Abstract
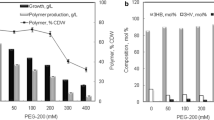
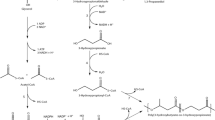
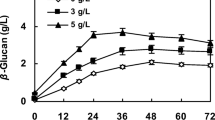
Poly (β-l-malic acid) (PMLA) is a water-soluble polyester with many attractive properties in chemical industry and medicine development. However, the low titer of PMLA in the available producer strains limits further industrialization efforts and restricts its many potential applications. In order to solve this problem, a new strain with the distinguished high productivity of PMLA was isolated from fresh plants samples. It was characterized as the candidate of Aureobasidium pullulans based on the morphology and phylogenetic analyses of the internal transcribed spacer sequences. After the optimization of culture conditions, the highest PMLA concentration (62.27 g l−1) could be achieved in the shake flask scale. In addition, the contribution of the carbon flux to exopolysaccharide (EPS) and PMLA could be regulated by the addition of CaCO3 in the medium. This high-level fermentation process was further scaled up in the 10 l benchtop fermentor with a high PMLA concentration (57.2 g l−1) and productivity (0.35 g l−1 h−1), which are the highest level in all the literature. Finally, the suitable acid hydrolysis conditions of PMLA were also investigated with regard to the production of l-malic acid, and the kinetics of PMLA acid hydrolysis was modeled to simulate the whole degradation process. The present work paved the road to produce this multifunctional biomaterial (PMLA) at industrial scale and promised one alternative method to produce l-malic acid in the future.






Similar content being viewed by others
References
Angerer B, Holler E (1995) Large complexes of β-poly (l-malate) with DNA polymerase-α, histones, and other proteins in nuclei of growing plasmodia of Physarum polycephalum. Biochemistry 34:14741–14751
Cammas S, Guerin P, Girault JP, Holler E, Gache Y, Vert M (1993) Natural poly (l-malic acid): NMR shows a poly (3-hydroxy acid)-type structure. Macromolecules 26:4681–4684
Carol A, Engel R, Straathof AJJ, Zijlmans TW, van Gulik WM, van der Wielen LAM (2008) Fumaric acid production by fermentation. Appl Microbiol Biotechnol 78:379–389
Chi ZM, Wang F, Chi Z, Yue LX, Liu GL, Zhang T (2009) Bioproducts from Aureobasidium pullulans, a biotechnologically important yeast. Appl Microbiol Biotechnol 82:793–804
Dubois M, Gilles KA, Hamilton J, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Fischer H, Erdmann S, Holler E (1989) An unusual polyanion from Physarum polycephalum that inhibits homologous DNA polymerase α in vitro. Biochemistry 28:5219–5226
Gasslmaier B, Holler E (1997) Specificity and direction of depolymerization of β-poly (l-malate) catalysed by polymalatase from Physarum polycephalum. Fluorescence labeling at the carboxy-terminus of β-poly(l-malate). Eur J Biochem 250:308–314
Goldberg I, Rokem JS, Pines O (2006) Organic acids: old metabolites, new themes. J Chem Technol Biotechnol 81:1601–1611
Jeanmougin F, Thompson JD, Gouy M, Higgins DG, Gibson TJ (1998) Multiple sequence alignment with Clustal X. Trends Biochem Sci 23:403–405
Leathers TD (2003) Biotechnological production and applications of pullulan. Appl Microbiol Biotechnol 62:468–473
Lee BS, Holler E (1999) Effects of culture conditions on β-poly(l-malate) production by Physarum polycephalum. Appl Microbiol Biotechnol 51:647–652
Lee BS, Holler E (2000) β-Poly (l-malate) production by non-growing microplasmodia of Physarum polycephalum effects of metabolic intermediates and inhibitors. FEMS Microbiol Lett 193:69–74
Lee BS, Maurer T, Kalbitzer HR, Holler E (1999) β-Poly(l-malate) production by Physarum polycephalum 13C nuclear magnetic resonance studies. Appl Microbiol Biotechnol 52:415–420
Lee BS, Vert M, Holler E (2002) Water-soluble aliphatic polyesters: poly(malic acid)s. In: Doi Y, Steinbuechel A (eds) Biopolymers, vol. 3, polyesters I. Wiley-VCH, Weinheim, pp 75–103
Li BX, Zhang N, Peng Q, Yin T, Guan FF, Wang GL, Li Y (2009) Production of pigment-free pullulan by swollen cell in Aureobasidium pullulans NG which cell differentiation was affected by pH and nutrition. Appl Microbiol Biotechnol 84:293–300
Liu SJ, Steinbüchel A (1997) Production of poly(malic acid) from different carbon sources and its regulation in Aureobasidium pullulans. Biotechnol Lett 19:11–14
Liu S, Steinübchel A (1996) Investigation of poly(β-l-malic acid) production by strains of Aureobasidium pullulans. Appl Microbiol Biotechnol 46:273–278
Ljubimova JY, Fujita M, Khazenzon NM, Lee BS, Wachsmann-Hogiu S, Farkas DL, Black KL, Holler E (2008) Nanoconjugate based on polymalic acid for tumor targeting. Chem Biol Interact 171:195–203
Nagata N, Nakahara T, Tabuchi T (1993) Fermentative production of poly(β-l-malic acid), a polyelectrolytic biopolyester, by Aureobasidium sp. Biosci Biotechnol Biochem 57:638–642
Portilla-Arias JA, García-Alvarez M, Galbis JA, Munoz-Guerra S (2008) Biodegradable nanoparticles of partially methylated fungal poly(β-l-malic acid) as a novel protein delivery carrier. Macromol Biosci 8:551–559
Rathberger K, Reisner H, Willibald B, Molitoris HP, Holler E (1999) Comparative synthesis and hydrolytic degradation of poly (l-malate) by myxomycetes and fungi. Mycol Res 103:513–520
Schmidt A, Windisch C, Holler E (1996) Nuclear accumulation and homeostasis of the unusual polymer poly (l-malate) in plasmodia of Physarum polycephalum. Eur J Cell Biol 70:373–380
Schwartz H, Radler F (1988) Formation of l (−) malate by Saccharomyces cerevisiae during fermentation. Appl Microbiol Biotechnol 27:553–560
Singh RS, Saini GK (2008) Pullulan-hyperproducing color variant strain of Aureobasidium pullulans FB-1 newly isolated from phylloplane of Ficus sp. Bioresour Technol 99:3896–3899
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Vert M, Lenz RW (1979) Preparation and properties of poly-β-malic acid: a functional polyester of potential biomedical importance. Polym Prep 20:608–611
Vert M, Li SM, Spenlehauer G, Guerin P (1992) Bioresorbability and biocompatibility of aliphatic polyesters. J Mater Sci Mater Med 3:432–446
Werpy T, Peterson G (2004) Top value added chemicals from biomass, volume 1. Results of screening for potential candidates from sugars and synthesis gas. US Department of Energy, Washington, DC
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA et al (eds) PCR protocols. Academic, San Diego, pp 315–322
Zalar P, Gostinčar C, de Hoog GS, Uršič V, Sudhadham M, Gunde-Cimerman N (2008) Redefinition of Aureobasidium pullulans and its varieties. Stud Mycol 61:21–38
Zelle RM, de Hulster E, Kloezen W, Pronk JT, van Maris AJA (2010) Key process conditions for production of C4 dicarboxylic acids in bioreactor batch cultures of an engineered Saccharomyces cerevisiae strain. Appl Environ Microbiol 76:744–750
Acknowledgments
This work was financially supported by the Ministry of Science and Technology of China (Grant No. 2009AA02Z206 and 2007CB707805), The National Natural Science Foundation of China (Grant No. 20736008), and The Natural Science Foundation of Zhejiang Province (Grant No. R4090041), People’s Republic of China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, H., Cai, J., Dong, J. et al. High-level production of poly (β-l-malic acid) with a new isolated Aureobasidium pullulans strain. Appl Microbiol Biotechnol 92, 295–303 (2011). https://doi.org/10.1007/s00253-011-3358-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3358-4