of mesenchymal stromal cells derived from adipose tissue. Cytotherapy 13(8):933–943. https://doi.org/10.3109/14653249.2011.583232
Omar Zaki SS, Kanesan L, Leong MYD et al (2019) The influence of serum-supplemented culture media in a transwell migration assay. Cell Biol Int 43(10):1201–1204. https://doi.org/10.1002/cbin.11122
Gorzelniak K, Janke J, Engeli S, Sharma AM (2001) Validation of endogenous controls for gene expression studies in human adipocytes and preadipocytes. Horm Metab Res 33(10):625–627. https://doi.org/10.1055/s-2001-17911
Guerrerosantos J, Guerrerosantos F, Orozco J (2007) Classification and treatment of facial tissue atrophy in Parry–Romberg disease. Aesthet Plast Surg 31:424–434
Hu J, Yin L, Tang X et al (2011) Combined skeletal and soft tissue reconstruction for severe Parry–Romberg syndrome. J Craniofac Surg 22:937–941
Raposo-Amaral C, Denadai R, Camargo D et al (2013) Parry–Romberg syndrome: severity of the deformity does not correlate with quality of life. Aesthet Plast Surg 37:792–801
Palmero ML, Uziel Y, Laxer RM, et al (2010) En coup de sabre scleroderma and Parry–Romberg syndrome in adolescents: surgical options and patient-related outcomes. J Rheumatol 37(10):2174–2179. https://doi.org/10.3899/jrheum.100062. Epub 2010 Sep 15. Erratum in. (2010): J Rheumatol 37(11):2444.
Saccomanno F, Bernardi C, Vittorini P (1997) The expanded polytetrafluoroethylene (ePTFE) in the surgical treatment of Parry–Romberg syndrome: case report. Aesthet Plast Surg 21:342–345
Onesti MG, Monarca C, Rizzo MI et al (2009) Minimally invasive combined treatment for Parry–Romberg syndrome. Aesthet Plast Surg 33(3):452–456. https://doi.org/10.1007/s00266-008-9287-7
Ortega VG, Sastoque D (2015) New and successful technique for the management of Parry–Romberg syndrome’s soft tissue atrophy. J Craniofac Surg 26(6):e507–e510. https://doi.org/10.1097/SCS.0000000000002023
Avelar RL, Göelzer JG, Azambuja FG et al (2010) Use of autologous fat graft for correction of facial asymmetry stemming from Parry–Romberg syndrome. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 109(2):e20–e25
Billings E Jr, May JW Jr (1989) Historical review and present status of free fat graft autotransplantation in plastic and reconstructive surgery. Plast Reconstr Surg 83(2):368–381. https://doi.org/10.1097/00006534-198902000-00033
Leaf N, Zarem HA (1972) Correction of contour defects of the face with dermal and dermal-fat grafts. Arch Surg 105:715–719
Chajchir A, Benzaquen I (1986) Liposuction fat grafts in face wrinkles and hemifacial atrophy. Aesthet Plast Surg 10(2):115–117. https://doi.org/10.1007/BF01575279
Moscona R, Ullman Y, Har-Shai Y et al (1989) Free-fat injections for the correction of hemifacial atrophy. Plast Reconstr Surg 84:501–507
Tobin H, Middleton W (1989) Hemifacial atrophy: a case report of fat transplantation. J Otolaryngol 18:125–127
Liu SW, Cohen GF (2010) Idiopathic hemi-facial lipoatrophy treated with autologous fat transfer. J Cosmet Dermatol 9(3):226–229. https://doi.org/10.1111/j.1473-2165.2010.00507.x
Alencar JC, Andrade SH, Pessoa SG et al (2011) Autologous fat transplantation for the treatment of progressive hemifacial atrophy (Parry–Romberg syndrome: case report and review of medical literature). An Bras Dermatol 86((4 Suppl 1)):S85–S88. https://doi.org/10.1590/s0365-05962011000700022 (English, Portuguese)
Roddi R, Riggio E, Gilbert PM et al (1994) Clinical evaluation of techniques used in the surgical treatment of progressive hemifacial atrophy. J Craniomaxillofac Surg 22(1):23–32. https://doi.org/10.1016/s1010-5182(05)80292-6
Roddi R, Riggio E, Gilbert P et al (1994) The value of microfat injection ‘“lipofilling”’ in the treatment of soft tissue deformities of the face in Parry–Romberg syndrome. Eur J Plast Surg 17:79–83
Cortese A, Savastano G, Felicetta L (2000) Free fat transplantation for facial tissue augmentation. J Oral Maxillofac Surg. 58(2):164–9. https://doi.org/10.1016/s0278-2391(00)90331-8 (discussion 169-70)
Rupnick MA, Panigrahy D, Zhang CY et al (2002) Adipose tissue mass can be regulated through the vasculature. Proc Natl Acad Sci USA 99(16):10730–5. https://doi.org/10.1073/pnas.162349799
Eto H, Kato H, Suga H et al (2012) The fate of adipocytes after nonvascularized fat grafting: evidence of early death and replacement of adipocytes. Plast Reconstr Surg 129(5):1081–1092. https://doi.org/10.1097/PRS.0b013e31824a2b19
Yoshimura K, Sato K, Aoi N et al (2008) Cell-assisted lipotransfer for facial lipoatrophy: efficacy of clinical use of adipose-derived stem cells. Dermatol Surg 34(9):1178–1185. https://doi.org/10.1111/j.1524-4725.2008.34256.x
Chang Q, Li J, Dong Z, Liu L, Lu F (2013) Quantitative volumetric analysis of progressive hemifacial atrophy corrected using stromal vascular fraction-supplemented autologous fat grafts. Dermatol Surg 39(10):1465–1473. https://doi.org/10.1111/dsu.12310
Cervelli V, Gentile P (2009) Use of cell fat mixed with platelet gel in progressive hemifacial atrophy. Aesthet Plast Surg 33(1):22–27. https://doi.org/10.1007/s00266-008-9223-x
Tiryaki T, Findikli N, Tiryaki D (2011) Staged stem cell-enriched tissue (SET) injections for soft tissue augmentation in hostile recipient areas: a preliminary report. Aesthet Plast Surg 35(6):965–971. https://doi.org/10.1007/s00266-011-9716-x
Zhu M, Zhou Z, Chen Y et al (2010) Supplementation of fat grafts with adipose-derived regenerative cells improves long-term graft retention. Ann Plast Surg 64(2):222–228. https://doi.org/10.1097/SAP.0b013e31819ae05c
CastroGovea Y, De La Garza-Pineda O, Lara-Arias J et al (2012) Cell-assisted lipotransfer for the treatment of Parry–Romberg syndrome. Arch Plast Surg 39(6):659–62. https://doi.org/10.5999/aps.2012.39.6.659
Doi K, Ogata F, Eto H et al (2015) Differential contributions of graft-derived and host-derived cells in tissue regeneration/remodeling after fat grafting. Plast Reconstr Surg 135(6):1607–1617. https://doi.org/10.1097/PRS.0000000000001292
Shih YC, Lee PY, Cheng H et al (2013) Adipose-derived stem cells exhibit antioxidative and antiapoptotic properties to rescue ischemic acute kidney injury in rats. Plast Reconstr Surg 132(6):940e–951e. https://doi.org/10.1097/PRS.0b013e3182a806ce
Yuan YI, Gao J, Lu F (2015) Effect of exogenous adipose-derived stem cells in the early stages following free fat transplantation. Exp Ther Med 10(3):1052–1058. https://doi.org/10.3892/etm.2015.2605
Li C, Chen K, Jia M et al (2018) AMPK promotes survival and adipogenesis of ischemia-challenged ADSCs in an autophagy-dependent manner. Biochim Biophys Acta Mol Cell Biol Lipids 1863(12):1498–1510. https://doi.org/10.1016/j.bbalip.2018.10.002
Rosen ED, MacDougald OA (2006) Adipocyte differentiation from the inside out. Nat Rev Mol Cell Biol 7(12):885–896. https://doi.org/10.1038/nrm2066
Cristancho AG, Lazar MA (2011) Forming functional fat: a growing understanding of adipocyte differentiation. Nat Rev Mol Cell Biol 12(11):722–734. https://doi.org/10.1038/nrm3198
Isakovic AM, Dulovic M, Markovic I et al (2017) Autophagy suppression sensitizes glioma cells to IMP dehydrogenase inhibition-induced apoptotic death. Exp Cell Res 350(1):32–40. https://doi.org/10.1016/j.yexcr.2016.11.001
Jung CH, Ro SH, Cao J, Otto NM, Kim DH (2010) mTOR regulation of autophagy. FEBS Lett. 584(7):1287–95. https://doi.org/10.1016/j.febslet.2010.01.017
Ariosa AR, Klionsky DJ (2016) Autophagy core machinery: overcoming spatial barriers in neurons. J Mol Med. 94(11):1217–1227. https://doi.org/10.1007/s00109-016-1461-9
Dejean LM, Martinez-Caballero S, Guo L et al (2005) Oligomeric Bax is a component of the putative cytochrome c release channel MAC, mitochondrial apoptosis-induced channel. Mol Biol Cell. 16(5):2424–32. https://doi.org/10.1091/mbc.e04-12-1111
Kinoshita T, Nohata N, Watanabe-Takano H et al (2012) Actin-related protein 2/3 complex subunit 5 (ARPC5) contributes to cell migration and invasion and is directly regulated by tumor-suppressive microRNA-133a in head and neck squamous cell carcinoma. Int J Oncol 40(6):1770–1778. https://doi.org/10.3892/ijo.2012.1390
Hayflick L (1965) The limited in vitro lifetime of human diploid cell strains. Exp Cell Res 37:614–636. https://doi.org/10.1016/0014-4827(65)90211-9
Munk R, Panda AC, Grammatikakis I et al (2017) Senescence-associated MicroRNAs. Int Rev Cell Mol Biol 334:p177-205. https://doi.org/10.1016/bs.ircmb.2017.03.008
Ohtani N, Takahashi A, Mann DJ et al (2012) Cellular senescence: a double-edged sword in the fight against cancer. Exp Dermatol 21(Suppl 1):1–4. https://doi.org/10.1111/j.1600-0625.2012.01493.x
He C, Zheng S, Luo Y, Wang B (2018) Exosome theranostics: biology and translational medicine. Theranostics 8(1):237–255. https://doi.org/10.7150/thno.21945
Funding
The authors received National Multi-disciplinary Collaborative Diagnosis and Treatment capacity Building project for Critical Diseases (No.19038) and CAMS-Innovation Fund for Medical Sciences (No.2020-12M-C&T-A-018) support for the research, authorship, and publication of this article.
Author information
Authors and Affiliations
Contributions
QG, ZY, YH, XY, XY, XJ and ZQ performed the research.QG,XJ and ZQ designed this study. QG and ZY analyzed the data. QG, ZY, XY, YH and XY followed up with the patients and wrote this paper. XJ and ZQ provided advice and critical discussions on the project. All of the authors read and approved the final version of this paper and participated in reviewing and editing the manuscript and approved the final version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest to disclose.
Ethical Approval
Written informed consent was obtained from two patients for their portrait rights in accordance with the institutional ethical committee of Plastic Surgery Hospital (Institute), Chinese Academy of Medical Sciences (CAMS), Peking Union Medical College, and the approval number was 2021-203.And this article did not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, Q., Qi, Z., Jin, X. et al. In Vitro Study on the Biological Characteristics of Adipose-Derived Stem Cells from Liposuction Area in Patients with Progressive Hemifacial Atrophy. Aesth Plast Surg 46, 2526–2538 (2022). https://doi.org/10.1007/s00266-022-02902-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-022-02902-x