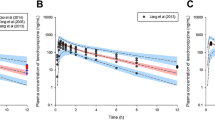
Abstract
This study aimed to quantify and explain inter-subject variability in morniflumate pharmacokinetics and identify effective covariates through population pharmacokinetics modeling. Models were constructed using bioequivalence pharmacokinetics results from healthy Korean males and individual physiological and biochemical parameters. Additionally, we incorporated previously reported pharmacokinetics results of niflumic acid, a major active metabolite of morniflumate, to extend the established population pharmacokinetics model and predict niflumic acid pharmacokinetics. Moreover, we used quantitative reports of leukotriene B4 (LTB4) synthesis inhibition in response to niflumic acid exposure to predict drug efficacy using Sigmoid Emax model. Population pharmacokinetics profiles of morniflumate were described using a multi-absorption (5-sequential) two-compartment model, and analysis of inter-individual variability suggested that volume of distribution in peripheral compartment was correlated with body mass index (BMI). Model simulation results showed that individuals with lower BMI had higher plasma concentrations of morniflumate and niflumic acid, resulting in increased and sustained inhibition of LTB4 synthesis. Under steady-state conditions, average plasma concentrations of morniflumate and niflumic acid were 2.66–2.68 times higher in group with a BMI of 17.36 kg/m2 compared to the group with a BMI of 28.41 kg/m2. Additionally, inhibition of LTB4 synthesis was 1.02 times higher in group with a BMI of 17.36 kg/m2 compared to group with a BMI of 28.41 kg/m2, and the fluctuation was significantly reduced from 6.06 to 0.01%. These findings suggest that the concentration of active metabolite in plasma following morniflumate exposure was lower in the obese group compared to the normal group, thus potentially reducing the drug’s efficacy.







Similar content being viewed by others
Data availability
All data and related materials are accessible in this manuscript and supplementary materials.
Abbreviations
- BMI:
-
Body mass index
- LTB4 :
-
Leukotriene B4
- NSAID:
-
Nonsteroidal anti-inflammatory drug
- TXB2 :
-
Thromboxane B2
- IIV:
-
Inter-individual variability
- T lag :
-
Lag-time
- NCA:
-
Noncompartment analysis
- AIC:
-
Akaike’s information criterion
- \(-\) 2LL:
-
Negative log likelihood
- GOF:
-
Goodness of fit
- VPC:
-
Visual predictive check
- NPDE:
-
Normalized prediction distribution error
- CWRES:
-
Conditional weighted residuals
- QQ:
-
Quantile-quantile
- WRES:
-
Weighted residuals
- OFV:
-
Objective function value
- BSA:
-
Body surface area
- GFR:
-
Glomerular filtration rate
References
Al-Naamani N, Maarouf O, Wilt J, D’Ovidio F, Sonett J, Arcasoy S, Nickolas T, Lederer D, Kawut S (2008) 25: the modification of diet in renal disease (MDRD) formula predicts kidney failure after lung transplantation. J Heart Lung Transplant 27:S69
BMI Oc (1998) Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults. NOE Initiative 6:51S-209S
Cho HY, Park GK, Lee YB (2013) Simultaneous determination of morniflumate and its major active metabolite, niflumic acid, in human plasma by high-performance liquid chromatography in stability and pharmacokinetic studies. Biomed Chromatogr 27:1438–1443
Civelli M, Vigano T, Acerbi D, Caruso P, Giossi M, Bongrani S, Folco G (1991) Modulation of arachidonic acid metabolism by orally administered morniflumate in man. Agents Actions 33:233–239
Cremonesi G, Cavalieri L (2015) Efficacy and safety of morniflumate for the treatment of symptoms associated with soft tissue inflammation. Int J Med Res 43:290–302
DrugBank (2023) Morniflumate. https://go.drugbank.com/drugs/DB09285. Accessed 24 May 2023
Dutta S, Matsumoto Y, Ebling WF (1996) Is it possible to estimate the parameters of the sigmoid Emax model with truncated data typical of clinical studies? J Pharm Sci 85:232–239
El Edelbi R, Lindemalm S, Eksborg S (2012) Estimation of body surface area in various childhood ages–validation of the Mosteller formula. Acta Paediatr 101:540–544
Hermsen ED, Maiefski MM, Florescu MC, Qiu F, Rupp ME (2009) Comparison of the modification of diet in renal disease and Cockcroft-Gault equations for dosing antimicrobials. Pharmacotherapy 29:649–655
Hotta M, Li Y, Anme T, Ushijima H (2005) Risk factors for low Kaup index among children in rural ethnic minority areas of Yunnan, China. Pediatr Int 47:147–153
Jang J-H, Jeong S-H, Lee Y-B (2023a) Establishment of a fexofenadine population pharmacokinetic (PK)–pharmacodynamic (PD) model and exploration of dosing regimens through simulation. J Pharm Investig 53:427–441
Jang J-H, Jeong S-H, Lee Y-B (2023b) Quantitative assessment of the relevance of organic-anion-transporting-polypeptide 1B1 and 2B1 polymorphisms in fexofenadine pharmacokinetic variants via pharmacometrics. J Pharm Anal 13:660–672
Jang JH, Jeong SH, Lee YB (2023c) Dosage exploration of meloxicam according to CYP2C9 genetic polymorphisms based on a population pharmacokinetic-pharmacodynamic model. Pharmacotherapy 43:145–157
Jeong S-H, Jang J-H, Cho H-Y, Lee Y-B (2021) Population pharmacokinetic analysis of cefaclor in healthy Korean subjects. Pharmaceutics 13:754
Jeong S-H, Jang J-H, Lee Y-B (2022) Population pharmacokinetic analysis of lornoxicam in healthy Korean males considering creatinine clearance and CYP2C9 genetic polymorphism. J Pharm Investig 52:109–127
Knechtle P, Shapiro S, Morrissey I, De Piano C, Belley A (2021) Sigmoid E max modeling to define the fixed concentration of enmetazobactam for MIC testing in combination with cefepime. Antimicrob Agents Chemother 65:e00926-e921
Kucherenko YV, Lang F (2014) Niflumic acid affects store-operated Ca 2+-permeable (SOC) and Ca 2+-dependent K+ and Cl− ion channels and induces apoptosis in K562 cells. J Membr Biol 247:627–638
Lee S-H, Han C-D, Yang I-H, Ha C-W (2011) Prescription pattern of NSAIDs and the prevalence of NSAID-induced gastrointestinal risk factors of orthopaedic patients in clinical practice in Korea. J Korean Med Sci 26:561–567
Matheu V, Sierra Z, Gracia M, Caloto M, Alcazar M, Martinez M, Zapatero L (1998) Morniflumate-induced urticaria-angioedema. Allergy 53:812–813
Meher CP, Purohit D, Kumar A, Singh R, Dubey A (2022) An updated review on morpholine derivatives with their pharmacological actions. Int J Health Sci 6:2218–2249
Mero F, Nettis E, Aloia A, Di Leo E, Ferrannini A, Vacca A (2013) Short-term tolerability of morniflumate in patients with cutaneous hypersensitivity reactions to non-steroidal anti-inflammatory drugs. Int J Immunopathol Pharmacol 26:247–250
Paleari D, Cavalieri L (2022) Morniflumate in the treatment of upper airways affections in adults: a review and meta-analysis. Minerva Med 113:853–863
Savic RM, Jonker DM, Kerbusch T, Karlsson MO (2007) Implementation of a transit compartment model for describing drug absorption in pharmacokinetic studies. J Pharmacokinet Pharmacodyn 34:711–726
Schiantarelli P, Cadel S, Acerbi D (1984) A gastroprotective anti-inflammatory agent: the β-morpholinoethyl ester of niflumic acid (morniflumate). Agents Actions 14:247–256
Talath S, Gadad AK (2006) Synthesis, stability studies, anti-inflammatory activity and ulcerogenicity of morpholinoalkyl ester prodrugs of niflumic acid. Arzneimittelforschung 56:744–752
Funding
This work was supported by the Medical Research Program of Handok Jeseok Foundation.
Author information
Authors and Affiliations
Contributions
Seung-Hyun Jeong: Conceptualization, investigation, methodology, writing-original draft, writing-review and editing, software, data analysis, and visualization; Ji-Hun Jang: Conceptualization, investigation, methodology, writing-review and editing, software, and data analysis; Yong-Bok Lee: Conceptualization, methodology, writing-review and editing, and supervision. All authors reviewed the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
Clinical studies were conducted in accordance with the Rules of Good Clinical Practice and the revised Declaration of Helsinki for Biomedical Research with Human Subjects. All subjects had no prior history of hypersensitivity or related reactions to steroid drugs and were physically normal. In addition, healthy adults aged 19–55 years have no history of digestive, liver, kidney, cardiovascular, central nervous system, endocrine, or blood diseases in the past and have not been taking any other drugs. All subjects underwent a physical examination, clinical screening, complete blood count, urinalysis, and blood chemistry analysis prior to participation in this clinical study to confirm their physical health. The clinical study protocol used in this study was thoroughly reviewed and approved by the Institutional Review Board of the Institute of Bioequivalence and Bridging Study, Chonnam National University, Gwangju, Republic of Korea (bioequivalence study permit numbers: 100329; 01.28.2010).
Consent to participate
All subjects provided written informed consent prior to their participation in the bioequivalence and pharmacokinetics studies.
Consent for publication
All data were anonymized and participants were informed that the results of this study may be subject to publication and presentation in meetings.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jeong, SH., Jang, JH. & Lee, YB. Modeling population pharmacokinetics of morniflumate in healthy Korean men: extending pharmacometrics analysis to niflumic acid, its major active metabolite. Naunyn-Schmiedeberg's Arch Pharmacol 397, 843–856 (2024). https://doi.org/10.1007/s00210-023-02640-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02640-0