Abstract
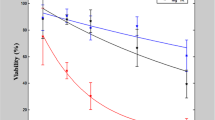
Electrotransfection and electrofusion, both widely used in research and medical applications, still have to face a range of problems, including the existence of electroporation-resistant cell types, cell mortality and also great batch-to-batch variations of the transfection and fusion yields. In the present study, a systematic analysis of the parameters critical for the efficiency and robustness of electromanipulation protocols was performed on five mammalian cell types. Factors examined included the sugar composition of hypotonic pulse media (trehalose, sorbitol or inositol), the kinetics of cell volume changes prior to electropulsing, as well as the growth medium additives used for post-pulse cell cultivation. Whereas the disaccharide trehalose generally allowed regulatory volume decrease (RVD), the monomeric sugar alcohols sorbitol and inositol inhibited RVD or even induced secondary swelling. The different volume responses could be explained by the sugar selectivity of volume-sensitive channels (VSC) in the plasma membrane of all tested cell types. Based on the volumetric data, highest transfection and fusion yields were mostly achieved when the target cells were exposed to hypotonicity for about 2 min prior to electropulsing. Longer hypotonic treatment (10–20 min) decreased the yields of viable transfected and hybrid cells due to (1) the cell size reduction upon RVD (trehalose) or (2) the excessive losses of cytosolic electrolytes through VSC (inositol/sorbitol). Doping the plasma membrane with lipophilic anions prevented both cell shrinkage and ion losses (probably due to VSC inhibition), which in turn resulted in increased transfection and fusion efficiencies.






Similar content being viewed by others
References
Antonenko Y.N., Rokitskaya T.I., Kotova E.A. 1999. Effect of dipole modifiers on the kinetics of sensitized photoinactivation of gramicidin channels in bilayer lipid membranes. Membr. Cell Biol. 13:111–120
Barrau C., Teissie J., Gabriel B. 2004. Osmotically induced membrane tension facilitates the triggering of living cell electropermeabilization. Bioelectrochem. 63:327–332
Braet K., Mabilde C., Cabooter L., Rapp G., Leybaert L. 2004. Electroporation loading and photoactivation of caged InsP(3): tools to investigate the relation between cellular ATP release in response to intracellular InsP(3) elevation. J. Neurosci. Meth. 132:81–89
Cegovnik U., Novakovic S. 2004. Setting optimal parameters for in vitro electrotransfection of B16F1, SA1, LPB, SCK, L929 and CHO cells using predefined exponentially decaying electric pulses. Bioelectrochem. 62:73–82
Cseh R., Benz R. 1998. The adsorption of phloretin to lipid monolayers and bilayers cannot be explained by Langmuir adsorption isotherms alone. Biophys J. 74:1399–1408
Friedrich U., Stachowicz N., Simm A., Fuhr G., Lukas K., Zimmermann U. 1998. High efficiency electrotransfection with aluminum electrodes using microsecond controlled pulses. Bioelectrochem. Bioenerg. 47:103–111
Fuhr G., Zimmermann U., Shirley S.G. 1996. Cell motion in time-varying fields: Principles and potential. In: Electromanipulation of Cells. Zimmermann U., Neil G. (eds). CRC, Boca Raton, FL pp. 259–328
Fürst J., Gschwentner M., Ritter M., Bottà G., Jakab ML, Mayer M., Garavaglia L., Bazzini C., Rodighiero S., Meyer G., Eichmüller S., Wöll E., Paulmichl M. 2002. Molecular and functional aspects of anionic channels activated during regulatory volume decrease in mammalian cells. Pfluegers Arch. -Eur. J. Physiol. 444:1–25
Gaynor P., Wells D.N., Oback B. 2005. Couplet alignment and improved electrofusion by dielectrophoresis for a zona-free high-throughput cloned embryo production system. Med. Biol. Eng. Comput. 43:150–154
Grund E.M., Muise-Helmericks R.C. 2005. Cost efficient and effective gene transfer into the human natural killer cell line, NK92. J. Immun. Meth. 296:31–36
Hertel C., Terzi E., Hauser N., Jakob-Rotne R., Seelig J., Kemp J.A. 1997. Inhibition of the electrostatic interaction between beta-amyloid peptide and membranes prevents beta-amyloid-induced toxicity. Proc. Natl. Acad. Sci. USA 94:9412–9416
Hojo S., Shimizu K., Yositake H., Muraji M., Tsujimoto H., Tatebe W. 2003. The relationship between electropermeabilization and cell cycle and cell size of Saccharomyces cerevisiae. IEEE Tram. Nanobiosci. 2:35–39
Isambert H. 1998. Understanding the electroporation of cells and artificial bilayer membranes. Phys. Rev. Lett. 80:3404–3407
Jackson P.S., Strange K. 1993. Volume-sensitive anion channels mediate swelling-activated inositol and taurine efflux. Am. J. Physiol. 265:C1489–C1500
Jones T.B. 1995. Electromechanics of Particles. Cambridge University Press, New York
Kürschner M., Nielsen K., Andersen C., Sukhorukov V.L., Schenk W.A., Benz R., Zimmermann U. 1998. Interaction of lipophilic ions with the plasma membrane of mammalian cells studied by electrorotation. Biophys. J. 74:3031–3043
Lang F. 1998. Cell Volume Regulation, Karger, Basel
Li S.L. 2004. Electroporation gene therapy: New developments in vivo and in vitro. Curr. Gene Ther. 4:309–316
Lynch P.T., Davey M.R. 1996. Electrical Manipulation of Cells, Chapman & Hall, NY
Muller K.J., Sukhorukov V.L., Zimmermann U. 2001. Reversible electropermeabilization of mammalian cells by high-intensity, ultra-short pulses of submicrosecond duration. J. Membrane Biol. 184:161–170
Mussauer H., Sukhorukov V.L., Zimmermann U. 2001. Trehalose improves survival of electrotransfected mammalian cells. Cytometry 45:161–169
Okada Y. 1998. Cell Volume Regulation. The Molecular Mechanism and Volume Sensing Machinery, Elsevier, Amsterdam
Pucihar G., Kotnik T., Kanduser M., Miklavcic D. 2001. The influence of medium conductivity on electropermeabilization and survival of cells in vitro. Bioelectrochem. 54:107–115
Reuss R., Ludwig J., Shirakashi R., Ehrhart F., Zimmermann H., Schneider S., Weber M.M., Zimmermann U., Schneider H., Sukhorukov V.L. 2004. Intracellular delivery of carbohydrates into mammalian cells through swelling-activated pathways. J. Membrane. Biol. 200:67–81
Schmidt E., Leinfelder U., Gessner P., Zillikens D., Brocker E.B., Zimmermann U. 2001. CD19+ B lymphocytes are the major source of human antibody-secreting hybridomas generated by electrofusion. J. Immunol. Methods 255:93–102
Schmitt J.J., Zimmermann U. 1989. Enhanced hybridoma production by electrofusion in strongly hypo-osmolar solutions. Biochim. Biophys. Acta 983:42–50
Shimizu K., Kuriyama H., Kjaergaard J., Lee W., Tanaka H., Shu S. 2004. Comparative analysis of antigen loading strategies of dendritic cells for tumor immunotherapy. J. Immunother. 27:265–272
Shirakashi R., Köstner C.M., Müller K.J., Kürschner M., Zimmermann U., Sukhorukov V.L. 2002. Intracellular delivery of trehalose into mammalian cells by electropermeabilization. J. Membrane Biol. 189:45–54
Smejtek P., Wang S. 1991. Domains and anomalous adsorption-isotherms of dipalmitoylphosphatidylcholine membranes and lipophilic ions — pentachlorophenolate, tetraphenylborate, and dipicrylamine. Biophys. J. 59:1064–1073
Sukhorukov V.L., Zimmermann U. 1996. Electrorotation of erythrocytes treated with dipicrylamine: mobile charges within the membrane show their “signature” in rotational spectra. J. Membrane Biol. 153:161–169
Sukhorukov V.L., Kürschner M., Dilsky S., Lisec T., Wagner B., Schenk W.A., Benz R., Zimmermann U. 2001. Phloretin-induced changes of lipophilic ion transport across the plasma membrane of mammalian cells. Biophys. J. 81:1006–1013
Sukhorukov V.L., Mussauer H., Zimmermann U. 1998. The effect of electrical deformation forces on the electropermeabilization of erythrocyte membranes in low- and high- conductivity media. J. Membrane Biol. 163:235–245
Weaver J.C. 2003. Electroporation of biological membranes from multicellular to nano scales. IEEE Trans. Diel. Electr. Insul. 10:754–768
Weise J.B., Maune S., Gorogh T., Kabelitz D., Arnold N., Pfisterer J., Hilpert F., Heiser A. 2004. A dendritic cell based hybrid cell vaccine generated by electrofusion for immunotherapy strategies in HNSCC. Auris Nasus Larynx. 31:149–153
Zarnitsyn V.G., Prausnitz A.R., Chizmadzhev Y.A. 2004. Physical methods of nucleic acid delivery into cells and tissues. Biol. Membrany 21:355–373
Zimmermann U., Friedrich U., Mussauer H., Gessner P., Hämel K., Sukhorukov V.L. 2000. Electromanipulation of mammalian cells: fundamentals and application. IEEE Trans. Plasma Sci. 28:72–82
Zimmermann U., Gessner P., Schnettler R., Perkins S., Foung S.K.H. 1990. Efficient hybridization of mouse-human cell lines by means of hypo-osmolar electrofusion. J. Immunolog. Meth. 134:43–50
Zimmermann U., Neil G.A. 1996. Electromanipulation of Cells, CRC, Boca Raton, FL
Zimmermann U., Pilwat G., Riemann F. 1974. Reversible dielectric breakdown of cell membranes by electrostatic fields. Z. Naturforsch. 29c:304–305
Acknowledgment
This work was supported by grants from the Deutsche Forschungsgemeinschaft to U.Z. and V.L.S (Zi 99/12), and to W.A.S. and V.L.S. (SCHE209/17).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sukhorukov, V., Reuss, R., Zimmermann, D. et al. Surviving High-Intensity Field Pulses: Strategies for Improving Robustness and Performance of Electrotransfection and Electrofusion. J Membrane Biol 206, 187–201 (2005). https://doi.org/10.1007/s00232-005-0791-2
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s00232-005-0791-2