Abstract
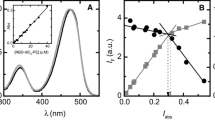
Melittin, an amphiphathic peptide, affects the permeability of vesicles. This can be demonstrated using the dye release technique. Calcein, a fluorescent marker, is trapped in large unilamellar 1-palmitoyl-2-oleoyl-phosphatidylcholine (POPC) vesicles and melittin-induced leakage of the dye can be monitored directly by increasing fluorescence intensity. First, we characterized the effect of increasing cholesterol content in the membrane on melittin-induced leakage and our results reveal that cholesterol inhibits the lytic activity of the peptide. Using intrinsic fluorescence of the single tryptophan of melittin and 2H-NMR of headgroup deuterated phosphatidylcholine, we demonstrated that the affinity of melittin for phosphatidylcholine vesicles is reduced in the presence of cholesterol; this is associated with the tighter lipid packing of the cholesterol-containing bilayer. This reduced binding is responsible for the reduced melittin-induced leakage from cholesterol-containing membranes. The pathway of release was determined to be an all-or-none mechanism. Finally, we investigated the possibility of achieving specific membrane targeting with melittin, when vesicles of different lipid composition are simultaneously present. Melittin incubated together with vesicles made of pure POPC and POPC containing 30(mol)% cholesterol can empty nearly all the cholesterol-free vesicles while the cholesterol-containing vesicles remain almost intact. Owing to the preferential interaction of melittin with the pure POPC vesicles, we were able to achieve controlled release of encapsulated material from a specific vesicle population.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 8 May 1996 / Accepted: 12 September 1996
Rights and permissions
About this article
Cite this article
Benachir, T., Monette, M., Grenier, J. et al. Melittin-induced leakage from phosphatidylcholine vesicles is modulated by cholesterol: a property used for membrane targeting. Eur Biophys J 25, 201–210 (1997). https://doi.org/10.1007/s002490050032
Issue Date:
DOI: https://doi.org/10.1007/s002490050032