Abstract
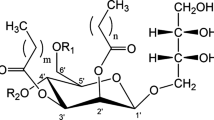
Mannosylerythritol lipids (MELs), which are glycolipid biosurfactants produced by Pseudozyma yeasts, show not only excellent interfacial properties but also versatile biochemical actions. In the course of MEL production from glucose as the sole carbon source, P. antarctica was found to produce unknown glycolipids more hydrophilic than conventional “di-acylated MELs,” which have two fatty acyl esters on the mannose moiety. Based on a detailed characterization, the most hydrophilic one was identified as 4-O-(3′-O-alka(e)noyl-β-d-mannopyranosyl)-d-erythritol namely, “mono-acylated MEL.” The mono-acylated MEL reduced the surface tension of water to 33.8 mN/m at a critical micelle concentration (CMC) of 3.6 × 10−4 M, and its hydrophilic–lipophilic balance was tentatively calculated to be 12.15. The observed CMC was 100-fold higher than that of the MELs hitherto reported. Interestingly, of the yeast strains of the genus Pseudozyma, only P. antarctica and P. parantarctica gave the mono-acylated MEL from glucose, despite a great diversity of di-acylated MEL producers in the genus. These strains produced MELs including the mono-acylated one at a rate of 20–25%. From these results, the new MEL is likely to have great potential for use in oil-in-water-type emulsifiers and washing detergents because of its higher water solubility compared to conventional MELs and will thus contribute to facilitating a broad range of applications for the environmentally advanced surfactants.








Similar content being viewed by others
References
Abalos A, Pinazo A, Infante MR, Casals M, García F, Manresa A (2001) Physicochemical and antimicrobial properties of new rhamnolipids produced by Pseudomonas aeruginosa AT10 from soybean oil refinery wastes. Langmuir 17:1367–1371
Banat IM, Makkar RS, Cameotra SS (2000) Potential commercial applications of microbial surfactants. Appl Microbiol Biotechnol 53:495–508
Bistline RG, Maurer EW, Smith FD, Lifield WM (1980) Fatty acid amides and anilides, synthesis and antimicrobial properties. J Am Oil Chem Soc 57:98–103
Fukuoka T, Morita T, Konishi M, Imura T, Kitamoto D (2007) Characterization of new glycolipid biosurfactants, tri-acylated mannosylerythritol lipids, produced by Pseudozyma yeasts. Biotechnol Lett 29:1111–1118
Griffin WC (1954) Calculations of HLB values of nonionic surfactants. J Soc Cosmet Chem 5:249–256
Im JH, Ikegami T, Yanagishita H, Takeyama Y, Idemoto Y, Koura N, Kitamoto D (2003) Mannosylerythritol lipids, yeast glycolipid biosurfactants, are potential affinity ligand materials for human immunoglobulin G. J Biomed Mater Res 65:379–385
Imura T, Yanagishita H, Kitamoto D (2004) Coacervate formation from natural glycolipid: one acetyl group on the headgroup triggers coacervate-to-vesicle transition. J Am Chem Soc 126:10804–10805
Imura T, Yanagishita H, Ohira J, Sakai H, Abe M, Kitamoto D (2005) Thermodynamically stable vesicle formation from glycolipid biosurfactant sponge phase. Colloids Surf B Biointerfaces 43:114–121
Imura T, Ohta N, Inoue K, Yagi H, Negishi H, Yanagishita H, Kitamoto D (2006) Naturally engineered glycolipid biosurfactants leading to distinctive self-assembled structures. Chem Eur J 12:2434–2440
Imura T, Hikosaka Y, Worakitkanchanakul W, Sakai H, Abe M, Konishi M, Minamikawa H, Kitamoto D (2007) Aqueous-phase behavior of natural glycolipid biosurfactant mannosylerythritol lipid A: sponge, cubic, and lamellar phases. Langmuir 23:1659–1663
Isoda H, Shinmoto H, Kitamoto D, Matsumura M, Nakahara T (1997) Differentiation of human promyelocytic leukemia cell line HL60 by microbial extracellular glycolipids. Lipids 32:263–271
Kakugawa K, Tamai M, Imamura K, Miyamoto K, Miyoshi S, Morinaga Y, Suzuki O, Miyakawa T (2002) Isolation of yeast Kurtzmanomyces sp. I-11, novel producer of mannosylerythritol lipid. Biosci Biotechnol Biochem 66:188–191
Kawakami Y (1953) Surface active agents. Kagaku 23:546–551 (in Japanese)
Kim HS, Jeon JW, Kim BH, Ahn CY, Oh HM, Yoon BD (2006) Extracellular production of a glycolipid biosurfactant, mannosylerythritol lipid, by Candida sp. SY16 using fed-batch fermentation. Appl Microbiol Biotechnol 70:391–396
Kitamoto D, Akiba S, Hioki C, Tabuchi T (1990) Extracellular accumulation of mannosylerythritol lipids by a strain of Candida antarctica. Agric Biol Chem 54:31–36
Kitamoto D, Yanagishita H, Shinbo T, Nakane T, Kamisawa C, Nakahara T (1993) Surface active properties and antimicrobial activities of mannosylerythritol lipids as biosurfactants produced by Candida antarctica. J Biotechnol 29:91–96
Kitamoto D, Yanagishita H, Haraya K, Kitamoto HK (1998) Contribution of a chain-shortening pathway to the biosynthesis of the fatty acids of mannosylerythritol lipid (biosurfactant) in the yeast Candida antarctica: Effect of β-oxidation inhibitors on biosurfactant synthesis. Biotechnol Lett 20:813–818
Kitamoto D, Ghosh S, Ourisson G, Nakatani Y (2000) Formation of giant vesicles from diacylmannosyleryhtritols, and their binding to concanavalin A. Chem Commun 10:861–862
Kitamoto D, Ikegami T, Suzuki T, Sasaki A, Takeyama Y, Idemoto Y, Koura N, Yanagishita H (2001) Microbial conversion of n-alkanes into glycolipid biosurfactants, mannosylerythritol lipids, by Pseudozyma antarctica. Biotechnol Lett 23:1709–1714
Kitamoto D, Isoda H, Nakahara T (2002) Functions and potential applications of glycolipid biosurfactants—from energy-saving materials to gene delivery carriers. J Biosci Bioeng 94:187–201
Konishi M, Morita T, Fukuoka T, Imura T, Kakugawa K, Kitamoto D (2007) Production of different types of mannosylerythritol lipids as biosurfactants by the newly isolated yeast strains belonging to the genus Pseudozyma. Appl Microbiol Biotechnol 75:521–531
Lang S (2002) Biological amphiphiles (microbial biosurfactants). Curr Opin Colloid Interface Sci 7:12–20
Morita T, Konishi M, Fukuoka T, Imura T, Kitamoto D (2006) Discovery of Pseudozyma rugulosa NBRC 10877 as a novel producer of the glycolipid biosurfactants, mannosylerythritol lipids, based on rDNA sequence. Appl Microbiol Biotechnol 73:305–313
Morita T, Konishi M, Fukuoka T, Imura T, Kitamoto D (2007a) Physiological differences in the formation of the glycolipid biosurfactants, mannosylerythritol lipids, between Pseudozyma antarctica and Pseudozyma aphidis. Appl Microbiol Biotechnol 74:307–315
Morita T, Konishi M, Fukuoka T, Imura T, Kitamoto KH, Kitamoto D (2007b) Characterization of the genus Pseudozyma by the formation of glycolipid biosurfactants, mannosylerythritol lipids. FEMS Yeast Res 7:286–292
Nakamura S (1999) Application of sucrose fatty acid esters as food emulsifiers. Spec Publ R Chem Soc 230:73–87
Rau U, Nguyen LA, Schulz S, Wray V, Nimtz M, Roper H, Koch H, Lang S (2005) Formation and analysis of mannosylerythritol lipids secreted by Pseudozyma aphidis. Appl Microbiol Biotechnol 66:551–559
Rodorigues L, Banat IM, Teixeria J, Oliveira R (2006) Biosurfactants: potential applications in medicine. J Antimicrob Chemother 57:609–618
Wakamatsu Y, Zhao X, Jin C, Day N, Shibahara M, Nomura N, Nakahara T, Murata T, Yokoyama KK (2001) Mannosylerythritol lipid induces characteristics of neuronal differentiation in PC12 cells through an ERK-related signal cascade. Eur J Biochem 268:374–383
Zhao X, Murata T, Ohno S, Day N, Song J, Nomura N, Nakahara T, Yokoyama KK (2001) Protein kinase Ca plays a critical role in mannosylerythritol lipid-induced differentiation of melanoma B16 cells. J Biol Chem 276:9903–9910
Acknowledgments
We would like to thank Ms. Akiko Sugimura, a fellow of the Japan Industrial Technology Association, for her technical assistance. This work was supported by the Industrial Technology Research Grant Program in 06A17501c from the New Energy and Industrial Technology Development Organization (NEDO) of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukuoka, T., Morita, T., Konishi, M. et al. Structural characterization and surface-active properties of a new glycolipid biosurfactant, mono-acylated mannosylerythritol lipid, produced from glucose by Pseudozyma antarctica . Appl Microbiol Biotechnol 76, 801–810 (2007). https://doi.org/10.1007/s00253-007-1051-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-007-1051-4