Abstract
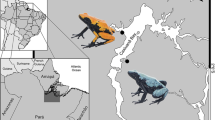
Intraspecific morphological variation may correspond to behavioral variation that helps determine the nature of species interactions. Color variation among populations of variably toxic organisms has been shown to associate with alternative anti-predator behaviors. However, the effects of these alternative behavioral tendencies on the outcomes of interspecific interactions other than predator–prey remain largely unexplored. We investigated how coloration and body size variation in Oophaga pumilio, one of the most phenotypically diverse amphibians known, associated with territorial aggressiveness and how this association influenced the outcome of agonistic male–male interactions with conspecifics and heterospecifics of two sympatric species (Andinobates claudiae and Phyllobates lugubris). Irrespective of body size, resident frogs from more conspicuous, red-colored O. pumilio populations responded to same-morph conspecifics and P. lugubris more quickly and exhibited more aggressive behaviors and more energetically expensive behaviors than resident frogs from green populations under these same treatments. Furthermore, red-colored resident frogs dominated most of the interactions in which they were involved, whereas green residents dominated only a few of the interactions, despite their status as residents. Because conspecific and heterospecific intruders did not behave more aggressively toward red resident frogs, aggressiveness of red residents does not appear to be a response to higher aggression being directed toward them. These results suggest that coloration in O. pumilio is a good indicator of aggressiveness that associates with the outcome of intraspecific and some interspecific behavioral male–male interactions, providing support for a positive association among anti-predator traits, agonistic behavior, and dominance in both intraspecific and interspecific, intraguild interactions.



Similar content being viewed by others
References
Adams DC (2004) Character displacement via aggressive interference in Appalachian salamanders. Ecology 85:2664–2670
Anderson CN, Grether GF (2010) Interspecific aggression and character displacement of competitor recognition in Hetaerina damselflies. Proc R Soc Lond B 277:549–555
Bastiaans E, Morinaga G, Gaytan JGC, Marshall JC, Sinervo B (2013) Male aggression varies with throat color in 2 distinct populations of the mesquite lizard. Behav Ecol 24:968–981
Baugh JR, Forester DC (1994) Prior residence effect in the dart-poison frog, Dendrobates pumilio. Behaviour 131:207–224
Bee MA (2003) A test of the “dear enemy effect” in the strawberry dart-poison frog (Dendrobates pumilio). Behav Ecol Sociobiol 54:601–610
Bernays EA, Singer M (2002) Contrasted foraging tactics in two species of polyphagous caterpillars. Acta Zool Hung 48:117–135
Blomberg SP, Garland T Jr, Ives AR (2003) Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57:717–45
Brown JL, Maan ME, Cummings ME, Summers K (2010) Evidence for selection on coloration in a Panamanian poison frog: a coalescent-based approach. J Biogeogr 37:891–901
Cohen J (1988) Statistical power analyses for the behavioral sciences, 2nd edn. Erlbaum, Hillsdale
Crothers LR, Cummings ME (2013) Warning signal brightness variation: sexual selection may work under the radar of natural selection in populations of a polytypic poison frog. Am Nat 181:E116–E124
Crothers LR, Cummings ME (2015) A multifunctional warning signal behaves as an agonistic status signal in a poison frog. Behav Ecol 26:560–568
Crothers LR, Gering E, Cummings ME (2011) Aposematic signal variation predicts male-male interactions in a polymorphic poison frog. Evolution 65:599–605
Daly JW, Myers CW (1967) Toxicity of Panamanian poison frogs (Dendrobates): some biological and chemical aspects. Science 156:970–973
Dijkstra PD, Groothuis TG (2011) Male-male competition as a force in evolutionary diversification: evidence in haplochromine cichlid fish. Int J Evol Biol 2011:689254
Dijkstra PD, Seehausen O, Groothuis TGG (2005) Direct male-male competition can facilitate invasion of new colour types in Lake Victoria cichlids. Behav Ecol Sociobiol 58:136–143
Dijkstra PD, van Dijk S, Groothuis TGG, Pierotti MER, Seehausen O (2009) Behavioral dominance between female color morphs of a Lake Victoria cichlid fish. Behav Ecol 20:593–600
Donnelly MA (1989) Demographic effects of reproductive resource supplementation in a territorial frog, Dendrobates pumilio. Ecol Monogr 59:207–221
Dreher CE, Cummings ME, Proehl H (2015) An analysis of predator selection to affect aposematic coloration in a poison frog species. PLoS One 10, e0130571
Duckworth RA, Badyaev AV (2007) Coupling of dispersal and aggression facilitates the rapid range expansion of a passerine bird. Proc Natl Acad Sci U S A 104:15017–15022
Dugas MB, Halbrook SR, Killius AM, Sol JF, Richards‐Zawacki CL (2015) Colour and escape behaviour in polymorphic populations of an aposematic poison frog. Ethology 121:813–822
Evans MR, Norris K (1996) The importance of carotenoids in signalling during aggressive interactions between male firemouth cichlids (Cichlasoma meeki). Behav Ecol 7:1–6
Forester D, Cover J, Wisnieski A (1993) The influence of time of residency on the tenacity of territorial defense by the dart-poison frog Dendrobates pumilio. Herpetologica 49:94–99
Gehara M, Summers K, Brown JL (2013) Population expansion, isolation and selection: novel insights on the evolution of color diversity in the strawberry poison frog. Evol Ecol 27:797–824
Grant JB (2007) Ontogenetic colour change and the evolution of aposematism: a case study in panic moth caterpillars. J Anim Ecol 76:439–447
Grether GF, Anderson CN, Drury JP, Kirschel ANG, Losin N, Okamoto K, Peiman KS (2013) The evolutionary consequences of interspecific aggression. Ann N Y Acad Sci 1289:48–68
Healey M, Uller T, Olsson M (2007) Seeing red: morph-specific contest success and survival rates in a colour-polymorphic agamid lizard. Anim Behav 74:337–341
Hegna RH, Saporito RA, Donnelly MA (2013) Not all colors are equal: predation and color polytypism in the aposematic poison frog Oophaga pumilio. Evol Ecol 27:831–845
Heying H (2004) Reproductive limitation by oviposition site in a treehole breeding Madagascan poison frog (Mantella laevigata). Misc Publ Mus Zool Univ Mich 193:23–30
Hödl W, Amézquita A (2001) Visual signaling in anuran amphibians. In: Ryan MJ (ed) Anuran communication. Smithsonian Institute Press, Washington, pp 121–141
Hover EL (1985) Differences in aggressive-behavior between two throat color morphs in a lizard, Urosaurus ornatus. Copeia 1985:933–940
Hsu Y, Earley RL, Wolf LL (2006) Modulation of aggressive behaviour by fighting experience: mechanisms and contest outcomes. Biol Rev Camb Philos 81:33–74
Kenward MG, Roger JH (1997) Small sample inference for fixed effects from restricted maximum likelihood. Biometrics 53:983–997
Laiolo P (2012) Interspecific interactions drive cultural co-evolution and acoustic convergence in syntopic species. J Anim Ecol 81:594–604
Lehtonen TK, McCrary JK, Meyer A (2010) Territorial aggression can be sensitive to the status of heterospecific intruders. Behav Process 84:598–601
Lieberman SS (1986) Ecology of the leaf litter herpetofauna of a neotropical rain forest: La Selva, Costa Rica. Acta Zool Mex 15:1–72
Lötters S, Jungfer KH, Henkel FW, Schmidt W (2007) Poison frogs, biology, species & captive husbandry. In: Edition Chimaira & Serpents Tale. Frankfurt am Main, Germany
Maan ME, Cummings ME (2008) Female preferences for aposematic signal components in a polymorphic poison frog. Evolution 62:2334–2345
Maan ME, Cummings ME (2009) Sexual dimorphism and directional sexual selection on aposematic signals in a poison frog. Proc Natl Acad Sci U S A 106:19072–19077
Maan ME, Cummings ME (2012) Poison frog colors are honest signals of toxicity, particularly for bird predators. Am Nat 179:E1–E14
Merilaita S, Tullberg BS (2005) Constrained camouflage facilitates the evolution of conspicuous warning coloration. Evolution 59:38–45
Mikami OK, Kawata M (2004) Does interspecific territoriality reflect the intensity of ecological interactions? A theoretical model for interspecific territoriality. Evol Ecol Res 6:765–775
Mochida K (2009) A parallel geographical mosaic of morphological and behavioural aposematic traits of the newt, Cynops pyrrhogaster (Urodela: Salamandridae). Biol J Linn Soc 97:613–622
Peiman KS, Robinson BW (2007) Heterospecific aggression and adaptive divergence in brook stickleback (Culaea inconstans). Evolution 61:1327–1338
Peiman KS, Robinson BW (2010) Ecology and evolution of resource-related heterospecific aggression. Q Rev Biol 85:133–158
Pigliucci M (2003) Phenotypic integration: studying the ecology and evolution of complex phenotypes. Ecol Lett 6:265–272
Pröhl H (1997) Territorial behaviour of the strawberry poison-dart frog, Dendrobates pumilio. Amphibia-Reptilia 18:437–442
Pröhl H, Berke O (2001) Spatial distributions of male and female strawberry poison frogs and their relation to female reproductive resources. Oecologia 129:534–542
Pröhl H, Hagemann S, Karsch J, Höbel G (2007) Geographic variation in male sexual signals in strawberry poison frogs (Dendrobates pumilio). Ethology 113:825–837
Pröhl H, Ostrowski T (2011) Behavioural elements reflect phenotypic colour divergence in a poison frog. Evol Ecol 25:993–1015
Prudic KL, Skemp AK, Papaj DR (2007) Aposematic coloration, luminance contrast, and the benefits of conspicuousness. Behav Ecol 18:41–46
Pryke SR, Astheimer LB, Buttemer WA, Griffith SC (2007) Frequency-dependent physiological trade-offs between competing colour morphs. Biol Lett 3:494–497
Pryke SR, Griffith SC (2006) Red dominates black: agonistic signalling among head morphs in the colour polymorphic Gouldian finch. Proc R Soc Lond B 273:949–957
Reynolds RG, Fitzpatrick BM (2007) Assortative mating in poison-dart frogs based on an ecologically important trait. Evolution 61:2253–2259
Richards-Zawacki CL, Cummings ME (2010) Intraspecific reproductive character displacement in a polymorphic poison dart frog, Dendrobates pumilio. Evolution 65:259–267
Robinson SK, Terborgh J (1995) Interspecific aggression and habitat selection by Amazonian birds. J Anim Ecol 64:1–11
Rojas B, Devillechabrolle J, Endler JA (2014) Paradox lost: variable colour-pattern geometry is associated with differences in movement in aposematic frogs. Biol Lett 10:20140193
Rudh A, Breed MF, Qvarnstrom A (2013) Does aggression and explorative behaviour decrease with lost warning coloration? Biol J Linn Soc 108:116–126
Rudh A, Rogell B, Hastad O, Qvarnstrom A (2011) Rapid population divergence linked with co-variation between coloration and sexual display in strawberry poison frogs. Evolution 65:1271–1282
Rudh A, Rogell B, Hoglund J (2007) Non-gradual variation in colour morphs of the strawberry poison frog Dendrobates pumilio: genetic and geographical isolation suggest a role for selection in maintaining polymorphism. Mol Ecol 16:4284–4294
Sacchi R, Pupin F, Gentilli A, Rubolini D, Scali S, Fasola M, Galeotti P (2009) Male-male combats in a polymorphic lizard: residency and size, but not color, affect fighting rules and contest outcome. Aggress Behav 35:274–283
SAS Institute I (2003) SAS/STAT user’s guide Release 91 SAS Institute. Cary, North Carolina
Savage JM (2002) The amphibians and reptiles of Costa Rica: a herpetofauna between two continents, between two seas. University of Chicago Press, Chicago
Seehausen O, Schluter D (2004) Male-male competition and nuptial-colour displacement as a diversifying force in Lake Victoria cichlid fishes. Proc R Soc Lond B 271:1345–1353
Siddiqi A, Cronin TW, Loew ER, Vorobyev M, Summers K (2004) Interspecific and intraspecific views of color signals in the strawberry poison frog Dendrobates pumilio. J Exp Biol 207:2471–2485
Sinervo B, Lively CM (1996) The rock-paper-scissors game and the evolution of alternative male strategies. Nature 380:240–243
Sinervo B, Miles DB, Frankino WA, Klukowski M, DeNardo DF (2000) Testosterone, endurance, and Darwinian fitness: natural and sexual selection on the physiological bases of alternative male behaviors in side-blotched lizards. Horm Behav 38:222–233
Speed MP, Brockhurst MA, Ruxton GD (2010) The dual benefits of aposematism: predator avoidance and enhanced resource collection. Evolution 64:1622–1633
Stamp NE, Wilkens RT (1993) On the cryptic side of life: being unapparent to enemies and the consequences for foraging and growth of caterpillars. In: Caterpillars: ecological and evolutionary constraints on foraging. Chapman & Hall, New York, pp 283–330
Summers K, Symula R, Clough M, Cronin T (1999) Visual mate choice in poison frogs. Proc R Soc Lond B 266:2141–2145
Tazzyman SJ, Iwasa Y (2010) Sexual selection can increase the effect of random genetic drift—a quantitative genetic model of polymorphism in Oophaga pumilio, the strawberry poison-dart frog. Evolution 64:1719–1728
Toft CA (1980) Feeding ecology of thirteen syntopic species of anurans in a seasonal tropical environment. Oecologia 45:131–141
Wang IJ, Shaffer HB (2008) Rapid color evolution in an aposematic species: a phylogenetic analysis of color variation in the strikingly polymorphic strawberry poison-dart frog. Evolution 62:2742–2759
Wilczynski W, Lynch KS, O’Bryant EL (2005) Current research in amphibians: studies integrating endocrinology, behavior, and neurobiology. Horm Behav 48:440–450
Willink B, Brenes-Mora E, Bolanos F, Proehl H (2013) Not everything is black and white: color and behavioral variation reveal a continuum between cryptic and aposematic strategies in a polymorphic poison frog. Evolution 67:2783–2794
Wong S, Parada H, Narins PM (2009) Heterospecific acoustic interference: effects on calling in the frog Oophaga pumilio in Nicaragua. Biotropica 41:74–80
Acknowledgments
We are grateful to M. Caldwell, J. Cronin, J. Davis, B. Elderd, C. Richards-Zawacki, and R. Stevens for their helpful suggestions during the development and execution of this study. Many thanks to C. Richards-Zawacki for providing equipment and to L. Freeborn for providing information about frog localities. For their assistance in the field, we thank D. Gonzales, C. Harris, M. Alvarez, and A. Cuervo. The Smithsonian Tropical Research Institute provided logistical support, and we especially thank G. Jacome, P. Gondola, C. Jaramillo, and the Bocas del Toro Research Station staff. We thank M. Dugas, K. Hovanes, M. Klock, J. Touchon, A. Stuckert, K. Summers, members of the Richards-Zawaki Lab, and two anonymous reviewers for their comments on drafts of the manuscript and D. Blouin for providing statistical advice. This study was supported by the Sigma Xi Chapter of Louisiana State University and by two grants from the Louisiana Environmental and Education Commission from the Louisiana Department of Wildlife and Fisheries.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All applicable international, national, and institutional guidelines for the care and use of animals were followed. Experiments reported in this article comply with the current laws of Panama (ANAM permit No. SC/A-41-12). All procedures performed were in accordance with the ethical standards of Louisiana State University (IACUC No. 13-006) and the Smithsonian Tropical Research Institute (IACUC No. 2012-1015-2015).
Additional information
Communicated by K. Summers
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 244 kb)
Rights and permissions
About this article
Cite this article
Galeano, S.P., Harms, K.E. Coloration in the polymorphic frog Oophaga pumilio associates with level of aggressiveness in intraspecific and interspecific behavioral interactions. Behav Ecol Sociobiol 70, 83–97 (2016). https://doi.org/10.1007/s00265-015-2027-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-015-2027-5