Abstract
Rationale
Cabergoline (CAB) is an ergot derivative typically prescribed for the treatment of hyperprolactinemia. It suppresses the release of prolactin through agonist actions on dopamine (DA) D2 receptors; however, it possesses binding affinity for other DA and 5-HT receptors. Side effects that exacerbate valvular heart disease can occur with high doses.
Objective
The present study examined the acute, subchronic, and chronic dose–response effects of CAB and a derivative dimethylcabergoline (DMC) which acts as an antagonist instead of agonist at 5-HT 2B receptors, on appetitive and consummatory sexual behaviors of male rats.
Methods
CAB (0, 0.03, 0.15, or 0.3 mg/kg/ml) was administered daily to sexually experienced male rats (N = 10/dose) by oral gavage for a total of 68 days. Sexual behavior was tested every 4 days during this period for a total of 16 trials. On the 17th trial, rats were administered their dose of CAB, and 4 h after were overdosed with sodium pentobarbital, perfused intracardially, and their brains processed for Fos immunohistochemistry. DMC (0, 0.03, 0.15, 0.3 mg/kg/ml) was administered daily to sexually experienced male rats (N = 10/dose) by oral gavage for a total of 36 days. Sexual behavior was tested every 4 days for a total of 9 trials.
Results
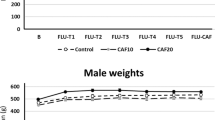
CAB increased anticipatory level changes, intromissions, and ejaculations significantly across all timepoints, with the medium and high doses being most potent. The medium and high doses also increased Fos protein significantly within the medial preoptic area, whereas in the nucleus accumbens shell, the low and medium doses decreased Fos protein but the high dose increased it significantly from control. Similar to CAB, the medium and high doses of DMC increased the number of ejaculations significantly. Rats in all drug dose groups appeared healthy for the duration of the experiments.
Conclusions
Both CAB and DMC facilitate ejaculations, and CAB further facilitates measures of anticipatory sexual motivation and intromissions. These data suggest that both could be used as treatments for sexual arousal disorders and ejaculation/orgasm disorders with little or no untoward side effects at low doses.



Similar content being viewed by others
References
Afonso VM, Mueller D, Stewart J, Pfaus JG (2009) Amphetamine pretreatment facilitates appetitive sexual behaviors in the female rat. Psychopharmacology 205:35–43
Ahlenius S, Engel J, Larsson K, Svensson L (1982) Effects of pergolide and bromocriptine on male rat sexual behavior. J Neural Transm 54:165–170
Allers KA, Dremencov E, Ceci A, Flik G, Ferger B, Cremers TI, Ittrich C, Sommer B (2010) Acute and repeated flibanserin administration in female rats modulates monoamines differentially across brain areas: a microdialysis study. J Sex Med 7:1757–1767
Antonini A, Poewe W (2007) Fibrotic heart-valve reactions to dopamine-agonist treatment in Parkinson’s disease. Lancet Neurol 6:826–829
Arnow BA, Millheiser L, Garrett A, Lake Polan M, Glover GH, Hill KR, Lightbody A, Watson C, Banner L, Smart T, Buchanan T, Desmond JE (2009) Women with hypoactive sexual desire disorder compared to normal females: a functional magnetic resonance imaging study. Neuroscience 158:484–502
Baum MJ, Everitt BJ (1992) Increased expression of c-fos in the medial preoptic area after mating in male rats: role of afferent inputs from the medial amygdala and midbrain central tegmental field. Neuroscience 50:627–646
Berg KA, Harvey JA, Spampinato U, Clarke WP (2008) Physiological and therapeutic relevance of constitutive activity of 5-HT 2A and 5-HT 2C receptors for the treatment of depression. Prog Brain Res 172:287–305
Bhattacharyya S, Schapira AH, Mikhailidis DP, Davar J (2009) Drug-induced fibrotic valvular heart disease. Lancet 374:577–585
Bitran D, Thompson JT, Hull EM, Sachs BD (1989) Quinelorane (LY163502), a D2 dopamine receptor agonist, facilitates seminal emission, but inhibits penile erection in the rat. Pharmacol Biochem Behav 34:453–458
Blackburn JR, Pfaus JG, Phillips AG (1992) Dopamine functions in appetitive and defensive behaviours. Prog Neurobiol 39:247–279
Buvat J (2003) Hyperprolactinemia and sexual function in men: a short review. Int J Impot Res 15:373–377
Cabilio S (1996) Behavioral scoring program Unpublished software. Concordia University, Montréal
Cantor JM, Binik YM, Pfaus JG (1999) Chronic fluoxetine inhibits sexual behavior in the male rat: reversal with oxytocin. Psychopharmacology 144:355–362
Colao A, di Sarno A, Pivonello R, di Somma C, Lombardi G (2002) Dopamine receptor agonists for treating prolactinomas. Expert Opin Investig Drugs 11:787–800
Coolen LM, Peters HJ, Veening JG (1998) Anatomical interrelationships of the medial preoptic area and other brain regions activated following male sexual behavior: a combined fos and tract-tracing study. J Comp Neurol 397:421–435
Corona G, Rastrelli G, Bianchi N, Sparano C, Sforza A, Vignozzi L, Maggi M (2023) Hyperprolactinemia and male sexual function: focus on erectile dysfunction and sexual desire. Int J Impot Res. https://doi.org/10.1038/s41443-023-00717-1
Cosyns B, Droogmans S, Rosenhek R, Lancellotti P (2013) Drug-induced valvular heart disease. Heart 99:7–12
Dalló J, Lekka N, Knoll J (1986) The ejaculatory behavior of sexually sluggish male rats treated with (-)deprenyl, apomorphine, bromocriptine and amphetamine. Pol J Pharmacol Pharm 38:251–255
Deebel NA, Thai K, Ramasamy R, Terlecki RP (2022) Understanding the dopaminergic pathway relative to men’s sexual dysfunction in patients with Parkinson’s disease: a narrative review with implications for future research. Int J Impot Res. https://doi.org/10.1038/s41443-022-00656-3
Devroye C, Cathala A, Di Marco B, Caraci F, Drago F, Piazza PV, Spampinato U (2015) Central serotonin(2B) receptor blockade inhibits cocaine-induced hyperlocomotion independently of changes of subcortical dopamine outflow. Neuropharmacology 97:329–337
Doherty PC, Bartke A, Smith MS (1981) Differential effects of bromocriptine treatment on LH release and copulatory behavior in hyperprolactinemic male rats. Horm Behav 15:436–450
Dosa PI, Ward T, Walters MA, Kim SW (2013) Synthesis of novel analogs of cabergoline: improving cardiovascular safety by removing 5-HT2B receptor agonism. ACS Med Chem Lett 4:254–258
Ferrara N, Bernardini R, Nicoletti F, Drago F, Matera M, Ceravolo A, Scapagnini U (1985) Prolactin influences sexual behavior during aging. J Endocrinol Invest 8(Suppl 2):23–32
Fitzgerald LW, Burn TC, Brown BS, Patterson JP, Corjay MH, Valentine PA, Sun JH, Link JR, Abbaszade I, Hollis JM, Largent BL, Hartig PR, Hollis GF, Meunier PC, Robichaud AJ, Robertson DW (2000) Possible role of valvular serotonin 5-HT(2B) receptors in the cardiopathy associated with fenfluramine. Mol Pharmacol 57:75–81
Gebeyehu NA, Gesese MM, Tegegne KD, Kebede YS, Kassie GA, Mengstie MA, Zemene MA, Bantie B, Feleke SF, Dejenie TA, Abebe EC, Anley DT, Dessie AM, Bayih WA, Adela GA (2023) Global prevalence of sexual dysfunction among diabetic patients from 2008 to 2022: systematic review and meta-analysis. Metabol Open 18:100247
Gelez H, Clement P, Compagnie S, Gorny D, Laurin M, Allers K, Sommer B, Giuliano F (2013) Brain neuronal activation induced by flibanserin treatment in female rats. Psychopharmacology 230:639–652
Giuliano F, Rampin O (2004) Neural control of erection. Physiol Behav 83:189–201
González Cautela BV, Quintana GR, Akerman J, Pfaus JG (2021) Acute caffeine reverses the disruptive effects of chronic fluoxetine on the sexual behavior of female and male rats. Psychopharmacology 238:755–764
Graham MD, Pfaus JG (2012) Differential effects of dopamine antagonists infused to the medial preoptic area on the sexual behavior of female rats primed with estrogen and progesterone. Pharmacol Biochem Behav 102:532–539
Graham MD, Gardner Gregory J, Hussain D, Brake WG, Pfaus JG (2015) Ovarian steroids alter dopamine receptor populations in the medial preoptic area of female rats: implications for sexual motivation, desire, and behaviour. Eur J Neurosci 42:3138–3148
Harding SM, McGinnis MY (2005) Microlesions of the ventromedial nucleus of the hypothalamus: effects on sociosexual behaviors in male rats. Behav Neurosci 119:1227–1234
Harding SM, McGinnis MY (2004) Androgen receptor blockade in the MPOA or VMN: effects on male sociosexual behaviors. Physiol Behav 81:671–680
Hollander AB, Pastuszak AW, Hsieh TC, Johnson WG, Scovell JM, Mai CK, Lipshultz LI (2016) Cabergoline in the treatment of male orgasmic disorder - a retrospective pilot analysis. Sex Med 4:e28–e33
Holt RI, Barnett AH, Bailey CJ (2010) Bromocriptine: old drug, new formulation and new indication. Diabetes Obes Metab 12:1048–1057
Horvath J, Fross RD, Kleiner-Fisman G, Lerch R, Stalder H, Liaudat S, Raskoff WJ, Flachsbart KD, Rakowski H, Pache JC, Burkhard PR, Lang AE (2004) Severe multivalvular heart disease: a new complication of the ergot derivative dopamine agonists. Mov Disord 19:656–662
Hull EM, Dominguez JM (2007) Sexual behavior in male rodents. Horm Behav 52:45–55
Ismail N, Laroche C, Girard-Bériault F, Ménard S, Greggain-Mohr JA, Pfaus JG (2010) Conditioned ejaculatory preference in male rats paired with haloperidol-treated females. Physiol Behav 100:116–121
Kippin TE, Cain SW, Pfaus JG (2003) Estrous odors and sexually-conditioned neutral odors activate independent neural pathways in the male rat. Neuroscience 117:971–979
Kleitz-Nelson HK, Dominguez JM, Cornil CA, Ball GF (2010) Is sexual motivational state linked to dopamine release in the medial preoptic area? Behav Neurosci 124:300–304
Konstantinos S, Vikelis M, Rapoport A (2020) Acute care treatment of migraine. J Neuroophthalmol 40:472–484
Krüger TH, Haake P, Haverkamp J, Krämer M, Exton MS, Saller B, Leygraf N, Hartmann U, Schedlowski M (2003) Effects of acute prolactin manipulation on sexual drive and function in males. J Endocrinol 179:357–365
Krüger THC, Keil L, Jung S, Kahl KG, Wittfoth M, Leeners B, Hartmann U (2018) Lack of increase in sexual drive and function after dopaminergic stimulation in women. J Sex Marital Ther 44:61–72
Kvernmo T, Härtter S, Burger E (2006) A review of the receptor-binding and pharmacokinetic properties of dopamine agonists. Clin Ther 28:1065–1078
Larner AJ (2008) Transient acute neurologic sequelae of sexual activity: headache and amnesia. J Sex Med 5:284–288
Mantegani S, Brambilla E, Varasi M (1999) Ergoline derivatives: receptor affinity and selectivity. Farmaco 54:288–296
Mayer J, Sato A, Kiupel M, DeCubellis J, Donnelly T (2011) Extralabel use of cabergoline in the treatment of a pituitary adenoma in a rat. J Am Vet Med Assoc 239:656–660
Melis MR, Sanna F, Argiolas A (2022) Dopamine, erectile function and male sexual behavior from the past to the present: a review. Brain Sci 12:826
Miyagawa Y, Tsujimura A, Fujita K, Matsuoka Y, Takahashi T, Takao T, Takada S, Matsumiya K, Osaki Y, Takasawa M, Oku N, Hatazawa J, Kaneko S, Okuyama A (2007) Differential brain processing of audiovisual sexual stimuli in men: comparative positron emission tomography study of the initiation and maintenance of penile erection during sexual arousal. Neuroimage 36:830–842
Nickel M, Moleda D, Loew T, Rother W, Pedrosa Gil F (2007) Cabergoline treatment in men with psychogenic erectile dysfunction: a randomized, double-blind, placebo-controlled study. Int J Impot Res 19:104–107
Parada M, Chamas L, Censi S, Coria-Avila G, Pfaus JG (2010) Clitoral stimulation induces conditioned place preference and Fos activation in the rat. Horm Behav 57:112–118
Paredes RG, Lopez ME, Baum MJ (1998) Testosterone augments neuronal Fos responses to estrous odors throughout the vomeronasal projection pathway of gonadectomized male and female rats. Horm Behav 33:48–57
Pattij T, Olivier B, Waldinger MD (2005) Animal models of ejaculatory behavior. Curr Pharm Des 11(31):4069–4077
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates. Elsevier, New York
Pellis SM, Teitelbaum P, Meyer ME (1990) Labyrinthine and visual involvement in the dorsal immobility response of adult rats. Behav Brain Res 39:197–204
Pfaus JG (2009) Pathways of sexual desire. J Sex Med 6:1506–1533
Pfaus JG, Heeb MM (1997) Implications of immediate-early gene induction in the brain following sexual stimulation of female and male rodents. Brain Res Bull 44:397–407
Pfaus JG, Phillips AG (1991) Role of dopamine in anticipatory and consummatory aspects of sexual behavior in the male rat. Behav Neurosci 105:727–743
Pfaus JG, Giuliano F, Gelez H (2007) Bremelanotide: an overview of preclinical CNS effects on female sexual function. J Sex Med 4(Suppl 4):269–279
Pfaus JG, Mendelson SD, Phillips AG (1990a) A correlational and factor analysis of anticipatory and consummatory measures of sexual behavior in the male rat. Psychoneuroendocrinology 15:329–340
Pfaus JG, Damsma G, Nomikos GG, Wenkstern DG, Blaha CD, Phillips AG, Fibiger HC (1990b) Sexual behavior enhances central dopamine transmission in the male rat. Brain Res 530:345–348
Pivonello R, Auriemma RS, Delli Veneri A, Dassie F, Lorusso R, Ragonese M, Liotta M, Sala E, Zarino B, Lai E, Urbani C, Bogazzi F, Mantovani G, Cannavò S, Maffei P, Chiodini P, Colao A (2022) Global psychological assessment with the evaluation of life and sleep quality and sexual and cognitive function in a large number of patients with acromegaly: a cross-sectional study. Eur J Endocrinol 187:823–845
Porter MC, Appiah-Kubf LS, Chaudhuri KR (2002) Treatment of Parkinson’s disease and restless legs syndrome with cabergoline, a long-acting dopamine agonist. Int J Clin Pract 56:468–474
Pradeep R, Sundarmurthy H, Karan V, Kulkarni P (2019) Prevalence and predictors of femalesexual dysfunction in migraine. Ann Indian Acad Neurol 22:291–294
Rubio-Casillas A, Rodríguez-Quintero CM, Rodríguez-Manzo G, Fernández-Guasti A (2015) Unraveling the modulatory actions of serotonin on male rat sexual responses. Neurosci Biobehav Rev 55:234–246
Sachs BD, Barfield RJ (1976) Functional analysis of masculine copulatory behavior in the rat. Adv Stud Behav 7:91–154
Simonsen U, Comerma-Steffensen S, Andersson KE (2016) Modulation of dopaminergic pathways to treat erectile dysfunction. Basic Clin Pharmacol Toxicol 119(Suppl 3):63–74
Smith SR, Weissman NJ, Anderson CM, Sanchez M, Chuang E, Stubbe S, Bays H, Shanahan WR (2010) Multicenter, placebo-controlled trial of lorcaserin for weight management. N Engl J Med 363:245–256
Stolzenberg DS, Numan M (2011) Hypothalamic interaction with the mesolimbic DA system in the control of the maternal and sexual behaviors in rats. Neurosci Biobehav Rev 35:826–847
Tobiansky DJ, Roma PG, Hattori T, Will RG, Nutsch VL, Dominguez JM (2013) The medial preoptic area modulates cocaine-induced activity in female rats. Behav Neurosci 127:293–302
Vallette S, Serri K, Serri O (2010) Cabergoline therapy for prolactinomas: is valvular heart disease a real safety concern? Expert Rev Cardiovasc Ther 8:49–54
Wang AT, Mullan RJ, Lane MA, Hazem A, Prasad C, Gathaiya NW, Fernández-Balsells MM, Bagatto A, Coto-Yglesias F, Carey J, Elraiyah TA, Erwin PJ, Gandhi GY, Montori VM, Murad MH (2012) Treatment of hyperprolactinemia: a systematic review and meta-analysis. Syst Rev 1:33
Yonezawa A, Yoshizumi M, Ebiko M, Ise SN, Watanabe C, Mizoguchi H, Kimura Y, Sakurada S (2008) Ejaculatory response induced by a 5-HT2 receptor agonist m-CPP in rats: differential roles of 5-HT2 receptor subtypes. Pharmacol Biochem Behav 88:367–373
Yonezawa A, Yoshizumi M, Ise SN, Watanabe C, Mizoguchi H, Furukawa K, Tsuru H, Kimura Y, Kawatani M, Sakurada S (2009) Synergistic actions of apomorphine and m-chlorophenylpiperazine on ejaculation, but not penile erection in rats. Biomed Res 30:71–78
Yoshizumi M, Yonezawa A, Kimura Y, Watanabe C, Sakurada S, Mizoguchi H (2021) Central mechanisms of apomorphine and m-Chlorophenylpiperazine on synergistic action for ejaculation in rats. J Sex Med 18:231–239
Zanettini R, Antonini A, Gatto G, Gentile R, Tesei S, Pezzoli G (2007) Valvular heart disease and the use of dopamine agonists for Parkinson’s disease. N Engl J Med 356:39–46
Acknowledgements
The authors would like to thank Noeli Devoto and Brunella González Cautela for help conducting the behavioral experiments, and Dr. Ellen Zakreski for help with the figures.
Funding
This work was supported in part by a Discovery grant from the Natural Sciences and Engineering Research Council (NSERC) of Canada (OGP-138878) to JGP and by an infrastructure grant from Fonds de la recherche en santé du Québec (FRSQ F00757) to the Center for Studies in Behavioral Neurobiology at Concordia University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The authors declare that all animal procedures conformed to the guidelines of the Canadian Council for Animal Care. All procedures were approved by the Concordia University Animal Research Ethics Committee (Protocol #30000300 to JGP).
Conflict of interest
Two of the authors, PID and SWK, are owners of the patent for DMC.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pfaus, J.G., Antonie, R.A., Dosa, P.I. et al. Effects of cabergoline and dimethylcabergoline on the sexual behavior of male rats. Psychopharmacology 241, 717–726 (2024). https://doi.org/10.1007/s00213-023-06501-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-023-06501-9