Abstract
A simple method to determine hazardous silver nanoparticles (AgNPs) based on ionic liquid (IL) dispersive liquid–liquid microextraction and back-extraction is described. This approach involves AgNP stabilization using a cationic surfactant followed by extraction from the sample matrix by means of an IL as an extraction phase. Certain ILs have high affinity for metals, and preliminary experiments showed that those ILs consisting of imidazolium cation efficiently extracted AgNPs in the presence of a cationic surfactant and a chelating agent. Afterward, histamine was used as a dispersing agent to promote phase transfer of differently coated AgNPs from the IL in aqueous solution to be subsequently analyzed by UV–visible spectrometry. The analytical procedure allows AgNPs to be recovered from the sample matrix in an aqueous medium, the enrichment factor being up to 4, preserving both AgNP size and AgNP shape as demonstrated by transmission electron microscopy images and the localized surface plasmon resonance band characteristic of each AgNP. The present method exhibited a linear response for AgNPs in the range from 3 to 20 μg/mL, the limit of detection being 0.15 μg/mL. Method efficiency was assessed in spiked orange juice and face cream, yielding recoveries ranging from 75.7% to 96.6%. The method was evaluated in the presence of other nanointerferents (namely, gold nanoparticles). On the basis of diverse electrophoretic mobilities and surface plasmon resonance bands for metal nanoparticles, capillary electrophoresis was used to prove the lack of interaction of the target AgNPs with gold nanoparticles during the whole protocol; thus, interferents do not affect AgNP determination. As a consequence, the analytical approach described has great potential for the analysis of engineered nanosilver in consumer products.

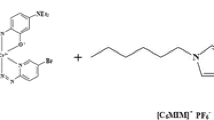
Simple protocol for the determination of silver nanoparticles (AgNPs) based on dispersive liquid–liquid extraction with a specific short alkyl side chain ionic liquid and their quantitative detection with a UV–visible spectrometer. HMIM•PF6 1-hexyl-3-methylimidazolium hexafluorophosphate, NP nanoparticle, SPR surface plasmon resonance




Similar content being viewed by others
Abbreviations
- AgNP:
-
Silver nanoparticle
- AuNP:
-
Gold nanoparticle
- CAPS:
-
3-(Cyclohexylamino)-1-propanesulfonic acid
- CE:
-
Capillary electrophoresis
- CTAB:
-
Cetyltrimethylammonium bromide
- CTAC:
-
Cetyltrimethylammonium chloride
- EDTA:
-
Ethylenediaminetetraacetic acid
- HMIM-PF6 :
-
1-Hexyl-3-methylimidazolium hexafluorophosphate
- IL:
-
Ionic liquid
- NP:
-
Nanoparticle
- PVP:
-
Polyvinylpyrrolidone
- RSD:
-
Relative standard deviation
- SDS:
-
Sodium dodecyl sulfate
- SPR:
-
Surface plasmon resonance
- TEM:
-
Transmission electron microscopy
References
He X, Deng H, Hwang H-m. The current application of nanotechnology in food and agriculture. J Food Drug Anal. 2019;27(1):1–21.
Kessler R. Engineered nanoparticles in consumer products: understanding a new ingredient. Environ Health Perspect. 2011;119(3):A120–5.
Higashisaka K, Nagano K, Yoshioka Y, Tsutsumi Y. Nano-safety research: examining the associations among the biological effects of nanoparticles and their physicochemical properties and kinetics. Biol Pharm Bull. 2017;40(3):243–8.
K. Aschberger, H. Rauscher, H. Crutzen, K. Rasmussen, F.M. Christensen, B. Sokull-Klüttgen, H. Stamm. Considerations on information needs for nanomaterials in consumer products. Discussion of a labelling and reporting scheme for nanomaterials in consumer products in the EU. Report EUR 26560. Ispra: Joint Research Centre; 2014. https://doi.org/10.2788/3044.
Guo H, Zhang Z, Xing B, Mukherjee A, Musante C, White JC, et al. Analysis of silver nanoparticles in antimicrobial products using surface-enhanced Raman spectroscopy (SERS). Environ Sci Technol. 2015;49(7):4317–24.
Wei G, Liu FK, Wang CRC. Size-exclusion chromatography of metal nanoparticles and quantum dots. Trends Anal Chem. 2016;80:311–20.
Li L, Leopold K, Schuster M. Effective and selective extraction of noble metal nanoparticles from environmental water through a noncovalent reversible reaction on an ionic exchange resin. Chem Commun. 2012;48:9165–7.
Feichmeier NS, Leopold K. Detection of silver nanoparticles in parsley by solid sampling high-resolution-continuum source atomic absorption spectrometry. Anal Bioanal Chem. 2014;406(16):3887–94.
Schwertfeger DM, Velicogna JR, Jesmer AH, Saatcioglu S, McShane H, Scroggins RP, et al. Extracting metallic nanoparticles from soils for quantitative analysis: method development using engineered silver nanoparticles and SP-ICP-MS. Anal Chem. 2017;89(4):2505–13.
Hoque ME, Khosravi K, Newman K, Metcalfe CD. Detection and characterization of silver nanoparticles in aqueous matrices using asymmetric-flow field flow fractionation with inductively coupled plasma mass spectrometry. J Chromatogr A. 2012;1233:109–15.
Chao JB, Liu JF, Yu SJ, Feng YD, Tan ZQ, Liu R, et al. Speciation analysis of silver nanoparticles and silver ions in antibacterial products and environmental waters via cloud point extraction based separation. Anal Chem. 2011;83:6875–82.
Mattarozzi M, Suman M, Cascio C, Calestani D, Weigel S, Undas A, et al. Analytical approaches for the characterization and quantification of nanoparticles in food and beverages. Anal Bioanal Chem. 2017;409(1):63–80.
Bolea E, Jiménez-Lamana J, Laborda F, Abad-Álvaro I, Bladé C, Arola L, et al. Detection and characterization of silver nanoparticles and dissolved species of silver in culture medium and cells by AsFlFFF-UV-Vis-ICPMS: application to nanotoxicity tests. Analyst. 2014;139:914–22.
Huynh KA, Siska E, Heithmar E, Tadjiki S, Pergantis SA. Detection and quantification of silver nanoparticles at environmentally relevant concentrations using asymmetric flow field–flow fractionation online with single particle inductively coupled plasma mass spectrometry. Anal Chem. 2016;88(9):4909–16.
Motellier S, Pelissier N, Mattei JG. Contribution of single particle inductively coupled plasma mass spectrometry and asymmetrical flow field-flow fractionation for the characterization of silver nanosuspensions. Comparison with other sizing techniques. J Anal At Spectrom. 2017;32(7):1348–58.
Cayuela A, Soriano ML, Valcárcel M. Reusable sensor based on functionalized carbon dots for the detection of silver nanoparticles in cosmetics via inner filter effect. Anal Chim Acta. 2015;872:70–6.
Ruiz-Palomero C, Soriano ML, Valcárcel M. Sulfonated nanocellulose for the efficient dispersive micro solid-phase extraction and determination of silver nanoparticles in food products. J Chromatogr A. 2016;1428:352–8.
Ruiz-Palomero C, Soriano ML, Valcárcel M. Gels based on nanocellulose with photosensitive ruthenium bipyridine moieties as sensors for silver nanoparticles in real samples. Sensors Actuators B Chem. 2016;229:352–8.
Kumar A, Joshi H, Pasricha R, Mandale AB, Sastry M. Phase transfer of silver nanoparticles from aqueous to organic solutions using fatty amine molecules. J Colloid Interface Sci. 2013;264(2):396–401.
Lei Z, Chen B, Koo Y-M, MacFarlane DR. Introduction: ionic liquids. Chem Rev. 2017;117(10):6633–5.
Chen YL, Cao SR, Zhang L, Xi CX, Li XL, Chen ZQ, et al. Preparation of size-controlled magnetite nanoparticles with a graphene and polymeric ionic liquid coating for the quick, easy, cheap, effective, rugged and safe extraction of preservatives from vegetables. J Chromatogr A. 2016;1448:9–19.
Del Sesto RE, Koppisch AT, Fox DT, Jones MR, Lovejoy KS, Stevens TE, et al. Biphasic extraction, recovery and identification of organic and inorganic compounds with ionic liquids. In: Shiflett MB, Scurto AM, editors. Ionic liquids: current state and future directions. ACS symposium series, vol. 1250. Washington: American Chemical Society; 2017. p. 283–302.
Berthod A, Ruiz-Ángel MJ, Carda-Broch S. Recent advances on ionic liquid uses in separation techniques. J Chromatogr A. 2018;1559:2–16. https://doi.org/10.1016/j.chroma.2017.09.044.
Ventura SPM, e Silva FA, Quental MV, Mondal D, Freire MG, Coutinho JAP. Ionic-liquid-mediated extraction and separation processes for bioactive compounds: past, present, and future trends. Chem Rev. 2017;117(10):6984–7052.
Justin SJ, Peter B, Jarno M, Lea R. Environmental aspects of metals removal from waters and gold recovery. AIChE J. 2015;61:2739–48.
He Z, Alexandridis P. Nanoparticles in ionic liquids: interactions and organization. Phys Chem Chem Phys. 2015;17(28):18238–61.
Chen S, Sun Y, Chao J, Cheng L, Chen Y, Liu J. Dispersive liquid–liquid microextraction of silver nanoparticles in water using ionic liquid 1-octyl-3-methylimidazolium hexafluorophosphate. J Environ Sci. 2016;41:211–7.
López-Lorente AI, Soriano ML, Valcárcel M. Analysis of citrate-capped gold and silver nanoparticles by thiol ligand exchange capillary electrophoresis. Microchim Acta. 2014;181:1789–96.
Dueñas-Mas MJ, Soriano ML, Ruiz-Palomero C, Valcárcel M. Modified nanocellulose as promising material for the extraction of gold nanoparticles. Microchem J. 2018;138:379–83.
Lopez-Lorente AI, Simonet BM, Valcárcel M. Rapid analysis of gold nanoparticles in liver and river water samples. Analyst. 2012;137:3528–34.
Docherty KM, Dixon JK, Kulpa CF Jr. Biodegradability of imidazolium and pyridinium ionic liquids by an activated sludge microbial community. Biodegradation. 2007;18:481–93.
Liu FK, Tsai MH, Hsu YC, Chu TC. Analytical separation of Au/Ag core/shell nanoparticles by capillary electrophoresis. J Chromatogr A. 2006;1133:340–6.
López-Lorente AI, Simonet B, Valcárcel M. Electrophoretic methods for the analysis of nanoparticles. Trends Anal Chem. 2011;30:58–71.
Acknowledgements
This research was supported by the European Commission within the Seventh Framework Programme for the project FP7-NMP-2007-2013-SME5-280550. MLS expresses her gratitude to the Junta de Comunidades de Castilla-La Mancha-FEDER Funds for funding the project SBPLY/17/180501/000333.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
ABC Highlights: authored by Rising Stars and Top Experts.
Electronic supplementary material
ESM 1
(PDF 136 kb)
Rights and permissions
About this article
Cite this article
Soriano, M.L., Ruiz-Palomero, C. & Valcárcel, M. Ionic-liquid-based microextraction method for the determination of silver nanoparticles in consumer products. Anal Bioanal Chem 411, 5023–5031 (2019). https://doi.org/10.1007/s00216-019-01889-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-019-01889-w