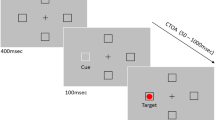
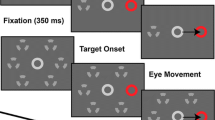
Abstract
The relationship between the latencies of saccadic eye movements and the number of response alternatives is complex. Previously, we have found a decrease in exogenous saccade latencies with an increase in the number of response alternatives (i.e., an anti-Hick’s effect). In the present study, we examined the effect of bottom up and top down influences on the effect. In Experiment 1, we found that the anti-Hick’s effect is dependent upon the number, as opposed to the configuration, of response alternatives, suggesting that the effect is not a purely bottom up phenomenon. In Experiment 2, we examined whether top down processes, such as expectancy, influence the magnitude and/or direction of the relationship between response alternatives and reaction time. We found that interleaving, as opposed to blocking, the number of alternatives negated the anti-Hick’s effect. Taken together, these results suggest that the anti-Hick’s effect arises not only from stimulus-based perceptual signals but also from higher-order control signals.




Similar content being viewed by others
References
Anliker J (1976) Eye movements: on-line measurement, analysis, and control. In: Monty RA, Senders JW (eds) Eye movements and psychological processes. Lawrence Erlbaum, New Jersey, pp 185–202
Bahill AT, Clark MR, Stark L (1975) The main sequence: a tool for studying human eye movements. Math Biosci 24:191–204
Ballanger B (2009) Top-down control of saccades as part of a generalized model of proactive inhibitory control. J Neurophysiol 102(5):2578–2580. doi:10.1152/jn.00717.2009
Basso MA, Wurtz RH (1997) Modulation of neuronal activity by target uncertainty. Nature 389(6646):66–69. doi:10.1038/37975
Basso MA, Wurtz RH (1998) Modulation of neuronal activity in superior colliculus by changes in target probability. J Neurosci 18(18):7519–7534
Boulinguez P, Ballanger B, Granjon L, Benraiss A (2009) The paradoxical effect of warning on reaction time: demonstrating proactive response inhibition with event-related potentials. Clin Neurophysiol 120(4):730–737. doi:10.1016/j.clinph.2009.02.167
Dyckman KA, Camchong J, Clementz BA, McDowell JE (2007) An effect of context on saccade-related behavior and brain activity. Neuroimage 36(3):774–784. doi:10.1016/j.neuroimage.2007.03.023
Ethridge LE, Brahmbhatt S, Gao Y, McDowell JE, Clementz BA (2009) Consider the context: blocked versus interleaved presentation of antisaccade trials. Psychophysiology 46(5):1100–1107. doi:10.1111/j.1469-8986.2009.00834.x
Fecteau JH, Munoz DP (2003) Exploring the consequences of the previous trial. Nat Rev Neurosci 4(6):435–443. doi:10.1038/Nrn1114
Heywood S, Churcher J (1980) Structure of the visual array and saccadic latency: implications for oculomotor control. Q J Exp Psychol 32(2):335–341
Hick WE (1952) On the rate of gain of information. Q J Exp Psychol 4:11–26
Hyman R (1953) Stimulus information as a determinant of reaction time. J Exp Psychol 45:188–196
Jaffard M, Benraiss A, Longcamp M, Velay JL, Boulinguez P (2007) Cueing method biases in visual detection studies. Brain Res 1179:106–118. doi:10.1016/j.brainres.2007.08.032
Jaffard M, Longcamp M, Velay JL, Anton JL, Roth M, Nazarian B, Boulinguez P (2008) Proactive inhibitory control of movement assessed by event-related fMRI. Neuroimage 42(3):1196–1206. doi:10.1016/j.neuroimage.2008.05.041
Kveraga K, Hughes HC (2005) Effects of stimulus-response uncertainty on saccades to near-threshold targets. Exp Brain Res 162(3):401–405. doi:10.1007/s00221-004-2155-z
Kveraga K, Boucher L, Hughes HC (2002) Saccades operate in violation of Hick’s law. Exp Brain Res 146(3):307–314. doi:10.1007/s00221-002-1168-8
Lawrence BM (2010) An anti-Hick’s effect for exogenous, but not endogenous, saccadic eye movements. Exp Brain Res 204(1):115–118. doi:10.1007/s00221-010-2301-8
Lawrence BM, Gardella AL (2009) Saccades and reaches, behaving differently. Exp Brain Res 195(3):413–418. doi:10.1007/s00221-009-1804-7
Lawrence BM, St John A, Abrams RA, Snyder LH (2008) An anti-Hick’s effect in monkey and human saccade reaction times. J Vis 8(3):21–27. doi:10.1167/8.3.26
Lee KM, Keller EL, Heinen SJ (2005) Properties of saccades generated as a choice response. Exp Brain Res 162(3):278–286. doi:10.1007/s00221-004-2186-5
Lo CC, Boucher L, Pare M, Schall JD, Wang XJ (2009) Proactive inhibitory control and attractor dynamics in countermanding action: a spiking neural circuit model. J Neurosci 29(28):9059–9071. doi:10.1523/JNEUROSCI.6164-08.2009
Marino RA, Munoz DP (2009) The effects of bottom-up target luminance and top-down spatial target predictability on saccadic reaction times. Exp Brain Res 197(4):321–335. doi:10.1007/s00221-009-1919-x
Pare M, Munoz DP (1996) Saccadic reaction time in the monkey: advanced preparation of oculomotor programs is primarily responsible for express saccade occurrence. J Neurophysiol 76(6):3666–3681
Posner MI, Cohen Y (1984) Components of visual orienting. In: Bouma H, Bouwhuis DG (eds) Attention and performance X: control of language processes. Erlbaum, Hillsdale, pp 531–556
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lawrence, B.M., Weaver, J.S. Manipulations of the relationship between response alternatives and exogenous saccade latencies. Exp Brain Res 214, 241–247 (2011). https://doi.org/10.1007/s00221-011-2824-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-011-2824-7