Abstract
Rationale
The precise diagnosis and treatment of cognitive impairment remains a major challenge in the field of schizophrenia (SCZ) research. Synaptic dysfunction and loss are thought to be closely related to the occurrence and development of SCZ and may be involved in cognitive dysfunction.
Objectives
The purpose of this study was to investigate whether neuronal pentraxins (NPTXs) plays a role in the etiology of SCZ and provide evidence of its possible therapeutic value a new target for drug development.
Methods
We recruited 275 participants, of whom 148 were SCZ from psychiatric hospital and 127 healthy control (HC) subjects from communities. Plasma concentrations of NPTXs were measured in HC and SCZ at baseline and after 8 weeks of antipsychotic treatment. The MATRICS Cognitive Consensus Battery was used to evaluate cognitive function. Furthermore, the brain is parcellated into 246 subregions using the Brainnetome atlas, and we extracted regional white matter volumes from magnetic resonance images of the SCZ groups.
Results
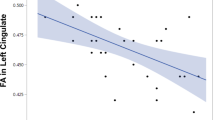
Plasma NPTX2 levels were significantly lower in SCZ compared with HC subjects, but were significantly raised in SCZ after 8 weeks of antipsychotic treatment compared to baseline. In addition, baseline plasma NPTX2 levels were positively correlated with cognitive performance.
Conclusions
These findings indicate that NPTX2 may reveal novel aspects of disease etiology and act as a promising target for new drug development.




Similar content being viewed by others
Data Availability
Data presented in this paper are available from the corresponding author upon reasonable request.
References
Andreasen NC, Pressler M, Nopoulos P, Miller D, Ho BC (2010) Antipsychotic dose equivalents and dose-years: a standardized method for comparing exposure to different drugs. Biol Psychiatry 67:255–62
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B (Methodological) 57:289–300
Butterfield DA, Halliwell B (2019) Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat Rev Neurosci 20:148–160
Chang MC, Park JM, Pelkey KA, Grabenstatter HL, Xu D, Linden DJ, Sutula TP, McBain CJ, Worley PF (2010) Narp regulates homeostatic scaling of excitatory synapses on parvalbumin-expressing interneurons. Nat Neurosci 13:1090–7
de San Gomez, Jose N, Massa F, Halbgebauer S, Oeckl P, Steinacker P, Otto M (2022) Neuronal pentraxins as biomarkers of synaptic activity: from physiological functions to pathological changes in neurodegeneration. J Neural Transm (Vienna) 129:207–230
Fan L, Li H, Zhuo J, Zhang Y, Wang J, Chen L, Yang Z, Chu C, Xie S, Laird AR, Fox PT, Eickhoff SB, Yu C, Jiang T (2016) The human Brainnetome Atlas: a new brain atlas based on connectional architecture. Cereb Cortex 26:3508–26
Fatemi SH, Folsom TD (2009) The neurodevelopmental hypothesis of schizophrenia, revisited. Schizophr Bull 35:528–48
Figueira ML, Brissos S (2011) Measuring psychosocial outcomes in schizophrenia patients. Curr Opin Psychiatry 24:91–9
Glausier JR, Lewis DA (2013) Dendritic spine pathology in schizophrenia. Neuroscience 251:90–107
Goverti D, Buyukluoglu N, Kaya H, Yuksel RN, Yucel C, Goka E (2022) Neuronal pentraxin-2 (NPTX2) serum levels during an acute psychotic episode in patients with schizophrenia. Psychopharmacology (Berl) 239:2585–2591
Gu Y, Huang S, Chang MC, Worley P, Kirkwood A, Quinlan EM (2013) Obligatory role for the immediate early gene NARP in critical period plasticity. Neuron 79:335–46
Havsteen I, Ohlhues A, Madsen KH, Nybing JD, Christensen H, Christensen A (2017) Are movement artifacts in magnetic resonance imaging a real problem?-a narrative review. Front Neurol 8:232
Heckers S, Konradi C (2015) GABAergic mechanisms of hippocampal hyperactivity in schizophrenia. Schizophr Res 167:4–11
Herrmann CS, Munk MH, Engel AK (2004) Cognitive functions of gamma-band activity: memory match and utilization. Trends Cogn Sci 8:347–55
Howard MW, Rizzuto DS, Caplan JB, Madsen JR, Lisman J, Aschenbrenner-Scheibe R, Schulze-Bonhage A, Kahana MJ (2003) Gamma oscillations correlate with working memory load in humans. Cereb Cortex 13:1369–74
Huang XF, Song X (2019) Effects of antipsychotic drugs on neurites relevant to schizophrenia treatment. Med Res Rev 39:386–403
Jauhar S, Johnstone M, McKenna PJ (2022) Schizophrenia. Lancet 399:473–486
Kahn RS, Keefe RS (2013) Schizophrenia is a cognitive illness: time for a change in focus. JAMA Psychiatry 70:1107–12
Kay SR, Fiszbein A, Opler LA (1987) The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr Bull 13:261–76
Kimoto S, Zaki MM, Bazmi HH, Lewis DA (2015) Altered markers of cortical gamma-aminobutyric acid neuronal activity in schizophrenia: role of the NARP gene. JAMA Psychiatry 72:747–56
Konradi C, Heckers S (2001) Antipsychotic drugs and neuroplasticity: insights into the treatment and neurobiology of schizophrenia. Biol Psychiatry 50:729–42
Kovács RÁ, Vadászi H, Bulyáki É, Török G, Tóth V, Mátyás D, Kun J, Hunyadi-Gulyás É, Fedor FZ, Csincsi Á, Medzihradszky K (2020) Identification of neuronal pentraxins as synaptic binding partners of C1q and the involvement of NP1 in synaptic pruning in adult mice. Front Immunol 11:599771
Krystal JH, Anticevic A, Yang GJ, Dragoi G, Driesen NR, Wang XJ, Murray JD (2017) Impaired tuning of neural ensembles and the pathophysiology of schizophrenia: a translational and computational neuroscience perspective. Biol Psychiatry 81:874–885
Lesh TA, Niendam TA, Minzenberg MJ, Carter CS (2011) Cognitive control deficits in schizophrenia: mechanisms and meaning. Neuropsychopharmacology 36:316–38
Li W, Lv L, Luo XJ (2022) In vivo study sheds new light on the dendritic spine pathology hypothesis of schizophrenia. Mol Psychiatry 27:1866–1868
Luchkina NV, Huupponen J, Clarke VR, Coleman SK, Keinanen K, Taira T, Lauri SE (2014) Developmental switch in the kinase dependency of long-term potentiation depends on expression of GluA4 subunit-containing AMPA receptors. Proc Natl Acad Sci U S A 111:4321–6
Manchia M, Piras IS, Huentelman MJ, Pinna F, Zai CC, Kennedy JL, Carpiniello B (2017) Pattern of gene expression in different stages of schizophrenia: Down-regulation of NPTX2 gene revealed by a meta-analysis of microarray datasets. Eur Neuropsychopharmacol 27:1054–1063
Mi R, Tang X, Sutter R, Xu D, Worley P, O’Brien RJ (2002) Differing mechanisms for glutamate receptor aggregation on dendritic spines and shafts in cultured hippocampal neurons. J Neurosci 22:7606–16
O’Brien RJ, Xu D, Petralia RS, Steward O, Huganir RL, Worley P (1999) Synaptic clustering of AMPA receptors by the extracellular immediate-early gene product Narp. Neuron 23:309–23
Osera C, Pascale A, Amadio M, Venturini L, Govoni S, Ricevuti G (2012) Pentraxins and Alzheimer’s disease: at the interface between biomarkers and pharmacological targets. Ageing Res Rev 11:189–98
Osimo EF, Beck K, Reis Marques T, Howes OD (2019) Synaptic loss in schizophrenia: a meta-analysis and systematic review of synaptic protein and mRNA measures. Mol Psychiatry 24:549–561
Pelkey KA, Barksdale E, Craig MT, Yuan X, Sukumaran M, Vargish GA, Mitchell RM, Wyeth MS, Petralia RS, Chittajallu R, Karlsson RM, Cameron HA, Murata Y, Colonnese MT, Worley PF, McBain CJ (2015) Pentraxins coordinate excitatory synapse maturation and circuit integration of parvalbumin interneurons. Neuron 85:1257–72
Romeo B, Brunet-Lecomte M, Martelli C, Benyamina A (2018) Kinetics of cytokine levels during antipsychotic treatment in schizophrenia: a meta-analysis. Int J Neuropsychopharmacol 21:828–836
Schmidt T, Samaras P, Frejno M, Gessulat S, Barnert M, Kienegger H, Krcmar H, Schlegl J, Ehrlich HC, Aiche S, Kuster B, Wilhelm M (2018) ProteomicsDB. Nucleic Acids Res 46:D1271–D1281
Schwarz DA, Barry G, Mackay KB, Manu F, Naeve GS, Vana AM, Verge G, Conlon PJ, Foster AC, Maki RA (2002) Identification of differentially expressed genes induced by transient ischemic stroke. Brain Res Mol Brain Res 101:12–22
Shi C, Kang L, Yao S, Ma Y, Li T, Liang Y, Cheng Z, Xu Y, Shi J, Xu X, Zhang C, Franklin DR, Heaton RK, Jin H, Yu X (2015) The MATRICS Consensus Cognitive Battery (MCCB): Co-norming and standardization in China. Schizophr Res 169:109–115
Sudhof TC (2008) Neuroligins and neurexins link synaptic function to cognitive disease. Nature 455:903–11
Sun J, Maller JJ, Guo L, Fitzgerald PB (2009) Superior temporal gyrus volume change in schizophrenia: a review on region of interest volumetric studies. Brain Res Rev 61:14–32
Swanson A, Wolf T, Sitzmann A, Willette AA (2018) Neuroinflammation in Alzheimer’s disease: pleiotropic roles for cytokines and neuronal pentraxins. Behav Brain Res 347:49–56
Uhlen M, Fagerberg L, Hallstrom BM, Lindskog C, Oksvold P, Mardinoglu A, Sivertsson A, Kampf C, Sjostedt E, Asplund A, Olsson I, Edlund K, Lundberg E, Navani S, Szigyarto CA, Odeberg J, Djureinovic D, Takanen JO, Hober S, Alm T, Edqvist PH, Berling H, Tegel H, Mulder J, Rockberg J, Nilsson P, Schwenk JM, Hamsten M, von Feilitzen K, Forsberg M, Persson L, Johansson F, Zwahlen M, von Heijne G, Nielsen J, Ponten F (2015) Proteomics. Tissue-based map of the human proteome. Science 347:1260419
van der Ende EL, Xiao M, Xu D, Poos JM, Panman JL, Jiskoot LC, Meeter LH, Dopper EG, Papma JM, Heller C, Convery R, Moore K, Bocchetta M, Neason M, Peakman G, Cash DM, Teunissen CE, Graff C, Synofzik M, Moreno F, Finger E, Sanchez-Valle R, Vandenberghe R, Laforce R Jr, Masellis M, Tartaglia MC, Rowe JB, Butler CR, Ducharme S, Gerhard A, Danek A, Levin J, Pijnenburg YA, Otto M, Borroni B, Tagliavini F, de Mendonca A, Santana I, Galimberti D, Seelaar H, Rohrer JD, Worley PF, van Swieten JC, Genetic Frontotemporal Dementia I (2020) Neuronal pentraxin 2: a synapse-derived CSF biomarker in genetic frontotemporal dementia. J Neurol Neurosurg Psychiatry 91:612–621
VanGuilder HD, Farley JA, Yan H, Van Kirk CA, Mitschelen M, Sonntag WE, Freeman WM (2011) Hippocampal dysregulation of synaptic plasticity-associated proteins with age-related cognitive decline. Neurobiol Dis 43:201–12
Vasek MJ, Garber C, Dorsey D, Durrant DM, Bollman B, Soung A, Yu J, Perez-Torres C, Frouin A, Wilton DK, Funk K, DeMasters BK, Jiang X, Bowen JR, Mennerick S, Robinson JK, Garbow JR, Tyler KL, Suthar MS, Schmidt RE, Stevens B, Klein RS (2016) A complement-microglial axis drives synapse loss during virus-induced memory impairment. Nature 534:538–43
Veleanu M, Urrieta-Chavez B, Sigoillot SM, Paul MA, Usardi A, Iyer K, Delagrange M, Doyle JP, Heintz N, Becamel C, Selimi F (2022) Modified climbing fiber/Purkinje cell synaptic connectivity in the cerebellum of the neonatal phencyclidine model of schizophrenia. Proc Natl Acad Sci U S A 119:e2122544119
Xiao MF, Roh SE, Zhou J, Chien CC, Lucey BP, Craig MT, Hayes LN, Coughlin JM, Leweke FM, Jia M, Xu D, Zhou W, Conover Talbot C Jr, Arnold DB, Staley M, Jiang C, Reti IM, Sawa A, Pelkey KA, McBain CJ, Savonenko A, Worley PF (2021) A biomarker-authenticated model of schizophrenia implicating NPTX2 loss of function. Sci Adv 7:eabf6935
Xiao MF, Xu D, Craig MT, Pelkey KA, Chien CC, Shi Y, Zhang J, Resnick S, Pletnikova O, Salmon D, Brewer J, Edland S, Wegiel J, Tycko B, Savonenko A, Reeves RH, Troncoso JC, McBain CJ, Galasko D, Worley PF (2017) NPTX2 and cognitive dysfunction in Alzheimer’s Disease. Elife 6. https://doi.org/10.7554/eLife.23798
Xu D, Hopf C, Reddy R, Cho RW, Guo L, Lanahan A, Petralia RS, Wenthold RJ, O’Brien RJ, Worley P (2003) Narp and NP1 form heterocomplexes that function in developmental and activity-dependent synaptic plasticity. Neuron 39:513–28
Xu Y, Hu Y, Geng Y, Zhao N, Jia C, Song H, Bai W, Guo C, Wang L, Ni Y, Qi X (2022) Pentraxin 3 depletion (PTX3 KD) inhibited myocardial fibrosis in heart failure after myocardial infarction. Aging 14:4036–4049
Yamamoto J, Suh J, Takeuchi D, Tonegawa S (2014) Successful execution of working memory linked to synchronized high-frequency gamma oscillations. Cell 157:845–57
Yi JM, Dhir M, Van Neste L, Downing SR, Jeschke J, Glockner SC, de Freitas Calmon M, Hooker CM, Funes JM, Boshoff C, Smits KM, van Engeland M, Weijenberg MP, Iacobuzio-Donahue CA, Herman JG, Schuebel KE, Baylin SB, Ahuja N (2011) Genomic and epigenomic integration identifies a prognostic signature in colon cancer. Clin Cancer Res 17:1535–45
Acknowledgements
The authors thank the patients and the healthy volunteers for their participation, as well as the physicians and technicians who helped us collect clinical data in the Second Affiliated Hospital of Xinxiang Medical University. We thank the International Science Editing (http://www.internationalscienceediting.com) for editing our manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (81971252, U1904130, 82171498, and 82001407), Major science and technology projects of Henan Province (201300310200), Science and technology research project of Henan Province (222102310130), Open program of Henan clinical research center for mental disorders (2021-zxkfkt-006), Medical science and technology research project of Henan Province (LHGJ20210541, LHGJ20220637, and LHGJ20190475), Youth project of medical science and technology of Henan Province (SBGJ202103094), and Practical Innovation Training Program for University Students of Henan Province (202210472017).
Author information
Authors and Affiliations
Contributions
LL, WL, WY, and JZ designed the study and wrote the protocol. JZ and XL managed the literature searches and analysis. XW, YY, HS, JYZ (Jingyuan Zhao), JHZ (Jianhong Zhang), and SD conducted sample selection. AN, YH, QL, LZ, TC, BL, XL, and JZ undertook the data management and statistical analysis. JZ wrote the first draft of the manuscript. JZ and WL revised the manuscript. All authors contributed to and have approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, J., Li, X., Wang, X. et al. Levels of neuronal pentraxin 2 in plasma is associated with cognitive function in patients with schizophrenia. Psychopharmacology 241, 865–874 (2024). https://doi.org/10.1007/s00213-023-06515-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-023-06515-3