Abstract
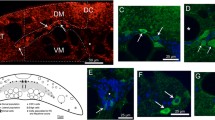
We demonstrated previously that the hypothalamic supraoptic nucleus (SON) undergoes an axonal sprouting response following a unilateral lesion of the hypothalamo-neurohypophysial tract in a 35-day-old rat to repopulate the partially denervated neural lobe (NL). However, no sprouting occurs following the same injury in a 125-day-old rat. We previously reported a significant increase in Thy-1 protein in the SON of a 125-day-old rat compared to a 35-day-old rat in the absence of injury. Thy-1 is a cell surface glycoprotein shown to inhibit axonal outgrowth following injury; however, we did not look at axotomy’s effect on Thy-1 in the SON. Therefore, we sought to determine the integrin ligands that bind Thy-1 in the SON and how axotomy impacts Thy-1. Like what others have shown, the co-immunoprecipitation analysis demonstrated that Thy-1 interacts with αvß3 and αvß5 integrin dimers in the SON. We used western blot analysis to examine protein levels of Thy-1 and integrin subunits following injury in the 35- and 125-day-old rat SON and NL. Our results demonstrated that Thy-1 protein levels increase in the lesion SON in a 35-day-old rat. The quantitative dual-fluorescent analysis showed that the increase in Thy-1 in the lesion SON occurred in astrocytes. There was no change in Thy-1 or integrin protein levels following injury in the 125-day-old following injury. Furthermore, the axotomy significantly decreased Thy-1 protein levels in the NL of both 35- and 125-day-old rats. These results provide evidence that Thy-1 protein levels are injury dependent in the magnocellular neurosecretory system.









Similar content being viewed by others
Data availability
The datasets generated during the current study are available from the corresponding author upon reasonable request.
References
Askvig JM, Dalzell TS, Toumeh N et al (2020) Age-dependent increase in Thy-1 protein in the rat supraoptic nucleus. Heliyon 6:e03501. https://doi.org/10.1016/j.heliyon.2020.e03501
Askvig JM, Leiphon LJ, Watt JA (2012) Neuronal activity and axonal sprouting differentially regulate CNTF and CNTF receptor complex in the rat supraoptic nucleus. Exp Neurol 233:243
Askvig JM, Watt JA (2015) The MAPK and PI3K pathways mediate CNTF-induced neuronal survival and process outgrowth in hypothalamic organotypic cultures. J Cell Commun Signal 9:217
Askvig JM, Watt JA (2019) Absence of axonal sprouting following unilateral lesion in 125-day-old rat supraoptic nucleus may be due to age-dependent decrease in protein levels of ciliary neurotrophic factor receptor alpha. J Comp Neurol 527:2291–2301. https://doi.org/10.1002/cne.24675
Avalos AM, Arthur WT, Schneider P, Quest AF, Burridge K, Leyton L (2004) Aggregation of integrins and RhoA activation are required for Thy-1-induced morphological changes in astrocytes. J Biol Chem 279:39139–39145. https://doi.org/10.1074/jbc.M403439200
Avalos AM, Labra CV, Quest AF, Leyton L (2002) Signaling triggered by Thy-1 interaction with beta 3 integrin on astrocytes is an essential step towards unraveling neuronal Thy-1 function. Biol Res 35:231–238. https://doi.org/10.4067/s0716-97602002000200015
Avalos AM, Valdivia AD, Munoz N et al (2009) Neuronal Thy-1 induces astrocyte adhesion by engaging syndecan-4 in a cooperative interaction with alphavbeta3 integrin that activates PKCalpha and RhoA. J Cell Sci 122:3462
Brown RH, Schweitzer J, Dichter MA (1984) Expression of the Thy-1 antigen in long-term cultures of embryonic mouse spinal cord. Brain Res 296:87–91. https://doi.org/10.1016/0006-8993(84)90513-4
Chen CH, Wang SM, Yang SH, Jeng CJ (2005) Role of Thy-1 in in vivo and in vitro neural development and regeneration of dorsal root ganglionic neurons. J Cell Biochem 94:684
Deacon RM, Rawlins JN (1996) Equithesin without chloral hydrate as an anaesthetic for rats. Psychopharmacology 124:288–290
Dellmann HD, Carithers J (1992) Development of neural lobe-like neurovascular contact regions after intrahypothalamic transection of the hypothalamo-neurohypophysial tract. Brain Res 585:19–27
Dohanics J, Hoffman GE, Smith MS, Verbalis JG (1992) Functional neurolobectomy induced by controlled compression of the pituitary stalk. Brain Res 575:215–222
Dohanics J, Hoffman GE, Verbalis JG (1996) Chronic hyponatremia reduces survival of magnocellular vasopressin and oxytocin neurons after axonal injury. J Neurosci 16:2373
Hermosilla T, Munoz D, Herrera-Molina R et al (2008) Direct Thy-1/alphaVbeta3 integrin interaction mediates neuron to astrocyte communication. Biochem Biophys Acta 1783:1111
Jaskolski F, Mulle C, Manzoni OJ (2005) An automated method to quantify and visualize colocalized fluorescent signals. J Neurosci Methods 146:42–49. https://doi.org/10.1016/j.jneumeth.2005.01.012
Kang SS, Keasey MP, Cai J, Hagg T (2012) Loss of neuron-astroglial interaction rapidly induces protective CNTF expression after stroke in mice. J Neurosci 32:9277–9287. https://doi.org/10.1523/JNEUROSCI.1746-12.2012
Keasey MP, Kang SS, Lovins C, Hagg T (2013) Inhibition of a novel specific neuroglial integrin signaling pathway increases STAT3-mediated CNTF expression. Cell Commun Signal 11:35
Kemshead JT, Ritter MA, Cotmore SF, Greaves MF (1982) Human Thy-1: expression on the cell surface of neuronal and glial cells. Brain Res 236:451–461. https://doi.org/10.1016/0006-8993(82)90727-2
Kiernan JA (1971) Pituicytes and the regenerative properties of neurosecretory and other axons in the rat. J Anat 109:97–114
Kukulansky T, Abramovitch S, Hollander N (1999) Cleavage of the glycosylphosphatidylinositol anchor affects the reactivity of thy-1 with antibodies. J Immunol 162:5993–5997
Leyton L, Díaz J, Martínez S, Palacios E, Pérez LA, Pérez RD (2019) Thy-1/CD90 a bidirectional and lateral signaling scaffold. Front Cell Dev Biol 7:132. https://doi.org/10.3389/fcell.2019.00132
Leyton L, Schneider P, Labra CV, Ruegg C, Hetz CA, Quest AF, Bron C (2001) Thy-1 binds to integrin beta(3) on astrocytes and triggers formation of focal contact sites. Curr Biol 11:1028
McLean IW, Nakane PK (1974) Periodate-lysine-paraformaldehyde fixative. A new fixation for immunoelectron microscopy. J Histochem Cytochem 22:1077
Miyata S, Takamatsu H, Maekawa S, Matsumoto N, Watanabe K, Kiyohara T, Hatton GI (2001) Plasticity of neurohypophysial terminals with increased hormonal release during dehydration: ultrastructural and biochemical analyses. J Comp Neurol 434:413
Moll J (1957) Regeneration of the supraoptico-hypophyseal and paraventriculo-hypophyseal tracts in the hypophysectomized rat. Z Zellforsch Mikrosk Anat 46:686–709
Morris R (1992) Thy-1, the enigmatic extrovert on the neuronal surface. BioEssays 14:715–722. https://doi.org/10.1002/bies.950141014
Raisman G (1973) Electron microscopic studies of the development of new neurohaemal contacts in the median eminence of the rat after hypophysectomy. Brain Res 55:245–261. https://doi.org/10.1016/0006-8993(73)90294-1
Reif AE, Allen JM (1964) The AKR thymic antigen and its distribution in leukemias and nervous tissues. J Exp Med 120:413–433. https://doi.org/10.1084/jem.120.3.413
Shahar T, House SB, Gainer H (2004) Neural activity protects hypothalamic magnocellular neurons against axotomy-induced programmed cell death. J Neurosci 24:6553
Silverman AJ, Zimmerman EA (1982) Adrenalectomy increases sprouting in a peptidergic neurosecretory system. Neuroscience 7:2705–2714. https://doi.org/10.1016/0306-4522(82)90094-X
Sofroniew MV (2005) Reactive astrocytes in neural repair and protection. Neuroscientist 11:400. https://doi.org/10.1177/1073858405278321
Tiveron MC, Barboni E, Pliego Rivero FB, Gormley AM, Seeley PJ, Grosveld F, Morris R (1992) Selective inhibition of neurite outgrowth on mature astrocytes by Thy-1 glycoprotein. Nature 355:745
Watt JA, Moffet CW, Zhou X, Short S, Herman JP, Paden CM (1999) Central peptidergic neurons are hyperactive during collateral sprouting and inhibition of activity suppresses sprouting. J Neurosci 19:1586–1598
Watt JA, Paden CM (1991) Compensatory sprouting of uninjured magnocellular neurosecretory axons in the rat neural lobe following unilateral hypothalamic lesion. Exp Neurol 111:9–24. https://doi.org/10.1016/0014-4886(91)90046-F
Whiteman ST, Askvig JM (2021) Astrocytic role of Thy-1 induced inhibition of axonal sprouting. Neural Regen Res 16:1192–1193. https://doi.org/10.4103/1673-5374.300429
Xue GP, Rivero BP, Morris RJ (1991) The surface glycoprotein Thy-1 is excluded from growing axons during development: a study of the expression of Thy-1 during axogenesis in hippocampus and hindbrain. Development 112:161–176
Zhou Y, Hagood JS, Lu B, Merryman WD, Murphy-Ullrich JE (2010) Thy-1-integrin alphav beta5 interactions inhibit lung fibroblast contraction-induced latent transforming growth factor-beta1 activation and myofibroblast differentiation. J Biol Chem 285:22382
Acknowledgements
The authors thank Dr. John Watt at the University of North Dakota for using the Olympus microscope. This material is based upon work supported by the National Science Foundation under Grant No. 1643998, anonymous donors that have financially supported research in the biology department, and the Office of URSCA (Undergraduate Research, Scholarship, and Creative Activity) at Concordia College.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial or nonfinancial interests directly or indirectly related to the work submitted for this publication.
Additional information
Communicated by Sreedharan Sajikumar.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Askvig, J.M., Irmen, R.E., Dalzell, T.S. et al. Axotomy results in an increase in Thy-1 protein in the 35-day-old rat supraoptic nucleus. Exp Brain Res 241, 851–864 (2023). https://doi.org/10.1007/s00221-023-06562-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-023-06562-4