Abstract
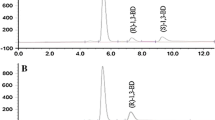
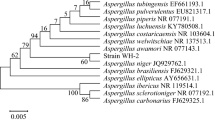
Biocatalytic asymmetric preparation of (R)-1,3-butanediol has been attracting much attention in pharmaceuticals industry. A new ideal strain, ZJB-09162, which exhibited high reduction activity and excellent (R)-stereospecificity towards 4-hydroxy-2-butanone, has been successfully isolated from soil samples. Based on morphology, physiological tests (API 20 C AUX), and 5.8S-ITS sequence, the isolate was identified as Candida krusei. Kinetic characterization demonstrated that carbonyl reductase from C. krusei ZJB-09162 preferred NADH to NADPH as cofactor, indicating it might be a new carbonyl reductase. (R)-1,3-Butanediol was produced in 19.8 g/L, 96.6% conversion, and 99.0% ee at optimal pH 8.5, 35 °C with a 2:1 molar ratio of glucose to 4H2B. In order to achieve higher product titer, the substrate loading was optimized in fixed catalysts and fixed substrate/catalysts ratio mode. The bioreduction of 4-hydroxy-2-butanone at a concentration of 45.0 g/L gave (R)-1,3-butanediol in 38.7 g/L and 83.9% conversion. Therefore, C. krusei ZJB-09162 was, for the first time, proven to be a promising biocatalyst for enzymatic preparation of (R)-1,3-butanediol.







Similar content being viewed by others
References
Arias CR, Burns JK, Friedrich LM, Goodrich RM, Parish ME (2002) Yeast species associated with orange juice: evaluation of different identification method. Appl Environ Microbiol 68:1955–1961
Asako H, Shimizu M, Makino Y, Itoh N (2010) Biocatalytic reduction system for the production of chiral methyl (R)/(S)-4-bromo-3-hydroxybutyrate. Tetrahedron Lett 51:2644–2666
Boaz NW, Ponasik JA, Large SE (2006) Ruthenium complexes of phosphine-aminophosphine ligands. Tetrahedron Lett 47:4033–4035
Bradford M (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principles of protein-dye binding. Anal Biochem 72:248–254
Chen J, Zheng RC, Zheng YG, Shen YC (2009) Microbial transformation of nitriles to high-value acids or amides. Adv Biochem Eng Biotechnol 113:33–77
Choi YH, Choi HJ, Kim D, Uhm K, Kim H (2010) Asymmetric sysnthesis of (S)-3-chloro-1-phenyl-1-propanol using Saccharomyces cerevisiae reductase with high enantioselectivity. Appl Microbiol Biotechnol 87:185–193
Eguchi T, Mochida K (1993) Lipase-catalyzed diacylation of 1,3-butanediol. Biotechnol Lett 15:955–960
Itoh N, Nakamura M, Inoue K, Makino Y (2007) Continuous production of chiral 1,3-butanediol using immobilized biocatalysts in a packed bed reactor: promising biocatalysis method with an asymmetric hydrogen-transfer bioreduction. Appl Microbial Biotechnol 75:1249–1256
Ju X, Yu HL, Pan J, Wei DZ, Xu JH (2010) Bioproduction of chiral mandelate by enantioselective deacylation of α-acetoxyphenylacetic acid using whole cells of newly isolated Pseudomonas sp, ECU1011. Appl Microbiol Biotechnol 86:83–91
Kataota M, Sakai H, Morikawa T, Katoh M, Miyoshi (1992) Characterization of aldehyde reductase of Sporobolomyces salmonicolor. Biochim Biophys Acta 1122:57–62
Katz M, Frejd T, Hahn-Hägerdal B, Gorwa-Grauslund MF (2003) Efficient anaerobic whole cell stereoselective bioreduction with recombinant Saccharomyces cerevisiae. Biotechnol Bioeng 84:573–582
Katzberg M, Skorupa-Parachin N, Gorwa-Granslund M-F, Bertau M (2010) Engineering cofactor preference of ketone reducing biocatalysts: a mutagenesis study on a γ-diketone reductase from the yeast Saccharomyces cerevisiae serving as an example. Int J Mol Sci 11:1735–1758
Kira I, Onishi N (2009) Purification and characterization of a (R)-1-phenyl-1,3-propanediol-producing enzyme from Trichosporon fermentans AJ-5152 and enzymatic (R)-1-phenyl-1,3-propanediol production. Biosci Biotechnol Biochem 73:1640–1646
Kumar S, Tamura K, Jakobsen IB, Nei M (2001) MEGA2: molecular evolutionary genetics analysis software. Bioinformatics 17:1244–1245
Kurtzman CP, Fell JW (eds) (1998) The yeasts. A taxonomic study, 4th edn. Elsevier Science B.V, Amsterdam
Larchevêque M, Mambu L, Petit Y (1991) Preparation of enantiomerically pure 1,3-butanediol from threonine. Synth Commun 21:2295–2300
Li N, Ni Y, Sun ZH (2010) Purification and characterization of carbonyl reductase from Candida krusei SW2026 involved in enantioselective reduction of ethyl 2-oxo-4-phenylbutyrate. J Mol Cata B Enzym 66:190–197
Liu ZQ, Zhang JF, Zheng YG, Shen YC (2008) Improvement of astaxanthin production by a newly isolated phaffia rhodozyma mutant with low energy ion beam implantation. J Appl Microbiol 104:861–872
Matsuda T, Yamanaka R, Nakamura K (2009) Recent progress in biocatalysis for asymmetric oxidation and reduction. Tetrahedron-Asymmetry 20:513–557
Matsuyama A, Kobayashi Y, Ohnishi H (1993) Microbial production of optically active 1,3-butanediol from 4-hydroxy-2-butanone. Biosci Biotechnol Biochem 57:348–349
Matsuyama A, Yamamoto H, Kawada N, Kobayashi Y (2001) Industrial production of (R)-1,3-butanediol by new biocatalysts. J Mol Catal B Enzym 11:513–521
Nakatsuka T, Iwata H, Tanaka R, Imajo S, Ishiguro M (1991) A facile conversion of the phenylthio group to acetoxy by copper reagents for a practical synthesis of 4-acetoxyazetidin-2-one derivatives from (R)-butane-1,3-diol. J Chem Soc Chem Commun 9:662–664
Nie Y, Xu Y, Hu QS, Xiao R (2009) A new strategy to improve the efficiency and sustainability of Candida parapsilosis catalyzing deracemization of (R, S)-1-phenyl-1,2-ethanediol under non-growing conditions: increase of NADPH availability. J Microbiol Biotechnol 19:65–71
Tao JH, Xu JH (2009) Biocatalysis in development of green pharmaceutical processes. Curr Opin Chem Biol 12:43–50
Thompson JD, Higgins DJ, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position specific gap penalties and weight matrix choice. Nucleic Acid Res 22:4673–4680
Wada M, Kataoka M, Kawabata H, Yasohara Y, Kizaki N, Hasegawa J, Shimizu S (1998) Purification and characterization of NADHP-dependent carbonyl reductase, involved in stereoselective reduction of ethyl 4-chloro-3-oxobutanote, from Candida magnoliae. Biosci Biotechnol Biochem 62:280–285
White TJ, Tayor J, Lee S, Burns T (1990) Amplification and direct sequencing of fungal ribosomal RNA for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic, San Diego, CA, pp 315–322
Yamamoto H, Kimoto N, Matsuyama A, Kobayashi Y (2002a) Purification and properties of a carbonyl reductase useful for production of ethyl (S)-4-chloro-3-hydroxybutanoate from Kluyveromyces lactis. Biosci Biotechnol Biochem 66:1775–1778
Yamamoto H, Matsuyama A, Kobayashi Y (2002b) Synthesis of (R)-1,3-butanediol by enantioselective oxidation using whole recombinant Esherichia coli cells expressing (S)-specific secondary alcohol dehydrogenase. Biosci Biotechnol Biochem 66:925–927
Yang W, Xu JH, Pan J, Xu Y, Wang ZL (2008) Efficient reduction of aromatic ketones with NADPH regeneration by using crude enzyme from Rhodotorula cells and mannitol as cosubstrate. Biochem Eng J 42:1–5
Ye Q, Yan M, Yao Z, Xu L, Cao H, Li ZJ, Chen Y, Li SY, Bai JX, Xiong J, Ying HJ, Ouyang PK (2009) A new member of the short-chain dehydrogenases/reductases superfamily: purification, characterization and substrate specificity of a recombinant carbonyl reductase from Pichia stipitis. Bioresour Technol 100:6022–6027
Ye Q, Zang GL, Lan M, Yan M, Wang Y, Zhang YY, Li XM, Li J, Xu L, Xiong J, Ouyang PK, Ying HJ (2010) Biocatalytic synthesis of (S)-4-chloro-3-hydroxybutanoate ethyl ester using a recombinant whole-cell catalyst. Appl Microbiol Biotechnol 88:1277–1285
Zhang W, Ni Y, Sun Z, Zheng P, Lin W, Zhu P, Ju N (2009a) Biocatalytic synthesis of ethyl (R)-2-hydroxy-4-phenylbutyrate with Candida krusei SW2026: a practical process for high enantiopurity and product titer. Process Biochem 44:1270–1275
Zhang R, Xu Y, Sun Y, Zhang W, Xiao R (2009b) Ser67Asp and His68Asp substitutions in Candida parapsilosis carbonyl reductase alter the coenzyme specificity and enantioselectivity of ketone reduction. Appl Environ Microbiol 75:2176–2183
Zheng RC, Wang YS, Liu ZQ, Xing LY, Zheng YG, Shen YC (2007) Isolation and characterization of Delfia tsuruhatensis ZJB-05174, capable of R-enantioselective degradation of 2,2-dimethylcyclopropanecarboxamide. Res Microbiol 158:258–264
Acknowledgements
We are grateful to the Key Scientific and Technological Programs of Zhejiang Province (no. 2009 C13033-3), 973 Program (no. 2011CB710806), Educational Commission of Zhejiang Province of China (no. Y201018390), and the Key Innovation Research Group Programs of Zhejiang Province (no. 2011R09043-04) for the financial support of this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zheng, RC., Ge, Z., Qiu, ZK. et al. Asymmetric synthesis of (R)-1,3-butanediol from 4-hydroxy-2-butanone by a newly isolated strain Candida krusei ZJB-09162. Appl Microbiol Biotechnol 94, 969–976 (2012). https://doi.org/10.1007/s00253-012-3942-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-3942-2