Abstract
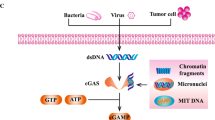
Reovirus, a naturally occurring oncolytic virus, initiates the lysis of tumor cells while simultaneously releasing tumor antigens or proapoptotic cytokines in the tumor microenvironment to augment anticancer immunity. However, reovirus has developed a strategy to evade antiviral immunity via its inhibitory effect on interferon production, which negatively affects the induction of antitumor immune responses. The mammalian adaptor protein Stimulator of Interferon Genes (STING) was identified as a key regulator that orchestrates immune responses by sensing cytosolic DNA derived from pathogens or tumors, resulting in the production of type I interferon. Recent studies reported the role of STING in innate immune responses to RNA viruses leading to the restriction of RNA virus replication. In the current study, we found that reovirus had a reciprocal reaction with a STING agonist regarding type I interferon responses in vitro; however, we found that the combination of reovirus and STING agonist enhanced anti-tumor immunity by enhancing cytotoxic T cell trafficking into tumors, leading to significant tumor regression and survival benefit in a syngeneic colorectal cancer model. Our data indicate the combination of reovirus and a STING agonist to enhance inflammation in the tumor microenvironment might be a strategy to improve oncolytic reovirus immunotherapy.







Similar content being viewed by others
Data availability statement
The data supporting this study's findings are available from the corresponding author (E.K.) upon reasonable request.
Abbreviations
- CRC:
-
Colorectal cancer
- cGAMP:
-
Cyclic GMP-AMP
- cGAS:
-
Cyclic GMP-AMP synthase
- dsDNA:
-
Double-stranded DNA
- FACS:
-
Fluorescence-activated cell sorting
- iNOS:
-
Inducible nitric oxide synthesis
- IFN:
-
Interferon
- IT:
-
Intratumoral
- MOI:
-
Multiplicity of infection
- MSI:
-
Microsatellite instability
- pfu:
-
Plaque-forming units
- PBS:
-
Phosphate buffered saline
- PD-1:
-
Programmed cell death 1
- PD-L1:
-
Programmed cell death ligand 1
- RT-PCR:
-
Reverse transcription–polymerase chain reaction
- STING:
-
Stimulator of Interferon Genes
- TAMs:
-
Tumor-associated macrophages
References
Hashiro G, Loh PC, Yau JT (1977) The preferential cytotoxicity of reovirus for certain transformed cell lines. Arch Virol 54:307–315. https://doi.org/10.1007/BF01314776
Duncan MR, Stanish SM, Cox DC (1978) Differential sensitivity of normal and transformed human cells to reovirus infection. J Virol 28:444–449. https://doi.org/10.1128/JVI.28.2.444-449.1978
Strong JE, Lee PW (1996) The v-erbB oncogene confers enhanced cellular susceptibility to reovirus infection. J Virol 70:612–616. https://doi.org/10.1128/JVI.70.1.612-616.1996
Coffey MC, Strong JE, Forsyth PA, Lee PW (1998) Reovirus therapy of tumors with activated Ras pathway. Science 282:1332–1334. https://doi.org/10.1126/science.282.5392.1332
Hamano S, Mori Y, Aoyama M et al (2015) Oncolytic reovirus combined with trastuzumab enhances antitumor efficacy through TRAIL signaling in human HER2-positive gastric cancer cells. Cancer Lett 356:846–854. https://doi.org/10.1016/j.canlet.2014.10.046
Inagaki Y, Kubota E, Mori Y, Aoyama M, Kataoka H, Johnston RN, Joh T (2017) Anti-tumor efficacy of oncolytic reovirus against gastrointestinal stromal tumor cells. Oncotarget 8:115632–115646. https://doi.org/10.18632/oncotarget.23361
Lawson KA, Mostafa AA, Shi ZQ, Spurrell J, Chen W, Kawakami J, Gratton K, Thakur S, Morris DG (2016) Repurposing sunitinib with oncolytic reovirus as a novel immunotherapeutic strategy for renal cell carcinoma. Clin Cancer Res 22:5839–5850. https://doi.org/10.1158/1078-0432.CCR-16-0143
Prestwich RJ, Errington F, Ilett EJ et al (2008) Tumor infection by oncolytic reovirus primes adaptive antitumor immunity. Clin Cancer Res 14:7358–7366. https://doi.org/10.1158/1078-0432.CCR-08-0831
Prestwich RJ, Ilett EJ, Errington F et al (2009) Immune-mediated antitumor activity of reovirus is required for therapy and is independent of direct viral oncolysis and replication. Clin Cancer Res 15:4374–4381. https://doi.org/10.1158/1078-0432.CCR-09-0334
Rajani K, Parrish C, Kottke T et al (2016) Combination therapy with reovirus and anti-PD-1 blockade controls tumor growth through innate and adaptive immune responses. Mol Ther 24:166–174. https://doi.org/10.1038/mt.2015.156
Ilett E, Kottke T, Thompson J et al (2017) Prime-boost using separate oncolytic viruses in combination with checkpoint blockade improves anti-tumour therapy. Gene Ther 24:21–30. https://doi.org/10.1038/gt.2016.70
Kelly KR, Espitia CM, Zhao W, Wu K, Visconte V, Anwer F, Calton CM, Carew JS, Nawrocki ST (2018) Oncolytic reovirus sensitizes multiple myeloma cells to anti-PD-L1 therapy. Leukemia 32:230–233. https://doi.org/10.1038/leu.2017.272
Groeneveldt C, Kinderman P, van den Wollenberg DJM et al (2020) Preconditioning of the tumor microenvironment with oncolytic reovirus converts CD3-bispecific antibody treatment into effective immunotherapy. J Immunother Cancer. https://doi.org/10.1136/jitc-2020-001191
Sherry B (2009) Rotavirus and reovirus modulation of the interferon response. J Interferon Cytokine Res 29:559–567. https://doi.org/10.1089/jir.2009.0072
Stanifer ML, Kischnick C, Rippert A, Albrecht D, Boulant S (2017) Reovirus inhibits interferon production by sequestering IRF3 into viral factories. Sci Rep 7:10873. https://doi.org/10.1038/s41598-017-11469-6
Corrales L, Glickman LH, McWhirter SM et al (2015) Direct activation of STING in the tumor microenvironment leads to potent and systemic tumor regression and immunity. Cell Rep 11:1018–1030. https://doi.org/10.1016/j.celrep.2015.04.031
Ramanjulu JM, Pesiridis GS, Yang J et al (2018) Design of amidobenzimidazole STING receptor agonists with systemic activity. Nature 564:439–443. https://doi.org/10.1038/s41586-018-0705-y
Berard A, Coombs KM (2009) Mammalian reoviruses: propagation, quantification, and storage. Curr Protoc Microbiol Chapter 15(Unit15C):1. https://doi.org/10.1002/9780471729259.mc15c01s14
Shi P, Zhong J, Hong J, Huang R, Wang K, Chen Y (2016) Automated Ki-67 quantification of immunohistochemical staining image of human nasopharyngeal carcinoma xenografts. Sci Rep 6:32127. https://doi.org/10.1038/srep32127
Morgan E, Arnold M, Gini A et al (2023) Global burden of colorectal cancer in 2020 and 2040: incidence and mortality estimates from GLOBOCAN. Gut 72:338–344. https://doi.org/10.1136/gutjnl-2022-327736
Marcus L, Lemery SJ, Keegan P, Pazdur R (2019) FDA approval summary: pembrolizumab for the treatment of microsatellite instability-high solid tumors. Clin Cancer Res 25:3753–3758. https://doi.org/10.1158/1078-0432.CCR-18-4070
Kawakami H, Zaanan A, Sinicrope FA (2015) Microsatellite instability testing and its role in the management of colorectal cancer. Curr Treat Options Oncol 16:30. https://doi.org/10.1007/s11864-015-0348-2
Muller MF, Ibrahim AE, Arends MJ (2016) Molecular pathological classification of colorectal cancer. Virchows Arch 469:125–134. https://doi.org/10.1007/s00428-016-1956-3
Hirasawa K, Nishikawa SG, Norman KL, Alain T, Kossakowska A, Lee PW (2002) Oncolytic reovirus against ovarian and colon cancer. Cancer Res 62:1696–1701
Adair RA, Scott KJ, Fraser S et al (2013) Cytotoxic and immune-mediated killing of human colorectal cancer by reovirus-loaded blood and liver mononuclear cells. Int J Cancer 132:2327–2338. https://doi.org/10.1002/ijc.27918
Gebremeskel S, Nelson A, Walker B, Oliphant T, Lobert L, Mahoney D, Johnston B (2021) Natural killer T cell immunotherapy combined with oncolytic vesicular stomatitis virus or reovirus treatments differentially increases survival in mouse models of ovarian and breast cancer metastasis. J Immunother Cancer. https://doi.org/10.1136/jitc-2020-002096
van den Wollenberg DJ, van den Hengel SK, Dautzenberg IJ, Cramer SJ, Kranenburg O, Hoeben RC (2008) A strategy for genetic modification of the spike-encoding segment of human reovirus T3D for reovirus targeting. Gene Ther 15:1567–1578. https://doi.org/10.1038/gt.2008.118
Kawagishi T, Kanai Y, Nouda R, Fukui I, Nurdin JA, Matsuura Y, Kobayashi T (2020) Generation of genetically RGD sigma1-modified oncolytic reovirus that enhances JAM-A-independent infection of tumor cells. J Virol. https://doi.org/10.1128/JVI.01703-20
Kottke T, Hall G, Pulido J et al (2010) Antiangiogenic cancer therapy combined with oncolytic virotherapy leads to regression of established tumors in mice. J Clin Invest 120:1551–1560. https://doi.org/10.1172/JCI41431
Roulstone V, Pedersen M, Kyula J et al (2015) BRAF- and MEK-targeted small molecule inhibitors exert enhanced antimelanoma effects in combination with oncolytic reovirus through ER stress. Mol Ther 23:931–942. https://doi.org/10.1038/mt.2015.15
Gong J, Sachdev E, Mita AC, Mita MM (2016) Clinical development of reovirus for cancer therapy: an oncolytic virus with immune-mediated antitumor activity. World J Methodol 6:25–42. https://doi.org/10.5662/wjm.v6.i1.25
Parakrama R, Fogel E, Chandy C, Augustine T, Coffey M, Tesfa L, Goel S, Maitra R (2020) Immune characterization of metastatic colorectal cancer patients post reovirus administration. BMC Cancer 20:569. https://doi.org/10.1186/s12885-020-07038-2
Castle JC, Loewer M, Boegel S et al (2014) Immunomic, genomic and transcriptomic characterization of CT26 colorectal carcinoma. BMC Genomics 15:190. https://doi.org/10.1186/1471-2164-15-190
Yu JW, Bhattacharya S, Yanamandra N et al. (2018) Tumor-immune profiling of murine syngeneic tumor models as a framework to guide mechanistic studies and predict therapy response in distinct tumor microenvironments. PLoS One 13:e0206223. https://doi.org/10.1371/journal.pone.0206223
Woo SR, Corrales L, Gajewski TF (2015) The STING pathway and the T cell-inflamed tumor microenvironment. Trends Immunol 36:250–256. https://doi.org/10.1016/j.it.2015.02.003
Sivick KE, Desbien AL, Glickman LH et al. (2018) Magnitude of therapeutic STING activation determines CD8(+) T cell-mediated anti-tumor immunity. Cell Rep 25: 3074–85. https://doi.org/10.1016/j.celrep.2018.11.047
Phillips MB, Stuart JD, Rodriguez Stewart RM, Berry JT, Mainou BA, Boehme KW (2018) Current understanding of reovirus oncolysis mechanisms. Oncolytic Virother 7:53–63. https://doi.org/10.2147/OV.S143808
Ishikawa H, Barber GN (2008) STING is an endoplasmic reticulum adaptor that facilitates innate immune signalling. Nature 455:674–678. https://doi.org/10.1038/nature07317
Ishikawa H, Ma Z, Barber GN (2009) STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 461:788–792. https://doi.org/10.1038/nature08476
Zevini A, Olagnier D, Hiscott J (2017) Crosstalk between cytoplasmic RIG-I and STING sensing pathways. Trends Immunol 38:194–205. https://doi.org/10.1016/j.it.2016.12.004
Ni G, Ma Z, Damania B (2018) cGAS and STING: at the intersection of DNA and RNA virus-sensing networks. PLoS Pathog 14:e1007148. https://doi.org/10.1371/journal.ppat.1007148
Franz KM, Neidermyer WJ, Tan YJ, Whelan SPJ, Kagan JC (2018) STING-dependent translation inhibition restricts RNA virus replication. Proc Natl Acad Sci U S A 115:E2058–E2067. https://doi.org/10.1073/pnas.1716937115
Farone AL, O’Brien PC, Cox DC (1993) Tumor necrosis factor-alpha induction by reovirus serotype 3. J Leukoc Biol 53:133–137. https://doi.org/10.1002/jlb.53.2.133
Hayman TJ, Baro M, MacNeil T et al (2021) STING enhances cell death through regulation of reactive oxygen species and DNA damage. Nat Commun 12:2327. https://doi.org/10.1038/s41467-021-22572-8
Kwon J, Bakhoum SF (2020) The cytosolic DNA-sensing cGAS-STING pathway in cancer. Cancer Discov 10:26–39. https://doi.org/10.1158/2159-8290.CD-19-0761
Kitajima S, Ivanova E, Guo S et al (2019) Suppression of STING associated with LKB1 loss in KRAS-driven lung cancer. Cancer Discov 9:34–45. https://doi.org/10.1158/2159-8290.CD-18-0689
Katayama Y, Tachibana M, Kurisu N et al (2018) Oncolytic reovirus inhibits immunosuppressive activity of myeloid-derived suppressor cells in a TLR3-dependent manner. J Immunol 200:2987–2999. https://doi.org/10.4049/jimmunol.1700435
Samson A, Bentham MJ, Scott K et al (2018) Oncolytic reovirus as a combined antiviral and anti-tumour agent for the treatment of liver cancer. Gut 67:562–573. https://doi.org/10.1136/gutjnl-2016-312009
Annels NE, Simpson GR, Denyer M, Arif M, Coffey M, Melcher A, Harrington K, Vile R, Pandha H (2021) Oncolytic reovirus-mediated recruitment of early innate immune responses reverses immunotherapy resistance in prostate tumors. Mol Ther Oncolytics 20:434–446. https://doi.org/10.1016/j.omto.2020.09.010
Acknowledgements
We would like to thank Mrs. Yukimi Ito from Nagoya City University for her technical assistance. We thank J. Ludovic Croxford, PhD, from Edanz Group (https://en-author-services.edanzgroup.com/ac) for editing a draft of this manuscript.
Funding
This work was supported by Grants-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science (JSPS), KAKENHI Grant Number 18K07951and 22K16002.
Author information
Authors and Affiliations
Contributions
SN, EK, YM, and HK were involved with the conception and performance of experiments, statistical analysis, and writing the manuscript. MT, TS, and ST assisted with some of the experiments. RNJ and MA were involved with the conception and design of experiments. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors state that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sugimura, N., Kubota, E., Mori, Y. et al. Reovirus combined with a STING agonist enhances anti-tumor immunity in a mouse model of colorectal cancer. Cancer Immunol Immunother 72, 3593–3608 (2023). https://doi.org/10.1007/s00262-023-03509-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-023-03509-0