Abstract
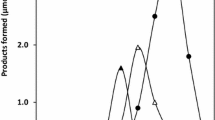
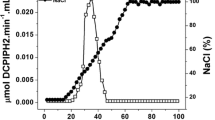
A membrane-bound NADH oxidase of an anaerobic alkaliphile, M-12 (a strain of Amphibacillus sp.), was solubilized with decanoyl N-methylglucamide and purified by chromatography on DEAE-Sepharose and hydroxyapatite. The purified enzyme appears to consist of a single polypeptide component with an apparent molecular mass of 56 kDa. The enzyme catalyzed the oxidation of NADH with the formation of H2O2 and exhibited a specific activity of 46 μmol NADH min–1 (mg protein)–1. NADPH did not serve as a substrate for the enzyme. The K m for NADH was estimated to be 0.05 mM. The enzyme exhibited a pH dependence for activity, with a pH optimum at approximately 9.5. The enzyme required a high concentration of salt and exhibited maximum activity in the presence of 600 mM NaCl.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 3 August 1998 / Accepted: 23 December 1998
Rights and permissions
About this article
Cite this article
Hamada, T., Wakagi, T., Shiba, H. et al. Purification and properties of the membrane-bound NADH oxidase of a facultatively anaerobic alkaliphile. Arch Microbiol 171, 237–242 (1999). https://doi.org/10.1007/s002030050705
Issue Date:
DOI: https://doi.org/10.1007/s002030050705