Abstract
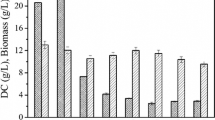
α, ω-Dicarboxylic acids (DCAs) are multipurpose chemicals widely used in polymers, perfumes, plasticizers, lubricants, and adhesives. The biotransformation of DCAs from alkanes and fatty acids by microorganisms has attracted recent interest, since synthesis via chemical oxidation causes problems in terms of the environment and safety. We isolated an ω-oxidizing yeast from a wastewater disposal facility of a petrochemical factory by chemostat enrichment culture. The haploid strain identified as Candida sorbophila DS02 grew on glucose and dodecane, exhibiting greater cell shrinkage on the latter. In flask cultures with mixed alkanes (C10–16) and fatty acid methyl esters (C10–16), DS02 used mixed alkanes simultaneously unlike Candida tropicalis and Yarrowia lipolytica and showed high substrate resistance. In flask cultures with acrylic acid—a known inhibitor of β-oxidation—DS02 produced 0.28 g/l dodecanedioic acid (DDDA) from dodecane, similar to wild-type C. tropicalis ATCC 20336. In fed-batch fermentation, DS02 produced 9.87 g/l DDDA, which was 5.7-fold higher than wild-type C. tropicalis. These results suggest that C. sorbophila strain DS02 has potential applications for the large-scale production of DCA.







Similar content being viewed by others
References
Alexandre H, Mathieu B, Charpentier C (1996) Alteration in membrane fluidity and lipid composition, and modulation of H+-ATPase activity in Saccharomyces cerevisiae caused by decanoic acid. Microbiology 142:469–475
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Cao Z, Gao H, Liu M, Jiao P (2006) Engineering the acetyl-CoA transportation system of Candida tropicalis enhances the production of dicarboxylic acid. Biotechnol J 1:68–74
Cornils B, Lappe P, Staff U (2014) Dicarboxylic acids, aliphatic. In: Ullmann’s encyclopedia of industrial chemistry. Wiley-VCH, Weinheim, pp 1–18
Craft DL, Madduri KM, Eshoo M, Wilson CR (2003) Identification and characterization of the CYP52 family of Candida tropicalis ATCC 20336, important for the conversion of fatty acids and alkanes to α, ω-dicarboxylic acids. Appl Environ Microbiol 69:5983–5991
Eirich LD, Craft DL, Steinberg L, Asif A, Eschenfeldt WH, Stols L, Donnelly MI, Wilson CR (2004) Cloning and characterization of three fatty alcohol oxidase genes from Candida tropicalis strain ATCC 20336. Appl Environ Microbiol 70:4872–4879
Eschenfeldt WH, Zhang Y, Samaha H, Stols L, Eirich LD, Wilson CR, Donnelly MI (2003) Transformation of fatty acids catalyzed by cytochrome P450 monooxygenase enzymes of Candida tropicalis. Appl Environ Microbiol 69:5992–5999
Fickers P, Benetti P-H, Wache Y, Marty A, Mauersberger S, Smit M, Nicaud J-M (2005) Hydrophobic substrate utilisation by the yeast Yarrowia lipolytica, and its potential applications. FEMS Yeast Res 5:527–543
Fukuda R (2013) Metabolism of hydrophobic carbon sources and regulation of it in n-alkane-assimilating yeast Yarrowia lipolytica. Biosci Biotechnol Biochem 77:1149–1154
Granot D, Levine A, Dor-Hefetz E (2003) Sugar-induced apoptosis in yeast cells. FEMS Yeast Res 4:7–13
Haase SB, Reed SI (2002) Improved flow cytometric analysis of the budding yeast cell cycle. Cell Cycle 1:117–121
Hettema EH, Tabak HF (2000) Transport of fatty acids and metabolites across the peroxisomal membrane. Biochim Biophys Acta 1486:18–27
Huf S, Krügener S, Hirth T, Rupp S, Zibek S (2011) Biotechnological synthesis of long-chain dicarboxylic acids as building blocks for polymers. Eur J Lipid Sci Technol 113:548–561
Iwama R, Kobayashi S, Ishimaru C, Ohta A, Horiuchi H, Fukuda R (2016) Functional roles and substrate specificities of twelve cytochromes P450 belonging to CYP52 family in n-alkane assimilating yeast Yarrowia lipolytica. Fungal Genet Biol 91:43–54
Kothavade RJ, Kura M, Valand AG, Panthaki M (2010) Candida tropicalis: its prevalence, pathogenicity and increasing resistance to fluconazole. J Med Microbiol 59:873–880
Kurtzman CP, Robnett CJ (1998) Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Antonie Van Leeuwenhoek 73:331–371
Lee H-W, Yoon S-J, Kim H-K, Park K-M, Oh T-K, Jung J-K (2000) Overexpression of an alkaline lipase gene from Proteus vulgaris K80 in Escherichia coli BL21/pKLE. Biotechnol Lett 22:1543–1547
Liu P, Chernyshov A, Najdi T, Fu Y, Dickerson J, Sandmeyer S, Jarboe L (2013) Membrane stress caused by octanoic acid in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 97:3239–3251
Liu S, Li C, Fang X, Za C (2004) Optimal pH control strategy for high-level production of long-chain α, ω-dicarboxylic acid by Candida tropicalis. Enzym Microb Technol 34:73–77
Lu X, Zhang J, Wu Q, Chen G-Q (2003) Enhanced production of poly (3-hydroxybutyrate-co-3-hydroxyhexanoate) via manipulating the fatty acid β-oxidation pathway in E. coli. FEMS Microbiol Lett 221:97–101
Luo Y, Wang J, Liu B, Wang Z, Yuan Y, Yue T (2015) Effect of yeast cell morphology, cell wall physical structure and chemical composition on patulin adsorption. PLoS One 10:e0136045
Mayer FL, Wilson D, Hube B (2013) Candida albicans pathogenicity mechanisms. Virulence 4:119–128
Mishra P, Park GY, Lakshmanan M, Lee HS, Lee H, Chang MW, Ching CB, Ahn J, Lee DY (2016) Genome-scale metabolic modeling and in silico analysis of lipid accumulating yeast Candida tropicalis for dicarboxylic acid production. Biotechnol Bioeng 113:1993–2004
Munoz AJ, Wanichthanarak K, Meza E, Petranovic D (2012) Systems biology of yeast cell death. FEMS Yeast Res 12:249–265
Nakase T (1975) Three new asporogenous yeasts found in industrial waste water. Antonie Van Leeuwenhoek 41:201–210
Picataggio S, Rohrer T, Deanda K, Lanning D, Reynolds R, Mielenz J, Eirich LD (1992) Metabolic engineering of Candida tropicalis for the production of long–chain dicarboxylic acids. Nat Biotechnol 10:894–898
Pscheidt B, Glieder A (2008) Yeast cell factories for fine chemical and API production. Microb Cell Factories 7:1
Qi Q, Steinbüchel A, Rehm BH (1998) Metabolic routing towards polyhydroxyalkanoic acid synthesis in recombinant Escherichia coli (fadR): inhibition of fatty acid β-oxidation by acrylic acid. FEMS Microbiol Lett 167:89–94
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Stanbury PF, Whitaker A, Hall SJ (1995) The isolation, preservation and improvement of industrially important micro-organisms. In: Principles of fermentation technology, 2nd edn. Pergamon Press, Oxford, pp 35–91
Świzdor A, Panek A, Milecka-Tronina N, Kołek T (2012) Biotransformations utilizing β-oxidation cycle reactions in the synthesis of natural compounds and medicines. Int J Mol Sci 13:16514–16543
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Thijsse G (1964) Fatty-acid accumulation by acrylate inhibition of β-oxidation in an alkane-oxidizing Pseudomonas. Biochim Biophys Acta 84:195–197
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Van Bogaert IN, Groeneboer S, Saerens K, Soetaert W (2011) The role of cytochrome P450 monooxygenases in microbial fatty acid metabolism. FEBS J 278:206–221
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: PCR protocols: a guide to methods and applications. Academic Press, San Diego, pp 315–322
Acknowledgements
This research was supported by the Research Initiative Program of Korea Research Institute of Bioscience and Biotechnology (KGM4231713) and the Global R&D Project Program of the Ministry of Trade, Industry and Energy of Korea (N0000677).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Electronic supplementary material
ESM 1
(PDF 203 kb).
Rights and permissions
About this article
Cite this article
Lee, H., Sugiharto, Y.E.C., Lee, S. et al. Characterization of the newly isolated ω-oxidizing yeast Candida sorbophila DS02 and its potential applications in long-chain dicarboxylic acid production. Appl Microbiol Biotechnol 101, 6333–6342 (2017). https://doi.org/10.1007/s00253-017-8321-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8321-6