Abstract
Purpose
To identify the imaging manifestations of splenic involvement in babesiosis, a potentially fatal tick-borne zoonosis with multi-organ involvement.
Methods
In our single center HIPAA compliant IRB-approved study, we performed a retrospective search of the electronic medical record at our institution to identify all patients with known or suspected acute babesiosis from 2000 to 2017. We then reviewed all abdominal imaging of patients with confirmed disease to identify incidence and characteristics of splenic involvement. Splenomegaly was determined using a height- and gender-adjusted reference.
Results
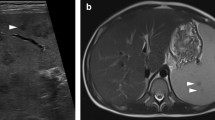
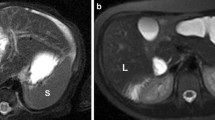
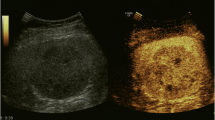
After exclusions, 63 patients with a confirmed diagnosis of babesiosis and contemporaneous imaging of the spleen were included in the final cohort. Within this cohort, 56 (89%) had splenomegaly at a minimum and 13 had splenic infarcts. Splenic rupture was present in eight patients with three having a pseudoaneurysm. In 14 patients with follow-up imaging, the spleen subsequently diminished in size. One additional patient with ruptured spleen underwent emergency splenectomy prior to imaging.
Conclusion
Although the literature suggests splenic involvement is a rare finding, acute parasitemia with babesiosis commonly affects the spleen. Recognition of this association can aid radiologists diagnosing splenic involvement in babesiosis and can lead to appropriate intervention in the minority with splenic hemorrhage.





Similar content being viewed by others
References
E. Vannier, P.J. Krause, Human Babesiosis, N. Engl. J. Med. 366 (2012) 2397–2407. https://doi.org/10.1056/NEJMra1202018.
Spielman, M.L. Wilson, J.F. Levine, J. Piesman, Ecology of Ixodes Dammini-Borne Human Babesiosis and Lyme Disease, Annu. Rev. Entomol. (1985). https://doi.org/10.1146/annurev.en.30.010185.002255.
J.T. Joseph, S.S. Roy, N. Shams, P. Visintainer, R.B. Nadelman, S. Hosur, J. Nelson, G.P. Wormser, Babesiosis in lower Hudson valley, New York, USA, Emerg. Infect. Dis. (2011). https://doi.org/10.3201/eid1705.101334.
D.J. White, J. Talarico, H.G. Chang, G.S. Birkhead, T. Heimberger, D.L. Morse, Human babesiosis in New York State: Review of 139 hospitalized cases and analysis of prognostic factors, Arch. Intern. Med. (1998). https://doi.org/10.1001/archinte.158.19.2149.
J.C. Hatcher, P.D. Greenberg, J. Antique, V.E. Jimenez-Lucho, Severe Babesiosis in Long Island: Review of 34 Cases and Their Complications, Clin. Infect. Dis. (2001). https://doi.org/10.1086/319742.
G.R. Healy, T.K. Ruebush, Morphology of Babesia microti in human blood smears, Am. J. Clin. Pathol. (1980). https://doi.org/10.1093/ajcp/73.1.107.
P.J. Krause, S. Telford, A. Spielman, R. Ryan, J. Magera, T. V. Rajan, D. Christianson, T. V. Alberghini, L. Bow, D. Persing, Comparison of PCR with blood smear and inoculation of small animals for diagnosis of Babesia microti parasitemia, J. Clin. Microbiol. (1996).
H.A. Del Portillo, M. Ferrer, T. Brugat, L. Martin-Jaular, J. Langhorne, M.V.G. Lacerda, The role of the spleen in malaria., Cell. Microbiol. 14 (2012) 343–55. https://doi.org/10.1111/j.1462-5822.2011.01741.x.
S. Li, B. Goyal, J.D. Cooper, A. Abdelbaki, N. Gupta, Y. Kumar, Splenic rupture from babesiosis, an emerging concern? A systematic review of current literature, Ticks Tick. Borne. Dis. (2018). https://doi.org/10.1016/j.ttbdis.2018.06.004.
Dumic, J. Patel, M. Hart, E.R. Niendorf, S. Martin, P. Ramanan, Splenic Rupture as the First Manifestation of Babesia Microti Infection: Report of a Case and Review of Literature., Am. J. Case Rep. 19 (2018) 335–341. https://doi.org/10.12659/ajcr.908453.
K.U. Chow, B. Luxembourg, E. Seifried, H. Bonig, Spleen Size Is Significantly Influenced by Body Height and Sex: Establishment of Normal Values for Spleen Size at US with a Cohort of 1200 Healthy Individuals, Radiology. (2016). https://doi.org/10.1148/radiol.2015150887.
H.K. Rosenberg, R.I. Markowitz, H. Kolberg, C. Park, A. Hubbard, R.D. Bellah, Normal splenic size in infants and children: Sonographic measurements, Am. J. Roentgenol. 157 (1991) 119–121. https://doi.org/10.2214/ajr.157.1.2048509.
E.B. Gray, B.L. Herwaldt, Babesiosis surveillance - United States, 2011-2015, MMWR Surveill. Summ. 68 (2019) 1–16. https://doi.org/10.15585/mmwr.ss6806a1.
Vinasco, W. Braga, O. Zegarra-Moro, M.H. Moro, Cellular Immune Responses in a Murine Model of Borrelia burgdorferi and Babesia microti coinfection (43.55), J. Immunol. 178 (2007).
K.L. Knapp, N.A. Rice, Human coinfection with Borrelia burgdorferi and Babesia microti in the United States, J. Parasitol. Res. (2015). https://doi.org/10.1155/2015/587131.
P.J. Krause, S.R. Telford, A. Spielman, V. Sikand, R. Ryan, D. Christianson, G. Burke, P. Brassard, R. Pollack, J. Peck, D.H. Persing, Concurrent Lyme disease and babesiosis: Evidence for increased severity and duration of illness, J. Am. Med. Assoc. (1996). https://doi.org/10.1001/jama.275.21.1657.
A.C. Steere, N.H. Bartenhagen, J.E. Craft, G.J. Hutchinson, J.H. Newman, D.W. Rahn, L.H. Sigal, P.N. Spieler, K.S. Stenn, S.E. Malawista, The early clinical manifestations of Lyme disease, Ann. Intern. Med. (1983). https://doi.org/10.7326/0003-4819-99-1-76.
F. Rosner, M.H. Zarrabi, J.L. Benach, G.S. Habicht, Babesiosis in splenectomized adults, Am. J. Med. 76 (1984) 696–701. https://doi.org/10.1016/0002-9343(84)90298-5.
J.M. Cullen, J.F. Levine, Pathology of experimental Babesia microti infection in the Syrian hamster., Lab. Anim. Sci. 37 (1987) 640–3. http://www.ncbi.nlm.nih.gov/pubmed/3695401 (accessed August 25, 2019).
I.G. Wright, B.V. Goodger, G.D. Buffington, I.A. Clark, F. Parrodi, D.J. Waltisbuhl, Immunopathophysiology of babesial infections, Trans. R. Soc. Trop. Med. Hyg. 83 (1989) 11–13. https://doi.org/10.1016/0035-9203(89)90596-8.
R. Siderits, N. Mikhail, C. Ricart, M.V. Abello-Poblete, C. Wilcox, J.J. Godyn, Babesiosis, Significance of Spleen Function Illustrated by Postsplenectomy Course in 3 Cases, Infect. Dis. Clin. Pract. 16 (2008) 182–186. https://doi.org/10.1097/IPC.0b013e31809fe523.
D.P. Kuwayama, R.J. Briones, Spontaneous Splenic Rupture Caused by Babesia microti Infection, Clin. Infect. Dis. 46 (2008) e92–e95. https://doi.org/10.1086/587175.
K.M. Patel, J.E. Johnson, R. Reece, L.A. Mermel, Babesiosis-associated Splenic Rupture: Case Series From a Hyperendemic Region, Clin. Infect. Dis. (2018). https://doi.org/10.1093/cid/ciy1060.
B.C. Urban, D.J.P. Ferguson, A. Pain, N. Willcox, M. Plebanski, J.M. Austyn, D.J. Roberts, Plasmodium falciparum-infected erythrocytes modulate the maturation of dendritic cells, Nature. 400 (1999) 73–77. https://doi.org/10.1038/21900.
Oo MM, Aikawa M, Than T, Aye TM, Myint PT, Igarashi I, Schoene WC, Human Cerebral Malaria, J. Neuropathol. Exp. Neurol. 46 (1987) 223–231. https://doi.org/10.1097/00005072-198703000-00009.
T. Akel, N. Mobarakai, Hematologic manifestations of babesiosis., Ann. Clin. Microbiol. Antimicrob. 16 (2017) 6. https://doi.org/10.1186/s12941-017-0179-z.
P.J. Krause, J. Daily, S.R. Telford, E. Vannier, P. Lantos, A. Spielman, Shared features in the pathobiology of babesiosis and malaria., Trends Parasitol. 23 (2007) 605–10. https://doi.org/10.1016/j.pt.2007.09.005.
C.L. Hutchings, A. Li, K.M. Fernandez, T. Fletcher, L.A. Jackson, J.B. Molloy, W.K. Jorgensen, C.T. Lim, B.M. Cooke, New insights into the altered adhesive and mechanical properties of red blood cells parasitized by Babesia bovis, Mol. Microbiol. 65 (2007) 1092–1105. https://doi.org/10.1111/j.1365-2958.2007.05850.x.
J.T. Joseph, K. Purtill, S.J. Wong, J. Munoz, A. Teal, S. Madison-Antenucci, H.W. Horowitz, M.E. Aguero-Rosenfeld, J.M. Moore, C. Abramowsky, G.P. Wormser, Vertical transmission of Babesia microti, United States, Emerg. Infect. Dis. (2012). https://doi.org/10.3201/eid1808.110988.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mojtahed, A., Bates, D.D.B. & Hahn, P.F. Splenic findings in patients with acute babesiosis. Abdom Radiol 45, 710–715 (2020). https://doi.org/10.1007/s00261-019-02362-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-019-02362-z