Abstract
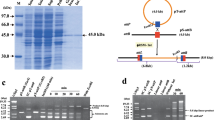
MrpA is the multimer resolution protein of the Streptomyces coelicolor A3(2) plasmid SCP2*. Previously, MrpA was found to significantly increase the stability of SCP2*-derived plasmids in Streptomyces lividans. The present report gives a functional characterization of MrpA. A sequence alignment revealed that MrpA shares highly conserved residues with members of the tyrosine recombinase family. After overexpression and Strep-tag purification, a DNase I footprint analysis and a gel mobility shift assay allowed for the identification of the 36-bp MrpA binding site mrpS. The mrpS site shows the configuration typical for tyrosine recombinases and contains two MrpA binding sites. The activity of MrpA was explored in vivo in E. coli cells and in vitro using purified MrpA. Depending on the position and orientation of the mrpS sites, three activities were detected: integration, resolution, and inversion. No accessory sites or proteins were required. Substitution of the conserved tyrosine (Y354F) by site-directed mutagenesis resulted in a complete loss of recombination activity but it still allowed the binding of MrpA to mrpS. The results define MrpA as a new site-specific tyrosine recombinase that acts with mrpS. In addition, we suggest that Y354 provides the nucleophile for DNA cleavage during recombination.










Similar content being viewed by others
References
Abremski K, Hoess R (1984) Bacteriophage P1 site-specific recombination. Purification and properties of the Cre recombinase protein. J Biol Chem 259:1509–1514
Altenbuchner J, Viell P, Pelletier I (1992) Positive selection vectors based on palindromic DNA sequences. Methods Enzymol 216:457–466
Arnold W, Pühler A (1988) A family of high-copy-number plasmid vectors with single end-label sites for rapid nucleotide sequencing. Gene 70:171–179
Azaro MA, Landy A (2002) The integration/excision cycle of lambda and other bacteriophages. In: Craig NL, Craigie R, Gellert M, Lambowitz A (eds) Mobile DNA II. ASM Press, Washington, pp 117–148
Bernard P, Couturier M (1992) Cell killing by the F plasmid CcdB protein involves poisoning of DNA-topoisomerase II complexes. J Mol Biol 226:735–745
Bernard P, Gabant P, Bahassi EM, Couturier M (1994) Positive-selection vectors using the F plasmid ccdB killer gene. Gene 148:71–74
Bibb MJ, Hopwood DA (1981) Genetic studies of the fertility plasmid SCP2 and its SCP2* variants in streptomyces coelicolor A3(2). J Gen Microbiol 126:427–442
Bibb MJ, Freeman RF, Hopwood DA (1977) Physical and genetical characterisation of a second sex factor, SCP2, for Streptomyces coelicolor A3(2). Mol Gen Genet 154:155–166
Bibb MJ, Schottel JL, Cohen SN (1980) A DNA cloning system for interspecies gene transfer in antibiotic-producing Streptomyces. Nature 284:526–531
Bolivar F et al (1977) Construction and characterization of new cloning vehicles. II. A multipurpose cloning system. Gene 2:95–113
Brasch MA, Pettis GS, Lee SC, Cohen SN (1993) Localization and nucleotide sequences of genes mediating site-specific recombination of the SLP1 element in Streptomyces lividans. J Bacteriol 175:3067–3074
Chang AC, Cohen SN (1978) Construction and characterization of amplifiable multicopy DNA cloning vehicles derived from the P15A cryptic miniplasmid. J Bacteriol 134:1141–1156
Chen Y, Narendra U, Iype LE, Cox MM, Rice PA (2000) Crystal structure of a Flp recombinase-Holliday junction complex: assembly of an active oligomer by helix swapping. Mol Cell 6:885–897
Esposito D, Scocca JJ (1997) The integrase family of tyrosine recombinases: evolution of a conserved active site domain. Nucleic Acids Res 25:3605–3614
Fried M, Crothers DM (1981) Equilibria and kinetics of lac repressor-operator interactions by polyacrylamide gel electrophoresis. Nucleic Acids Res 9:6505–6525
Garner MM, Revzin A (1981) A gel electrophoresis method for quantifying the binding of proteins to specific DNA regions: application to components of the Escherichia coli lactose operon regulatory system. Nucleic Acids Res 9:3047–3060
Grainge I, Jayaram M (1999) The integrase family of recombinase: organization and function of the active site. Mol Microbiol 33:449–456
Grainge I, Sherratt DJ (2007) Site-specific recombination. In: Aguilera A, Rothstein R (eds) Molecular genetics of recombination. Springer, Berlin, pp 443–463
Guo F, Gopaul DN, van Duyne GD (1997) Structure of Cre recombinase complexed with DNA in a site-specific recombination synapse. Nature 389:40–46
Hagege J, Boccard F, Smokvina T, Pernodet JL, Friedmann A, Guerineau M (1994) Identification of a gene encoding the replication initiator protein of the Streptomyces integrating element, pSAM2. Plasmid 31:166–183
Haug I, Weissenborn A, Brolle D, Bentley S, Kieser T, Altenbuchner J (2003) Streptomyces coelicolor A3(2) plasmid SCP2*: deductions from the complete sequence. Microbiology 149:505–513
Hickman AB, Waninger S, Scocca JJ, Dyda F (1997) Molecular organization in site-specific recombination: the catalytic domain of bacteriophage HP1 integrase at 2.7 A resolution. Cell 89:227–237
Jeske M, Altenbuchner J (2010) The Escherichia coli rhamnose promoter rhaP(BAD) is in Pseudomonas putida KT2440 independent of Crp-cAMP activation. Appl Microbiol Biotechnol 85:1923–1933
Katoh K, Kuma K, Toh H, Miyata T (2005) MAFFT version 5: improvement in accuracy of multiple sequence alignment. Nucleic Acids Res 33:511–518
Kieser T, Bibb MJ, Buttner MJ, Chater KF, Hopwood DA (2000) Practical streptomyces genetics. The John Innes Foundation, Norwich
Kovach ME et al (1995) Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene 166:175–176
Kwon HJ, Tirumalai R, Landy A, Ellenberger T (1997) Flexibility in DNA recombination: structure of the lambda integrase catalytic core. Science 276:126–131
Larson JL, Hershberger CL (1986) The minimal replicon of a streptomycete plasmid produces an ultrahigh level of plasmid DNA. Plasmid 15(3):199–209
Lydiate DJ, Malpartida F, Hopwood DA (1985) The streptomyces plasmid SCP2*: its functional analysis and development into useful cloning vectors. Gene 35:223–235
Malanowska K, Salyers AA, Gardner JF (2006) Characterization of a conjugative transposon integrase, IntDOT. Mol Microbiol 60:1228–1240
Maxam AM, Gilbert W (1977) A new method for sequencing DNA. Proc Natl Acad Sci USA 74:560–564
Maxam AM, Gilbert W (1980) Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol 65:499–560
Needleman SB, Wunsch CD (1970) A general method applicable to the search for similarities in the amino acid sequence of two proteins. J Mol Biol 48:443–453
Nordstroem K, Austin SJ (1989) Mechanisms that contribute to the stable segregation of plasmids. Annu Rev Genet 23:37–69
Nunes-Dueby SE, Kwon HJ, Tirumalai RS, Ellenberger T, Landy A (1998) Similarities and differences among 105 members of the Int family of site-specific recombinases. Nucleic Acids Res 26:391–406
Pargellis CA, Nunes-Dueby SE, de Vargas LM, Landy A (1988) Suicide recombination substrates yield covalent lambda integrase-DNA complexes and lead to identification of the active site tyrosine. J Biol Chem 263:7678–7685
Rajeev L, Salyers AA, Gardner JF (2006) Characterization of the integrase of NBU1, a Bacteroides mobilizable transposon. Mol Microbiol 61:978–990
Ringrose L, Lounnas V, Ehrlich L, Buchholz F, Wade R, Stewart AF (1998) Comparative kinetic analysis of FLP and cre recombinases: mathematical models for DNA binding and recombination. J Mol Biol 284:363–384
Sambrook J, Russell DWDW (2001) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schrempf H, Bujard H, Hopwood DA, Goebel W (1975) Isolation of covalently closed circular deoxyribonucleic acid from Streptomyces coelicolor A3(2). J Bacteriol 121:416–421
Subramanya HS, Arciszewska LK, Baker RA, Bird LE, Sherratt DJ, Wigley DB (1997) Crystal structure of the site-specific recombinase, XerD. EMBO J 16:5178–5187
Van Mellaert L, Mei L, Lammertyn E, Schacht S, Jozef A (1998) Site-specific integration of bacteriophage VWB genome into streptomyces venezuelae and construction of a VWB-based integrative vector. Microbiology 144(Pt 12):3351–3358
Volff JN, Eichenseer C, Viell P, Piendl W, Altenbuchner J (1996) Nucleotide sequence and role in DNA amplification of the direct repeats composing the amplifiable element AUD1 of Streptomyces lividans 66. Mol Microbiol 21:1037–1047
Wilkins MR et al (1999) Protein identification and analysis tools in the ExPASy server. Methods Mol Biol 112:531–552
Yanisch-Perron C, Vieira J, Messing J (1985) Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33:103–119
Acknowledgments
We would like to thank Prof. Dr. Ralf Mattes for his great and generous support during the past years of research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jean-Luc Pernodet.
The present work has been accomplished within a PhD thesis at Stuttgart University, Institute of Industrial Genetics (IIG).
Rights and permissions
About this article
Cite this article
Warth, L., Haug, I. & Altenbuchner, J. Characterization of the tyrosine recombinase MrpA encoded by the Streptomyces coelicolor A3(2) plasmid SCP2*. Arch Microbiol 193, 187–200 (2011). https://doi.org/10.1007/s00203-010-0662-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-010-0662-9