Abstract
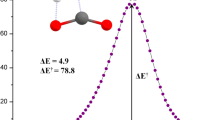
The mechanism of dihydrogen activation has been theoretically investigated by means of DFT calculation. An experimentally synthesized bridged P/B frustrated Lewis pair (FLP) and two designed FLPs are used for this purpose. The model FLPs 2 and 3 are more efficient than FLP 1 for H2 activation as revealed by the thermochemical and kinetic data. A significant amount of electron density is transferred from H2 molecule to the FLPs at the transition states (TSs) during the process of H2 activation, and this is greater at the corresponding TSs of FLPs 2 and 3 than that of FLP 1. The NICS(0) and NICS(1zz) of the boron heterocycle at the FLPs 2 and 3, and at the corresponding TSs and the product geometries of H2 activation demonstrate that the anti-aromatic character of the rings in the FLPs is remarkably reduced at the TSs and finally at the products and that is most likely responsible for enhanced activity of FLPs 2 and 3 by decreasing the activation barrier.




Similar content being viewed by others
References
Appelt C, Westenberg H, Bertini F, Ehlers AW, Slootweg JC, Lammertsma K, Uhl W (2011) Geminal Phosphorus/Aluminum-based frustrated Lewis pairs: C-H versus C-C activation and CO2 fixation. Angew Chem Int Ed 50:3925–3928
Ashley AE, Thompson AL, Hare DO (2009) Non-metal-mediated homogeneous hydrogenation of CO2 to CH3OH. Angew Chem Int Ed 48:9839–9843
Axenov KV, Kehr G, Frohlich R, Erker G (2009) Catalytic hydrogenation of sensitive organometallic compounds by antagonistic N/B Lewis pair catalyst systems. J Am Chem Soc 131:3454–3455
Bertini F, Lyaskovskyy V, Timmer BJJ, de Kanter FJJ, Lutz M, Ehlers AW, Chris Slootweg J, Lammertsma K (2012) Preorganized frustrated Lewis pairs. J Am Chem Soc 134:201–204
Cabrera-Trujillo JJ, Fernández I (2019) Aromaticity can enhance the reactivity of P-donor/borole frustrated Lewis pairs. Chem Commun 55:675–678
Cardenas AJP, Culotta BJ, Warren TH, Grimme S, Stute A, Froehlich R, Kehr G, Erker G (2011) Capture of NO by a frustrated Lewis pair: a new type of persistent N-Oxyl radical. Angew Chem Int Ed 50:7567–7571
Cedillo A, Chattaraj PK, Parr RG (2000) An atoms- in-molecules partitioning of the density. Int J Quantum Chem 77:403–407
Chase PA, Jurca T, Stephan DW (2008) Lewis acid-catalyzed hydrogenation: B(C6F5)3-mediated reduction of imines and nitriles with H2. ChemCommun 14:1701–1703
Chase PA, Welch GC, Jurca T, Stephan DW (2007) Metal-free catalytic hydrogenation. Angew Chem Int Ed 46:8050–8053
Chattaraj PK, Maiti B, Sarkar U (2003) Philicity: a unified treatment of chemical reactivity and selectivity. J Phys Chem A 107:4973
Cheeseman JR, Trucks GW, Keith TA, Frisch MJ (1996) A comparison of models for calculating nuclear magnetic resonance shielding tensors. J Chem Phys 104:5497–5509
Chernichenko K, Madarasz A, Papai I, Nieger M, Leskela M, Repo TA (2013) frustrated-Lewis-pair approach to catalytic reduction of alkynes to cis-alkenes. Nat Chem 5:718–723
Courtemanche M-A, Legare M-A, Maron L, Fontaine F-G (2014) Reducing CO2 to methanol using frustrated Lewis pairs: on the mechanism of phosphine−borane-mediated hydroboration of CO2. J Am Chem Soc 136:10708–10717
Courtemanche M-A, Pulis AP, Rochette E, Légaré M-A, Stephan DW, Fontaine F-G (2015) Intramolecular B/N frustrated Lewis pairs and the hydrogenation of carbon dioxide. Chem Commun 51:9797–9800
Dong S, Daniliuc CG, Kehr G, Erker G (2020) Formation of active cyclic five-membered frustrated Phosphane/Borane Lewis pairs and their cycloaddition reactions. ChemEur J 26:745–753
Dureen MA, Stephan DW (2010) Reactions of boron amidinates with CO2 and CO and other small molecules. J Am Chem Soc 132:13559–13568
Eicherb J, Vankova N (2015) Are intramolecular frustrated Lewis pairs also intramolecular catalysts? A theoretical study on H2 activation. Phys Chem Chem Phys 17:10687–10698
Eros G, Nagy K, Mehdi H, Papai I, Nagy P, Kiraly P, Tarkanyi G, Soos T (2012) Catalytic hydrogenation with frustrated Lewis pairs: selectivity achieved by size-exclusion design of Lewis acids. Chem - Eur J 18:574–585
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA et al. (2016) Gaussian 16, Revision B.01, Gaussian, Inc., Wallingford, CT
Gao B, Feng X, Meng W, Du H (2020) Asymmetric hydrogenation of ketones and enones with chiral Lewis base derived frustrated Lewis pairs. Angew Chem Int Ed 59:2–9
Ghara M, Chattaraj PK (2018a) Fixation of nitrous oxide (N2O) by 1, 4, 2, 5-diazadiborinine: a DFT study. Int J Quantum Chem 118:e25593
Ghara M, Chattaraj PK (2018b) A DFT study on trapping of nitric oxide by 1,3,2,5-diazadiborinine, a frustrated Lewis pair. J Indian Chem Soc 95:1019–1024
Ghara M, Chattaraj PK (2019) A computational study on hydrogenation of CO2, catalyzed by a bridged B/N frustrated Lewis pair. Struct Chem 30:1067–1077
Ghara M, Giri S, Chattaraj PK (2020) Cycloaddition Reactions between H2C = CHR (R = H, CN, CH3) and a cyclic P/B frustrated Lewis pair: a DFT study. J Phys Chem A 124:4455–4462
Ghara M, Pan S, Chattaraj PK (2019) A theoretical investigation on boron-ligand cooperation to activate molecular hydrogen by a frustrated Lewis pair and subsequent reduction of carbon dioxide. Phys Chem Chem Phys 21:21267–21277
Grimme S, Antony J, Ehrlich S, Krieg H (2010) A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J Chem Phys 132:154104
Grimme S, Kruse H, Goerigk L, Erker G (2010) The mechanism of dihydrogen activation by frustrated Lewis pairs revisited. Angew Chem Int Ed 49:1402–1405
Hamza A, Stirling A, Rokob TA, Pápai I (2009) Mechanism of hydrogen activation by frustrated Lewis pairs: a molecular orbital approach. Int J Quantum Chem 109:2416–2425
Hirshfeld FL (1977) Bonded-atom fragments for describing molecular charge densities. TheorChimActa 44:129–138
Hounjet LJ, Bannwarth C, Garon CN, Caputo CB, Grimme S, Stephan DW (2013) Combinations of ethers and B(C6F5)3 function as hydrogenation catalysts. Angew Chem Int Ed 52:7492–7495
Jiang CF, Blacque O, Fox T, Berke H (2011) Reversible, metal-free hydrogen activation by frustrated Lewis pairs. Dalton Trans 40:1091–1097
Kolychev EL, Bannenberg T, Freytag M, Daniliuc CG, Jones PG, Tamm M (2012) Reactivity of a frustrated Lewis pair and small-molecule activation by an isolable arduengocarbene-B{3,5-(CF3)2C6H3}3 complex. ChemEur J 18:16938–16946
Longobardi LE, Tang C, Stephan DW (2014) Stoichiometric reductions of alkyl-substituted ketones and aldehydes to borinic esters. Dalton Trans 43:15723–15726
Mahdi T, del Castillo JN, Stephan DW (2013) Metal-free hydrogenation of N-based heterocycles. Organometallics 32:1971–1978
Mahdi T, Stephan DW (2014) Enabling catalytic ketone hydrogenation by frustrated Lewis pairs. J Am Chem Soc 136:15809–15812
Mahdi T, Stephan DW (2015) Facile protocol for catalytic frustrated Lewis pair hydrogenation and reductive deoxygenation of ketones and aldehydes. Angew Chem Int Ed 54:8511–8514
Mardirossian N, Head-Gordon M (2016) How accurate are the minnesota density functionals for noncovalent interactions, isomerization energies, thermochemistry, and barrier heights involving molecules composed of main-group elements? J Chem Theory Comput 12:4303–4325
Marenich AV, Cramer CJ, Truhlar DG (2009) Performance of SM6, SM8, and SMD on the SAMPL1 test set for the prediction of small-molecule solvation free energies. J Phys Chem B 113:4538–4543
Mc Cahill JSJ, Welch GC, Stephan DW (2007) Reactivity of “Frustrated Lewis Pairs”: three-component reactions of phosphines, a borane, and olefins. Angew Chem Int Ed 46:4968–4971
Menard G, Stephan DW (2010) Room temperature reduction of CO2 to methanol by Al-based frustrated Lewis pairs and ammonia borane. J Am Chem Soc 132:1796–1797
Moemming CM, Otten E, Kehr G, Froehlich R, Grimme S, Stephan DW, Erker G (2009) Reversible metal-free carbon dioxide binding by frustrated Lewis pairs. Angew Chem Int Ed 48:6643–6646
Muck-Lichtenfeld C, Grimme S (2012) Theoretical analysis of cooperative effects of small molecule activation by frustrated Lewis pairs. Dalton Trans 41:9111–9118
Paradies J (2014) Metal-free hydrogenation of unsaturated hydrocarbons employing molecular hydrogen. Angew Chem Int Ed 53:3552–3557
Pereira JCM, Sajid M, Kehr G, Wright AM, Schirmer B, Qu Z-W, Grimme S, Erker G, Ford PC (2014) Reaction of a bridged frustrated Lewis pair with nitric oxide: a kinetics study. J Am Chem Soc 136:513–519
Pérez P, Yepes D, Jaque P, Chamorro E, Domingo LR, Rojas RS, Toro-Labbé A (2015) A computational and conceptual DFT study on the mechanism of hydrogen activation by novel frustrated Lewis pairs. Phys Chem Chem Phys 17:10715–10725
Piers WE, Marwitz AJV, Mercier LG (2011) Mechanistic aspects of bond activation with perfluoroarylboranes. Inorg Chem 50:12252–12262
Rajeev R, Sunoj RB (2009) On the origin of reversible hydrogen activation by phosphine–boranes. Chem -Eur J 15:12846–12855
Reed AE, Weinstock RB, Weinhold F (1985) Natural population analysis. J Chem Phys 83:735–746
Rokob TA, Bakó I, Stirling A, Hamza A, Pápai I (2013) Reactivity models of hydrogen activation by frustrated Lewis pairs: synergistic electron transfers or polarization by electric field? J Am Chem Soc 135:4425–4437
Rokob TA, Hamza A, Pápai I (2009) Rationalizing the reactivity of frustrated Lewis pairs: thermodynamics of H2 activation and the role of acid−base properties. J Am Chem Soc 131:10701–10710
Rokob TA, Hamza A, Stirling A, Pápai I (2009) On the mechanism of B(C6F5)3-catalyzed direct hydrogenation of imines: inherent and thermally induced frustration. J Am Chem Soc 131:2029–2036
Rokob TA, Hamza A, Stirling A, Soos T, Papai I (2008) Turning frustration into bond activation: a theoretical mechanistic study on heterolytic hydrogen splitting by frustrated Lewis pairs. Angew Chem Int Ed 47:2435–2438
Roy DR, Parthasarathi R, Padmanabhan J, Sarkar U, Subramanian V, Chattaraj PK (2006) Careful scrutiny of the philicity concept. J Phys Chem A 110:1084–1093
Sajid M, Kehr G, Daniliuc CG, Erker G (2014) Formylborane formation with frustrated Lewis pair templates. Angew Chem Int Ed 53:1118–1121
Samigullin K, Georg I, Bolte M, Lerner H-W, Wagner M (2016) A highly reactive geminal P/B frustrated Lewis pair: expanding the scope to C-X(X=Cl, Br) bond activation. ChemEur J 22:3478–3484
Schirmer B, Grimme S (2010) Electric field induced activation of H2 - Can DFT do the job? Chem Commun 46:7942–7944
Schleyer PVR, Maerker C, Dransfeld A, Jiao H, Hommes NJRVE (1996) Nucleus-independent chemical shifts: a simple and efficient aromaticity probe. J Am ChemSoc 118:6317
Scott DJ, Fuchter MJ, Ashley AE (2014) Nonmetal catalyzed hydrogenation of carbonyl compounds. J Am Chem Soc 136:15813–15816
Segawa Y, Stephan DW (2012) Metal-free hydrogenation catalysis of polycyclic aromatic hydrocarbons. Chem Commun 48:11963–11965
Sitte NA, Bursch M, Grimme S, Paradies J (2019) Frustrated lewis pair catalyzed hydrogenation of amides: halides as active Lewis base in the metal-free hydrogen activation. J Am Chem Soc 141:159–162
Skara G, De Vleeschouwer F, Geerlings P, De Proft F, Pinter B (2017) Heterolytic splitting of molecular hydrogen by frustrated and classical Lewis pairs: a unified reactivity concept. Sci Rep 7:16024
Spies P, Schwendemann S, Lange S, Kehr G, Frhlich R, Erker G (2008) Metal-free catalytic hydrogenation of enamines, imines, and conjugated phosphinoalkenylboranes. Angew Chem Int Ed 47:7543–7546
Stephan DW (2008) “Frustrated Lewis pairs”: a concept for new reactivity and catalysis. Org Biomol Chem 6:1535–1539
Stephan DW, Erker G (2010) Frustrated Lewis pairs: metal-free hydrogen activation and more. Angew Chem Int Ed 49:46–76
Stephan DW, Erker G (2014) Frustrated Lewis pair chemistry of carbon, nitrogen and sulfur oxides. Chem Sci 5:2625–2641
Stephan DW, Erker G (2015) Frustrated Lewis pair chemistry: development and perspectives. Angew Chem Int Ed 54:6400–6441
Sumerin V, Schulz F, Atsumi M, Wang C, Nieger M, Leskela M, Repo T, Pyykko P, Rieger B (2008) Molecular tweezers for hydrogen: synthesis, characterization, and reactivity. J Am Chem Soc 130:14117–14119
Theuergarten E, Schlüns D, Grunenberg J, Daniliuc CG, Jones PG, Tamm M (2010) Intramolecular heterolytic dihydrogen cleavage by a bifunctional frustrated pyrazolylborane Lewis pair. Chem Commun 46:8561–8563
Trunk M, Teichert JF, Thomas A (2017) Room-temperature activation of hydrogen by semi-immobilized frustrated Lewis pairs in microporous polymer networks. J Am Chem Soc 139:3615–3618
Welch GC, Juan RRS, Masuda JD, Stephan DW (2006) Reversible metal-free hydrogen activation. Science 314:1124–1126
Welch GC, Stephan DW (2007) Facile heterolytic cleavage of dihydrogen by phosphines and boranes. J Am Chem Soc 129:1880–1881
Wen M, Huang F, Lu G, Wang Z-X (2013) Density functional theory mechanistic study of the reduction of CO2 to CH4 catalyzed by an ammonium hydridoborate ion pair: CO2 activation via formation of a formic acid entity. Inorg Chem 52:12098–12107
Whittemore SM, Edvenson G, Camaioni DM, Karkamkar A, Neiner D, Parab K, Autrey T (2015) Catalytic reduction of polar substrates without metals: a thermodynamic and kinetic study of heterolytic activation of hydrogen by vacancies in frustrated Lewis pairs. Catal Today 251:28–33
Wolff NV, Lefevre G, Berthet J-C, Thuery P, Cantat T (2016) Implications of CO2 activation by frustrated Lewis pairs in the catalytic hydroboration of CO2: a view using N/Si+ frustrated Lewis pairs. ACS Catal 6:4526–4535
Wolinski K, Hilton JF, Pulay P (1990) Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J Am Chem Soc 112:8251–8260
Yanez RAA, Kehr G, Daniliuc CG, Schirmer B, Erker G (2014) Formation of a dihydroborole by catalytic isomerization of a divinylborane. Dalton Trans 43:10794–10800
Zhao X, Stephan DW (2011) Olefin–borane “van der Waals Complexes”: intermediates in frustrated Lewis pair addition reactions. J Am Chem Soc 133:12448–12450
Zhao Y, Truhlar DG (2008a) The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor Chem Acc 120:215–241
Zhao Y, Truhlar DG (2008b) Exploring the limit of accuracy of the global hybrid meta density functional for main-group thermochemistry, kinetics, and noncovalent interactions. J Chem Theory Comput 4:1849–1868
Acknowledgements
We are delighted to dedicate this article to Professor Ramon Carbó-Dorca on his 80th birth anniversary. We would like to thank Professors Gernot Frenking, Miquel Solà and Tanmoy Chakraborty for kindly inviting us to contribute this article to the Special Issue of the Theoretical Chemistry Accounts. PKC thanks the DST, New Delhi, for his J. C. Bose National Fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Published as part of the special collection of articles “Festschrift in honour of Prof. Ramon Carbó-Dorca”.
Rights and permissions
About this article
Cite this article
Ghara, M., Chattaraj, P.K. Can a decrease in anti-aromaticity increase the dihydrogen activation ability of a frustrated phosphorous/borane Lewis pair?: a DFT study. Theor Chem Acc 139, 183 (2020). https://doi.org/10.1007/s00214-020-02698-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-020-02698-6