Abstract
Purpose
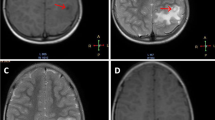
Tuberculoma and neurocysticercosis (NCC) are two of the most common intracranial infections in developing countries and cause of seizures or focal neurological deficit. They often present on neuroimaging as ring-enhancing lesions, and in absence of typical imaging features, can cause significant diagnostic dilemma. Differentiation is extremely important to avoid empirical exposure to anti-tubercular treatment or nonspecific treatment causing disease progression. The aim our study was to evaluate the MR perfusion characteristics of brain tuberculoma and NCC and to assess the potential role of the perfusion in differentiating the two entities.
Methods
Fifty untreated patients (20 tuberculoma and 30 NCC) were prospectively evaluated by using conventional imaging and dynamic susceptibility contrast (DSC) MR perfusion. Relative cerebral blood volume (rCBV) values were calculated from the wall and core of the lesion and were compared with contralateral normal white matter.
Results
rCBV values from the wall of the lesion were significantly different in tuberculoma and NCC (P < 0.05). Tuberculomas showed high mean rCBV values than NCC (3.3 and 1.3 respectively). The core of these lesions showed lower rCBV values in both the lesions which were lower than normal white matter. A cutoff value of rCBV from the wall of the lesion 1.965 for tuberculoma showed 90% sensitivity and 100% specificity.
Conclusion
Perfusion MRI in combination with conventional sequences can better characterize and differentiate similar-appearing tuberculoma and NCC and may be incorporated in routine protocol which may help in avoiding brain biopsy and empirical therapy.






Similar content being viewed by others
References
Garg RK, Desai P, Kar M, Kar AM (2008) Multiple ring enhancing brain lesions on computed tomography: an Indian perspective. J Neurol Sci 266:92–96
Singhi P (2011) Infectious causes of seizures and epilepsy in the developing world. Dev Med Child Neurol 53:600–609
Amudhan S, Gururaj G, Satishchandra P (2015) Epilepsy in India I: epidemiology and public health. Ann Indian Acad Neurol 18:263–277
Singhi P, Singhi S (2009) Neurocysticercosis in children. Indian J Pediatr 76:537–545
Garg RK (2004) Diagnostic criteria for neurocysticercosis: some modifications are needed for Indian patients. Neurol India 52:171–177
Rajshekhar V (1991) Etiology and management of single small CT lesions in patients with seizures: understanding a controversy. Acta Neurol Scand 84:465–470
Bagga A, Kalra V, Ghai OP (1988) Intracranial tuberculoma. Evaluation and treatment. Clin Pediatr 27:487–490
Regional Office for South-East Asia, World Health Organization (2017). Bending the curve - ending TB: Annual report 2017. WHO Regional Office for South-East Asia. http://www.who.int/iris/handle/10665/254762. Accessed 12 Feb 2018
Geneva. Word Health Organization; 2009. Global Tuberculosis Control 2009 Surveillance Planning Financing. WHO/HTM/TB/2009411
Ahuja GK, Mohan KK, Prasad K, Behari M (1994) Diagnostic criteria for tuberculous meningitis and their validation. Tuber Lung Dis 75:149–152
Jinkins JR, Gupta R, Chang KH, Carbajal JR (1995) MR imaging of central nervous system tuberculosis. Radiol Clin N Am 33:771–786
Kumar R, Shalimar BV, Khanal S, Sreenivas V, Gupta SD et al (2010) Antituberculosis therapy-induced acute liver failure: magnitude, profile, prognosis, and predictors of outcome. J Hepatol 51:443–444
Del Brutto OH, Nash TE, White AC Jr, Rajshekhar V, Wilkins PP, Singh G et al (2017) Revised diagnostic criteria for neurocysticercosis. J Neurol Sci 372:202–210
Sankhe S, Baheti A, Ihare A, Mathur S, Dabhade P, Sarode A (2013) Perfusion magnetic resonance imaging characteristics of intracerebral tuberculomas and its role in differentiating tuberculomas from metastases. Acta Radiol 54:307–312
Chu X-l, Zhao L-p, Ma J-x et al (2015) Application of diffusion tensor imaging in AIDS patients with brain opportunistic diseases: a comparative study of tuberculosis and toxoplasmosis. Radiol Infect Dis 2:11–15
Chatterjee S, Saini J, Kesavadas C, Arvinda HR, Jolappara M, Gupta AK (2010) Differentiation of tubercular infection and metastasis presenting as ring enhancing lesion by diffusion and perfusion magnetic resonance imaging. J Neuroradiol 37:167–171
Pretell EJ, Martinot C Jr, Garcia HH, Alvarado M, Bustos JA, Martinot C (2005) Differential diagnosis between cerebral tuberculosis and neurocysticercosis by magnetic resonance spectroscopy. J Comput Assist Tomo 29:112–114
Jayasundar R, Singh VP, Raghunathan P, Jain K, Banerji AK (1999) Inflammatory granulomas: evaluation with proton MRS. NMR Biomed 12:139–144
Ezaki T, Baluk P, Thurston G et al (2001) Time course of endothelial cell proliferation and microvascular remodeling in chronic inflammation. Am J Pathol 58:2043–2055
Gupta RK, Haris M, Husain N, Husain M, Prasad KN, Pauliah M, Srivastava C, Tripathi M, Rastogi M, Behari S, Singh A, Rathore D, Rathore RKS (2007) Relative cerebral blood volume is a measure of angiogenesis in brain tuberculoma. J Comput Assist Tomo 31:335–336
Dastur DK, Dave UP (1977) Ultrastructural basis of the vasculopathy in and around brain tuberculosis: possible significance of altered basement membrane. Am J Pathol 89:35–50
Misra UK, Kalita J, Singh AP, Prasad S (2012) Vascular endothelial growth factor in tuberculous meningitis. Int J Neurosci 123(2):128–132
Alvarez JI, Colegial CH, Castano CA, Trujillo J, Teale JM, Restrepo BI (2002) The human nervous tissue in proximity to granulomatous lesions induced by Taenia solium metacestodes displays an active response. J Neuroimmunol 127(1):139–144
Restrepo BI, Alvarez JI, Castano JA, Arias LF, Restrepo M, Trujillo J, Colegial CH, Teale JM (2001) Brain granulomas in neurocysticercosis patients are associated with a Th1 and Th2 profile. Tuomanen EI, editor Infect Immun 69(7):4554–60
Tuero I, Palma S, Cabeza F, Saleemi S, Rodriguez S, Gonzales I, Mayta H, Mahanty S, Garcia HH, Gilman RH, Cysticercosis Working Group in Perú (2015) A comparative study of peripheral immune responses to Taenia solium in individuals with parenchymal and subarachnoid neurocysticercosis. Brehm K, ed PLoS Negl Trop Dis 9(10): e0004143
Awasthi R, Verma A, Paliwal VK, Verma SK, Prasad KN, Rathore RK, Gupta RK (2012) Dynamic contrast enhanced (DCE) perfusion indices as a measure of blood brain barrier (BBB) disruption in different stages of neurocysticercosis. Magn Reson Imaging 30:104–111
Floriano VH, Torres US, Spotti AR, Ferraz-Filho JRL, Tognola WA (2013) the role of dynamic susceptibility contrast-enhanced perfusion MR imaging in differentiating between infectious and neoplastic focal brain lesions: Results from a Cohort of 100 consecutive patients. Harel N, editor PLoS ONE 2013; 8(12):e81509
Batra A, Tripathi RP (2003) Perfusion magnetic resonance imaging in intracerebral parenchymal tuberculosis: preliminary findings. J Comput Assist Tomo 27:882–888
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Ghosh, R.N., Vyas, S., Singh, P. et al. Perfusion magnetic resonance imaging in differentiation of neurocysticercosis and tuberculoma. Neuroradiology 61, 257–263 (2019). https://doi.org/10.1007/s00234-018-2118-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-018-2118-x