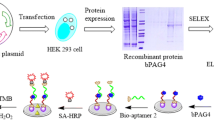
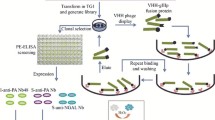
Abstract
Nanobodies (VHH) from camelid antibody libraries hold great promise as therapeutic agents and components of immunoassay systems. Synthetic antibody libraries that could be designed and generated once and for various applications could yield binders to virtually any targets, even for non-immunogenic or toxic ones, in a short term. One of the most difficult tasks is to obtain antibodies with a high affinity and specificity to polyglycosylated proteins. It requires antibody libraries with extremely high functional diversity and the use of sophisticated selection techniques. Here we report a development of a novel sandwich immunoassay involving a combination of the synthetic library-derived VHH-Fc fusion protein as a capture antibody and the immune single-chain fragment variable (scFv) as a tracer for the detection of pregnancy-associated glycoprotein (PAG) of cattle (Bos taurus). We succeeded in the generation of a number of specific scFv antibodies against PAG from the mouse immune library. Subsequent selection using the immobilized scFv-Fc capture antibody allowed to isolate 1.9 nM VHH binder from the diverse synthetic library without any overlapping with the capture antibody binding site. The prototype sandwich ELISA based on the synthetic VHH and the immune scFv was established. This is the first successful example of the combination of synthetic and immune antibody libraries in a single sandwich immunoassay. Thus, our approach could be used for the express isolation of antibody pairs and the development of sandwich immunoassays for challenging antigens.
Key points
• Heavily glycosylated PAG Bos Taurus were used for immune library construction and specific scFv isolation by phage display.
• Nanomolar affinity VHH for PAG was selected from the original synthetic nanobodies library.
• A novel VHH/scFv-based immunoassay for Bos Taurus pregnancy determination was developed.
Graphical abstract









Similar content being viewed by others
Change history
07 July 2022
The sizes of the images of Figs. 1, 4 and 6 are incorrect. This has been updated and corrected to the appropriate size.
References
Almagro JC, Pedraza-Escalona M, Arrieta HI, Pérez-Tapia SM (2019) Phage display libraries for antibody therapeutic discovery and development. Antibodies 8:44. https://doi.org/10.3390/ANTIB8030044
Barbato O, Melo de Sousa N, Barile VL, Canali C, Beckers J-F (2013) Purification of pregnancy-associated glycoproteins from late-pregnancy Bubalus bubalis placentas and development of a radioimmunoassay for pregnancy diagnosis in water buffalo females. BMC Vet Res 9:89. https://doi.org/10.1186/1746-6148-9-89
Bériot M, Tchimbou AF, Barbato O, Beckers J-F, de Sousa NM (2014) Identification of pregnancy-associated glycoproteins and alpha-fetoprotein in fallow deer (Dama dama) placenta. Acta Vet Scand 56:4. https://doi.org/10.1186/1751-0147-56-4
Bradbury ARM, Sidhu S, Dübel S, Mccafferty J (2011) Beyond natural antibodies- the power of in vitro display. 29:245–254
Charbgoo F, Mirshahi M, Sarikhani S, Saifi Abolhassan M (2012) Synthesis of a unique high-performance poly-horseradish peroxidase complex to enhance sensitivity of immunodetection systems. Biotechnol Appl Biochem 59:45–49. https://doi.org/10.1002/BAB.58
Dormeshkin DO, Kuprienko OS, Svirid AV, Gilep AA, Sviridov OV, Usanov SA (2016) Generation and characterization of biotinylated recombinant Fab antibody fragment against cortisol. Russ J Bioorganic Chem 42(1):22–28. https://doi.org/10.1134/S1068162016010039
Frenzel A, Frode D, Meyer T, Schirrmann T, Hust M (2012) Generating recombinant antibodies for research, diagnostics and therapy using phage display. Curr Biotechnology 1:33–41. https://doi.org/10.2174/2211550111201010033
Frey A, Di Canzio J, Zurakowski D (1998) A statistically defined endpoint titer determination method for immunoassays. J Immunol Methods 221:35–41. https://doi.org/10.1016/S0022-1759(98)00170-7
Friedrich M, Holtz W (2010) Establishment of an ELISA for measuring bovine pregnancy-associated glycoprotein in serum or milk and its application for early pregnancy detection. Reprod Domest Anim 45:142–146. https://doi.org/10.1111/J.1439-0531.2008.01287.X
Green JA, Xie S, Quan X, Bao B, Gan X, Mathialagan N, Beckers F, Roberts RM (2000) Pregnancy-associated bovine and ovine glycoproteins exhibit spatially and temporally distinct expression patterns during pregnancy. Biol Reprod 62:1624–1631. https://doi.org/10.1095/BIOLREPROD62.6.1624
Green JA, Parks TE, Avalle MP, Telugu BP, McLain AL, Peterson AJ, McMillan W, Mathialagan N, Hook RR, Xie S, Roberts RM (2005) The establishment of an ELISA for the detection of pregnancy-associated glycoproteins (PAGs) in the serum of pregnant cows and heifers. Theriogenology 63:1481–1503. https://doi.org/10.1016/j.theriogenology.2004.07.011
Harmsen MM, Fijten HPD (2012) Improved functional immobilization of llama single-domain antibody fragments to polystyrene surfaces using small peptides. J Immunoass Immunochem 33(3):234–251. https://doi.org/10.1080/15321819.2011.634473
Jones ML, Alfaleh MA, Kumble S, Zhang S, Osborne GW, Yeh M, Arora N, Hou JJC, Howard CB, Chin DY, Mahler SM (2016) Targeting membrane proteins for antibody discovery using phage display. Sci Rep 6:26240. https://doi.org/10.1038/srep26240
Karen A, De SNM, Beckers JF, Bajcsy ÁC, Tibold J, Mádl I, Szenci O (2015) Comparison of a commercial bovine pregnancy-associated glycoprotein ELISA test and a pregnancy-associated glycoprotein radiomimmunoassay test for early pregnancy diagnosis in dairy cattle. Anim Reprod Sci 159:31–37. https://doi.org/10.1016/j.anireprosci.2015.05.005
Kiracofe GH, Wright JM, Schalles RR, Ruder CA, Parish S, Sasser RG (1993) Pregnancy-specific protein B in serum of postpartum beef cows. J Anim Sci 71:2199–2205. https://doi.org/10.2527/1993.7182199X
Klisch K, Boos A, Friedrich M, Herzog K, Feldmann M, Sousa NM, Beckers JF, Leiser R, Schuler G (2006) The glycosylation of pregnancy-associated glycoproteins and prolactin-related protein-I in bovine binucleate trophoblast giant cells changes before parturition. Reproduction 132(5):791–798. https://doi.org/10.1530/REP-06-0040
Ljungars A, Svensson C, Carlsson A, Birgersson E, Tornberg UC, Frendéus B, Ohlin M, Mattsson M (2019) Deep mining of complex antibody phage pools generated by cell panning enables discovery of rare antibodies binding new targets and epitopes. Front Pharmacol 10:847. https://doi.org/10.3389/fphar.2019.00847
Madsen CB, Petersen C, Lavrsen K, Harndahl M, Buus S, Clausen H, Pedersen AE, Wandall HH (2012) Cancer associated aberrant protein O-glycosylation can modify antigen processing and immune response. PLoS ONE 7(11):e50139. https://doi.org/10.1371/journal.pone.0050139
Majewska M, Panasiewicz G, Klisch K, Olivera LVM, Mamani JM, Abd-Elnaeim MM, Szafranska B (2009) Pregnancy-associated glycoprotein (pag) family: transcripts and gene amplicons in camelids. Reprod Biol 9(2):127–150. https://doi.org/10.1016/S1642-431X(12)60022-9
Moutel S, Bery N, Bernard V, Keller L, Lemesre E, De Marco A, Ligat L, Rain JC, Favre G, Olichon A, Perez F (2016) NaLi-H1: a universal synthetic library of humanized nanobodies providing highly functional antibodies and intrabodies. Elife 5:e16228. https://doi.org/10.7554/eLife.16228
Oberli MA, Tamborrini M, Tsai Y-H, Werz DB, Horlacher T, Adibekian A, Gauss D, Möller HM, Pluschke G, Seeberger PH (2010) Molecular analysis of carbohydrate−antibody interactions: case study using a Bacillus anthracis Tetrasaccharide. J Am Chem Soc 132:10239–10241. https://doi.org/10.1021/ja104027w
Paduch M, Koide A, Uysal S, Rizk SS, Koide S, Kossiakoff AA (2013) Generating conformation-specific synthetic antibodies to trap proteins in selected functional states. Methods 60:3–14. https://doi.org/10.1016/J.YMETH.2012.12.010
Pardon E, Laeremans T, Triest S, Rasmussen SGF, Wohlkönig A, Ruf A, Muyldermans S, Hol WGJ, Kobilka BK, Steyaert J (2014) A general protocol for the generation of nanobodies for structural biology. Nat Protoc. https://doi.org/10.1038/nprot.2014.039
Salvador JP, Vilaplana L, Marco MP (2019) Nanobody: outstanding features for diagnostic and therapeutic applications. Anal Bioanal Chem 411(9):1703–1713. https://doi.org/10.1007/s00216-019-01633-4
Schaefer JV, Honegger A, Plückthun A (2010) Construction of scFv fragments from hybridoma or spleen cells by PCR assembly. Antibody Engineering 1:21–44. https://doi.org/10.1007/978-3-642-01144-3_3
Szenci O, Taverne MAM, Beckers JF, Sulon J, Varga J, Börzsönyi L, Hanzen C, Schekk G (1998) Evaluation of false ultrasonographic diagnoses in cows by measuring plasma levels of bovine pregnancy-associated glycoprotein 1. Vet Rec 142:304–306. https://doi.org/10.1136/vr.142.12.304
Telugu BPVL, Walker AM, Green JA (2009) Characterization of the bovine pregnancy-associated glycoprotein gene family--analysis of gene sequences, regulatory regions within the promoter and expression of selected genes. BMC Genomics 10. https://doi.org/10.1186/1471-2164-10-185
Vincke C, Loris R, Saerens D, Martinez-Rodriguez S, Muyldermans S, Conrath K (2009) General strategy to humanize a camelid single-domain antibody and identification of a universal humanized nanobody scaffold. J Biol Chem 284:3273–3284. https://doi.org/10.1074/jbc.M806889200
Wallace RM, Pohler KG, Smith MF, Green JA (2015) Placental PAGs: gene origins, expression patterns, and use as markers of pregnancy. Reproduction 149:R115–R126. https://doi.org/10.1530/REP-14-0485
Weber J, Peng H, Rader C (2017) From rabbit antibody repertoires to rabbit monoclonal antibodies. Exp Mol Med 493(49):e305–e305. https://doi.org/10.1038/emm.2017.23
Ylera F, Harth S, Waldherr D, Frisch C, Knappik A (2013) Off-rate screening for selection of high-affinity anti-drug antibodies. Anal Biochem 441(2):208–213. https://doi.org/10.1016/j.ab.2013.07.025
Zhang S, Garcia-D’Angeli A, Brennan JP, Huo Q (2013) Predicting detection limits of enzyme-linked immunosorbent assay (ELISA) and bioanalytical techniques in general. Analyst 139:439–445. https://doi.org/10.1039/c3an01835k
Zhang W, Li Z, Wang Z, Yue C, Zheng H, Li R, Zhou M, Hu Z, Wei Z, Li Q (2018) Generation of a monoclonal antibody recognizing the heavily glycosylated CD45 protein and its application on identifying circulating tumor cells. PLoS ONE 13:e0192506. https://doi.org/10.1371/journal.pone.0192506
Zoli AP, Guilbault LA, Delahaut P, Ortiz WB, Beckers JF (1992) Radioimmunoassay of a bovine pregnancy-associated glycoprotein in serum: its application for pregnancy diagnosis. Biol Reprod 46:83–92. https://doi.org/10.1095/biolreprod46.1.83
Acknowledgements
This study was financially supported by the State Committee on Science and Technology. We would like to thank Elena Tumar and Elena Kisileva at the Institute of Bioorganic Chemistry of NASB for their kind assistance with mouse immunizations.
Author information
Authors and Affiliations
Contributions
DD, MS, and AG designed the study; IK and ES performed bovine cotyledon extraction and bovine sample preparation; MS purified PAGs; DD performed phage display selections; MS and AlK purified and analyzed anti-PAG VHH and scFv; AnK produced VHH-Fc in FreeStyle™ 293-F cells; DD analyzed the data and wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The animal experiments were approved by the Institute of Experimental Veterinary Medicine named S. N. Vyshelesski, the Animal Care and Use Committee. The study was performed in accordance with the Belarussian legal and ethical requirements for appropriate animal procedures. Ruminants were not purposely euthanized for this study. Tissue samples were taken during the conventional slaughter process. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dormeshkin, D., Shapira, M., Karputs, A. et al. Combining of synthetic VHH and immune scFv libraries for pregnancy-associated glycoproteins ELISA development. Appl Microbiol Biotechnol 106, 5093–5103 (2022). https://doi.org/10.1007/s00253-022-12022-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-12022-w