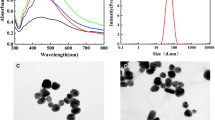
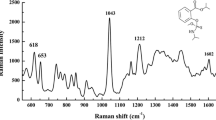
Abstract
Surface-enhanced Raman spectroscopy (SERS) has the potential to detect pesticide residues in agricultural products. However, some systemic pesticides, such as chlorpyrifos, can enter the plant tissue, and not just stay on the surface. Consequently, many SERS studies halted at practical application because of its complexity. In this work, SERS technology was used to detect chlorpyrifos residues in tea products at the semiquantitative level. A simple pretreatment method effectively avoided interference of other fluorescent substances, and all major peaks could be distinguished on the basis of a novel substrate. A principal component analysis algorithm was applied to form a regression model, and a nanogram detection limit was obtained. Furthermore, chlorpyrifos residues in the same tea products were also measured by gas chromatography–mass spectrometry, and the results show a small range of errors. From the comparative study of the two detection methods, the results suggest the great promise of SERS technology for rapid inspection of agricultural products.






Similar content being viewed by others
References
Zhang W, Li X, Hua F, Chen W, Wang W, Chu GX, et al. Interaction between ester-type tea catechins and neutrophil gelatinase-associated lipocalin: inhibitory mechanism. J. Agric. Food Chem. 2018;66(5):1147–56. https://doi.org/10.1021/acs.jafc.7b05399.
Yang X, Xu DC, Qiu JW, Ma Y, Wang J. Simultaneous determination of 118 pesticide residues in Chinese teas by gas chromatography-mass spectrometry. In: Central theme, technology for all: sharing the knowledge for development. Proceedings of the International Conference of Agricultural Engineering, Xxxvii Brazilian Congress of Agricultural Engineering, International Livestock Environment Symposium - Iles Viii, Iguassu Falls City, Brazil, August To September, 2008. pp 39-46
Hou R, Zhang Z, Pang S, Yang T, Clark JM, He L. Alteration of the nonsystemic behavior of the pesticide ferbam on tea leaves by engineered gold nanoparticles. Environ. Sci. Technol. 2016;50(12):6216–23. https://doi.org/10.1021/acs.est.6b01336.
Lee WJ, Hoppin JA, Rusiecki JA, Kamel F, Blair A, Sandler DP. Mortality among pesticide applicators exposed to chlorpyrifos in the Agricultural Health Study. Environ. Health Perspect. 2007;115(4):528–34.
Flaskos J. The developmental neurotoxicity of organophosphorus insecticides: a direct role for the oxon metabolites. Toxicol. Lett. 2012;209(1):86–93.
Galloway T, Handy R. Immunotoxicity of organophosphorous pesticides. Ecotoxicology. 2003;12(1-4):345–63.
Farag AT, Radwan AH, Sorour F, Okazy AE, El-Agamy ES, El-Sebae EK. Chlorpyrifos induced reproductive toxicity in male mice. Reprod. Toxicol. 2010;29(1):80–5.
Salic A, Mitchison TJ. A chemical method for fast and sensitive detection of DNA synthesis in vivo. Proc. Natl. Acad. Sci. U. S. A. 2008;105(7):2415–20.
Ghalwa NA, Abushawish HM, Hamada M, Hartani K, Basheer AAH. Studies on degradation of diquat pesticide in aqueous solutions using electrochemical method. Am. J. Anal. Chem. 2012;3(2):99–105.
Wang J, Cheung W, Leung D. Determination of pesticide residue transfer rates (percent) from dried tea leaves to brewed tea. J. Agric. Food Chem. 2014;62(4):966–83. https://doi.org/10.1021/jf404123h.
Zhu P, Miao H, Du J, Zou JH, Zhang GW, Zhao YF, et al. Organochlorine pesticides and pyrethroids in Chinese tea by screening and confirmatory detection using GC-NCI-MS and GC-MS/MS. J. Agric. Food Chem. 2014;62(29):7092–100. https://doi.org/10.1021/jf5012424.
Cao Yalin, Tang Hang, Chen Dazhou,Li Lie. A novel method based on MSPD for simultaneous determination of 16 pesticide residues in tea by tea by LC–MS/MS. Journal of Chromatography B. 2015;998–999:72–79.
Rahim AA, Nofrizal S, Saad B. Rapid tea catechins and caffeine determination by HPLC using microwave-assisted extraction and silica monolithic column. Food Chem. 2014;147:262–8. https://doi.org/10.1016/j.foodchem.2013.09.131.
Cialla-May D, Zheng XS, Weber K, Popp J. Recent progress in surface-enhanced Raman spectroscopy for biological and biomedical applications: from cells to clinics. Chem. Soc. Rev. 2017;46(13):3945–61. https://doi.org/10.1039/c7cs00172j.
Ruditskiy A, Xia Y. The science and art of carving metal nanocrystals. ACS Nano. 2017;11(1):23–7. https://doi.org/10.1021/acsnano.6b08556.
Tian Z-H, Ren B, Wu D-Y. Surface-Enhanced Raman Scattering: From Noble to Transition Metals and from Rough Surfaces to Ordered Nanostructures. The Journal of Physical Chemistry B. 2002;106(37):9463–9483.
Zhou Q, Meng G, Liu J, Huang Z, Han F, Zhu C, et al. A hierarchical nanostructure-based surface-enhanced Raman scattering sensor for preconcentration and detection of antibiotic pollutants. Adv. Mater. Technol. 2017;2(6):1700028. https://doi.org/10.1002/admt.201700028.
Panneerselvam R, Liu G-K, Wang Y-H, Liu J-Y, Ding S-Y, Li J-F, et al. Surface-enhanced Raman spectroscopy: bottlenecks and future directions. Chem. Commun. 2018. https://doi.org/10.1039/c7cc05979e.
Zhang D, Liang P, Yu Z, Huang J, Ni D, Shu H, et al. The effect of solvent environment toward optimization of SERS sensors for pesticides detection from chemical enhancement aspects. Sens Actuators B. 2018;256:721–8. https://doi.org/10.1016/j.snb.2017.09.209.
Zhang J, Huang F, Lin Z. Progress of nanocrystalline growth kinetics based on oriented attachment. Nanoscale. 2010;2(1):18–34. https://doi.org/10.1039/b9nr00047j.
Li C, Cheng Y, Xu S, Chao Z, Zhen L, Liu X, et al. Ag2O@Ag core-shell structure on PMMA as low-cost and ultra-sensitive flexible surface-enhanced Raman scattering substrate. J. Alloys Compd. 2017;695:1677–84.
Cabrera C, Artacho R, Giménez R. Beneficial effects of green tea—a review. J. Am. Coll. Nutr. 2006;25(2):79–99. https://doi.org/10.1080/07315724.2006.10719518.
Feng S, Hu Y, Ma L, Lu X. Development of molecularly imprinted polymers-surface-enhanced Raman spectroscopy/colorimetric dual sensor for determination of chlorpyrifos in apple juice. Sens. Actuators B. 2017;241:750–7. https://doi.org/10.1016/j.snb.2016.10.131.
Huang S, Hu J, Guo P, Liu M, Wu R. Rapid detection of chlorpyriphos residue in rice by surface-enhanced Raman scattering. Anal. Methods. 2015;7(10):4334–9. https://doi.org/10.1039/c5ay00381d.
Xu Q, Guo X, Xu L, Ying Y, Wu Y, Wen Y, et al. Template-free synthesis of SERS-active gold nanopopcorn for rapid detection of chlorpyrifos residues. Sens. Actuators B. 2017;241:1008–13. https://doi.org/10.1016/j.snb.2016.11.021.
Chen J, Huang Y, Kannan P, Zhang L, Lin Z, Zhang J, et al. Flexible and adhesive surface enhance Raman scattering active tape for rapid detection of pesticide residues in fruits and vegetables. Anal. Chem. 2016;88(4):2149–55. https://doi.org/10.1021/acs.analchem.5b03735.
Shende C, Inscore F, Sengupta A, Stuart J, Farquharson S. Rapid extraction and detection of trace Chlorpyrifos-methyl in orange juice by surface-enhanced Raman spectroscopy. Sensing and Instrumentation for Food Quality and Safety. 2010;4(3-4):101–7. https://doi.org/10.1007/s11694-010-9100-6.
He Y, Xiao S, Dong T, Nie P. Gold nanoparticles with different particle sizes for the quantitative determination of chlorpyrifos residues in soil by SERS. Int J Mol Sci 2019;20(11). doi:https://doi.org/10.3390/ijms20112817
Hui-Min MA, Wang YQ, Qian H. International comparative analysis of standards for tea pesticide residue limits. China Tea Processing 2012.
You T, Jiang L, Yin P, Shang Y, Zhang D, Guo L, et al. Direct observation of p,p '-dimercaptoazobenzene produced from p-aminothiophenol and p-nitrothiophenol on Cu2O nanoparticles by surface-enhanced Raman spectroscopy. J. Raman Spectrosc. 2014;45(1):7–14. https://doi.org/10.1002/jrs.4411.
Yang C, Liang P, Tang L, Zhou Y, Cao Y, Wu Y, et al. Synergistic effects of semiconductor substrate and noble metal nano-particles on SERS effect both theoretical and experimental aspects. Appl. Surf. Sci. 2018;436:367–72. https://doi.org/10.1016/j.apsusc.2017.12.074.
Manisekaran R. Literature survey on magnetic, gold, and core-shell nanoparticles. Design and Evaluation of Plasmonic/Magnetic Au-MFe2O4(M-Fe/Co/Mn). 2018. https://doi.org/10.1007/978-3-319-67609-8_2.
Chen Y, Bai X, Su L, Du Z, Shen A, Materny A, et al. combined labelled and label-free SERS probes for triplex three-dimensional cellular imaging. Sci. Rep. 2016;6:19173.
Wu YX, Liang P, Dong QM, Bai Y, Yu Z, Huang J, et al. Design of a silver nanoparticle for sensitive surface enhanced Raman spectroscopy detection of carmine dye. Food Chem. 2017;237:974–80. https://doi.org/10.1016/j.foodchem.2017.06.057.
Ratkaj M, Biljan T, Miljanić S. Quantitative analysis of entacapone isomers using surface-enhanced Raman spectroscopy and partial least squares regression. Appl. Spectrosc. 2012;66(12):1468.
Janči VT, Kljusurić D, Gajdoš MJ, Ivanda L, Vidaček M. Sanja Artificial neural network models for determination of histamine in fish by surface enhanced Raman spectroscopy. In: Wefta Conference, 2017.
Li SX, Zeng QY, Li LF, Zhang YJ, Wan MM, Liu ZM, et al. Study of support vector machine and serum surface-enhanced Raman spectroscopy for noninvasive esophageal cancer detection. J. Biomed. Opt. 2013;18(2):27008.
Acknowledgements
The project was financially supported by the Fundamental Research Funds for the Central Universities (program no. 2662017JC035), and the National Science Foundation for Young Scholars of China (grant no. 31000316), the Application Research Program of Commonweal Technology of Zhejiang Province (no. 2014C37042), the Zhejiang Province University Students in Scientific and Technological Innovation Activities (no. 2016R409011), the Science and Technology project of Zhejiang Province (no. 2016C33026), and the Science and Technology Project of Three Gorges Emigration. PL also thanks the National Demonstration Base for Micro/nano-fabrication & Optoelectronic Detection and the International Science and Technology Cooperation for the support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 346 kb)
Rights and permissions
About this article
Cite this article
Zhang, D., Liang, P., Ye, J. et al. Detection of systemic pesticide residues in tea products at trace level based on SERS and verified by GC–MS. Anal Bioanal Chem 411, 7187–7196 (2019). https://doi.org/10.1007/s00216-019-02103-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-019-02103-7