Abstract
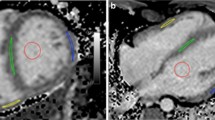
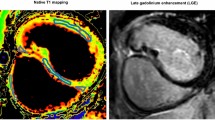
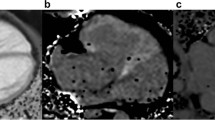
The assumption of the presence of diffuse myocardial fibrosis in long-standing cyanotic congenital heart disease (CHD) inspired us to noninvasively determine the myocardial extracellular volume (ECV) using contrast CMR. T1 maps were measured pre and 10 min after the injection of 0.15 mmol/kg of gadolinium in 25 subjects. Seven patients with long-standing cyanotic CHD and no previous cardiac surgery (aged 16–53 years and oxygen saturations of 69–90%), nine normal subjects (aged 14–49 years), and nine patients with previously cyanotic CHD, who had been corrected by open heart surgery (aged 2 months–58 years, mean 9 years). Late gadolinium enhancement was performed to exclude scar areas. The T1 values were measured in the interventricular septum and in the left lateral or inferior ventricular wall, such that same areas were assessed in every patient in the pre- and post-contrast T1 scan. ECV was calculated according to ΔR1myocardium/ΔR1blood * (1 − hematocrit). Cyanotic patients had significantly lower ECV percentage than the previous cyanotic patients (septum: 22 ± 2.7% vs 35 ± 4.6%, p = 0.002; LV wall: 22 ± 2.2% vs 30 ± 3.7%, p = 0.01, respectively). No significant differences were found between cyanotic patients and normal controls (septum: 22 ± 2.7% vs 24 ± 1.4%, p = 0.44; LV wall: 22 ± 2.2% vs 24 ± 2%, p = 0.57, respectively). Long-standing cyanosis in CHD without cardiac surgery does not cause diffuse myocardial fibrosis or expansion of the myocardial ECV.

Similar content being viewed by others
References
White SK, Sado DM, Flett AS, Moon JC (2012) Characterising the myocardial interstitial space: the clinical relevance of non-invasive imaging. Heart 98:773–779
Riesenkampff E, Messroghli DR, Redington AN, Grosse-Wortmann L (2015) Myocardial t1 mapping in pediatric and congenital heart disease. Circ Cardiovasc Imaging 8:e002504
Hopkins WE, Waggoner AD, Gussak H (1994) Quantitative ultrasonic tissue characterization of myocardium in cyanotic adults with an unrepaired congenital heart defect. Am J Cardiol 74:930–934
Babu-Narayan SV, Kilner PJ, Li W, Moon JC, Goktekin O, Davlouros PA, Khan M, Ho SY, Pennell DJ, Gatzoulis MA (2006) Ventricular fibrosis suggested by cardiovascular magnetic resonance in adults with repaired tetralogy of fallot and its relationship to adverse markers of clinical outcome. Circulation 113:405–413
Triedman JK (2002) Arrhythmias in adults with congenital heart disease. Heart 87:383–389
Hu J, Sun P, Ruan X, Chao A, Lin Y, Li XY (2005) Mechanism of myocardial microvessel formation in cyanotic congenital heart disease. Circ J 69:1089–1093
Kawel N, Nacif M, Zavodni A, Jones J, Liu S, Sibley CT, Bluemke DA (2012) T1 mapping of the myocardium: intra-individual assessment of the effect of field strength, cardiac cycle and variation by myocardial region. J Cardiovasc Magn Reson 14:27
Nacif MS, Turkbey EB, Gai N, Nazarian S, van der Geest RJ, Noureldin RA, Sibley CT, Ugander M, Liu S, Arai AE, Lima JA, Bluemke DA (2011) Myocardial t1 mapping with mri: comparison of look-locker and molli sequences. J Magn Reson Imaging 34:1367–1373
Messroghli DR, Radjenovic A, Kozerke S, Higgins DM, Sivananthan MU, Ridgway JP (2004) Modified look-locker inversion recovery (molli) for high-resolution t1 mapping of the heart. Magn Reson Med 52:141–146
Moon JC, Messroghli DR, Kellman P, Piechnik SK, Robson MD, Ugander M, Gatehouse PD, Arai AE, Friedrich MG, Neubauer S, Schulz-Menger J, Schelbert EB (2013) Myocardial t1 mapping and extracellular volume quantification: a society for cardiovascular magnetic resonance (scmr) and cmr working group of the European society of cardiology consensus statement. J Cardiovasc Magn Reson 15:92
Fratz S, Chung T, Greil GF, Samyn MM, Taylor AM, Valsangiacomo Buechel ER, Yoo SJ, Powell AJ (2013) Guidelines and protocols for cardiovascular magnetic resonance in children and adults with congenital heart disease: Scmr expert consensus group on congenital heart disease. J Cardiovasc Magn Reson 15:51
Allen BS (2004) Pediatric myocardial protection: where do we stand? J Thorac Cardiovasc Surg 128:11–13
Buckberg GD (1995) Update on current techniques of myocardial protection. Ann Thorac Surg 60:805–814
Aiello VD, Binotto MA (2007) Myocardial remodeling in congenital heart disease. Arq Bras Cardiol 88:e185–e186
Friedli B, Haenni B, Moret P, Opie LH (1977) Myocardial metabolism in cyanotic congenital heart disease studied by arteriovenous differences of lactate, phosphate, and potassium at rest and during atrial pacing. Circulation 55:647–652
Fujiwara T, Kurtts T, Anderson W, Heinle J, Mayer JE Jr (1988) Myocardial protection in cyanotic neonatal lambs. J Thorac Cardiovasc Surg 96:700–710
Graham TP Jr, Erath HG Jr, Boucek RJ Jr, Boerth RC (1980) Left ventricular function in cyanotic congenital heart disease. Am J Cardiol 45:1231–1236
Graham TP Jr, Erath HG Jr, Buckspan GS, Fisher RD (1979) Myocardial anaerobic metabolism during isoprenaline infusion in a cyanotic animal model: possible cause of myocardial dysfunction in cyanotic congenital heart disease. Cardiovasc Res 13:401–406
Lupinetti FM, Wareing TH, Huddleston CB, Collins JC, Boucek RJ Jr, Bender HW Jr, Hammon JW Jr (1985) Pathophysiology of chronic cyanosis in a canine model. Functional and metabolic response to global ischemia. J Thorac Cardiovasc Surg 90:291–296
Silverman NA, Kohler J, Levitsky S, Pavel DG, Fang RB, Feinberg H (1984) Chronic hypoxemia depresses global ventricular function and predisposes to the depletion of high-energy phosphates during cardioplegic arrest: implications for surgical repair of cyanotic congenital heart defects. Ann Thorac Surg 37:304–308
Visner MS, Arentzen CE, Ring WS, Anderson RW (1981) Left ventricular dynamic geometry and diastolic mechanics in a model of chronic cyanosis and right ventricular pressure overload. J Thorac Cardiovasc Surg 81:347–357
Broberg CS, Chugh SS, Conklin C, Sahn DJ, Jerosch-Herold M (2010) Quantification of diffuse myocardial fibrosis and its association with myocardial dysfunction in congenital heart disease. Circ Cardiovasc Imaging 3:727–734
Chowdhury UK, Sathia S, Ray R, Singh R, Pradeep KK, Venugopal P (2006) Histopathology of the right ventricular outflow tract and its relationship to clinical outcomes and arrhythmias in patients with tetralogy of fallot. J Thorac Cardiovasc Surg 132:270–277
Xie M, Li Y, Cheng TO, Wang X, Dong N, Nie X, Lu Q, Yang Y, He L, Li L, Ren P (2015) The effect of right ventricular myocardial remodeling on ventricular function as assessed by two-dimensional speckle tracking echocardiography in patients with tetralogy of fallot: a single center experience from china. Int J Cardiol 178:300–307
Broberg CS, Prasad SK, Carr C, Babu-Narayan SV, Dimopoulos K, Gatzoulis MA (2014) Myocardial fibrosis in Eisenmenger syndrome: a descriptive cohort study exploring associations of late gadolinium enhancement with clinical status and survival. J Cardiovasc Magn Reson 16:32
Kozak MF, Redington A, Yoo SJ, Seed M, Greiser A, Grosse-Wortmann L (2014) Diffuse myocardial fibrosis following tetralogy of fallot repair: a t1 mapping cardiac magnetic resonance study. Pediatr Radiol 44:403–409
Baker JE, Contney SJ, Singh R, Kalyanaraman B, Gross GJ, Bosnjak ZJ (2001) Nitric oxide activates the sarcolemmal k(atp) channel in normoxic and chronically hypoxic hearts by a cyclic gmp-dependent mechanism. J Mol Cell Cardiol 33:331–341
Baker JE, Holman P, Kalyanaraman B, Griffith OW, Pritchard KA Jr (1998) Adaptation to chronic hypoxia confers tolerance to subsequent myocardial ischemia by increased nitric oxide production. Ann N Y Acad Sci 874:236–253
Imura H, Caputo M, Parry A, Pawade A, Angelini GD, Suleiman MS (2001) Age-dependent and hypoxia-related differences in myocardial protection during pediatric open heart surgery. Circulation 103:1551–1556
Wen SB, Hu JG, Ma ZX, Zhou XM, Yang YF, Yu FL, Hu MS (2004) [expression of the heat shock protein-70 in the myocardial cells of cyanosis congenital heart diseases]. Zhong nan da xue xue bao. Yi xue ban = Journal of Central South University. Med Sci 29:322–325
Kawel-Boehm N, Maceira A, Valsangiacomo-Buechel ER, Vogel-Claussen J, Turkbey EB, Williams R, Plein S, Tee M, Eng J, Bluemke DA (2015) Normal values for cardiovascular magnetic resonance in adults and children. J Cardiovasc Magn Reson 17:29
Dabir D, Child N, Kalra A, Rogers T, Gebker R, Jabbour A, Plein S, Yu CY, Otton J, Kidambi A, McDiarmid A, Broadbent D, Higgins DM, Schnackenburg B, Foote L, Cummins C, Nagel E, Puntmann VO (2014) Reference values for healthy human myocardium using a t1 mapping methodology: results from the international t1 multicenter cardiovascular magnetic resonance study. J Cardiovasc Magn Reson 16:69
Miller CA, Naish JH, Bishop P, Coutts G, Clark D, Zhao S, Ray SG, Yonan N, Williams SG, Flett AS, Moon JC, Greiser A, Parker GJ, Schmitt M (2013) Comprehensive validation of cardiovascular magnetic resonance techniques for the assessment of myocardial extracellular volume. Circ Cardiovasc Imaging 6:373–383
Acknowledgements
We are very grateful to Sohrab Fratz, MD, PhD, who left us too early, for his scientific enthusiasm and knowledge. He was deeply involved in the scientific discussion and design of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
The study complies with the Declaration of Helsinki, and the protocol complies with the institution’s guidelines.
Informed Consent
A written informed consent was obtained from all patients or the accompanying parents.
Rights and permissions
About this article
Cite this article
Kharabish, A., Meierhofer, C., Hadamitzky, M. et al. Long-Standing Cyanosis in Congenital Heart Disease Does not Cause Diffuse Myocardial Fibrosis. Pediatr Cardiol 39, 105–110 (2018). https://doi.org/10.1007/s00246-017-1734-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00246-017-1734-2