Abstract
Background
The coronavirus disease 2019 outbreak (COVID-19) caused by the SARS-CoV coronavirus, has been declared as a pandemic by the World Health Organization on 11 March 2020, as a result of which about 315,000,000 people all over the world have been infected and more than 5,000,000 died.
Objective
Many scientific articles have been published concerning histopathological changes in different organs, but data concerning the lung changes of pregnant and postpartum period women who died of COVID-19 infections are still scarce. The aim of our study was to review and summarize autopsy findings and histopathological changes in lungs of pregnant and postpartum period women who died of COVID-19 infections in Armenia during 2020–2021.
Material and methods
Lung tissue specimens of 14 pregnant and postpartum period women who died of COVID-19 infections and its complications were examined. Hematoxylin-eosin and van Gieson staining methods as well as immunohistochemical examinations were used.
Results
The average age of the dead women was 33.9 years. From 14 cases in 9 there were comorbidities. All cases of death were in the 2nd and 3rd trimester of pregnancy or early and late postpartum period. Forensic medical diagnosis included COVID-19 infection with bilateral polysegmented pneumonia and acute respiratory failure. Histopathological examination revealed diffuse alveolar damage of lungs (DAD) in predominantly proliferation/organizing stage.
Conclusion
Histopathological examination of lungs showed proliferative stage of DAD with signs of fibrosing pneumonia. Early diagnosis and hospitalization of pregnant women may prevent late complications of COVID-19 infection and fibrosing pneumonia development, as well as future risks of fatal outcomes in pregnant and early postpartum period.
Zusammenfassung
Grundlagen
Der Ausbruch der Coronavirus-Krankheit 2019 (COVID-19), verursacht durch das Coronavirus SARS-CoV‑2, wurde am 11. März 2020 von der Weltgesundheitsorganisation zu einer Pandemie erklärt. Als Folge infizierten sich etwa 315.000.000 Menschen auf der ganzen Welt, von denen mehr als 5.000.000 starben.
Ziel
Es wurden viele wissenschaftliche Artikel über histopathologische Veränderungen in verschiedenen Organen veröffentlicht, aber Daten über die Lungenveränderungen von schwangeren und postpartalen Frauen, die an einer COVID-19-Infektion verstarben, sind noch rar. Das Ziel unserer Studie war es, die Autopsiebefunde und histopathologischen Veränderungen in der Lunge von Frauen in der Schwangerschaft und nach der Geburt, die im Zeitraum 2020–2021 in Armenien an einer COVID-19-Infektion starben, zu überprüfen und zusammenzufassen.
Materialien und Methoden
Es wurden Lungengewebeproben von 14 schwangeren und postpartalen Frauen entnommen, die an einer COVID-19-Infektion starben, und deren Komplikationen untersucht. Es wurden Hämatoxylin-Eosin und die Färbungsmethode nach van Gieson sowie eine immunhistochemische Untersuchung verwendet.
Ergebnisse
Das Durchschnittsalter der toten Frauen betrug 33,9 Jahre. In 9 von 14 Fällen lagen Komorbiditäten vor. Alle Todesfälle traten im 2. und 3. Trimenon der Schwangerschaft oder im frühen und späten Wochenbett auf. Die gerichtsmedizinische Diagnose umfasste eine COVID-19-Infektion mit bilateralen polysegmentierten Pneumonien und akutem Lungenversagen. Die histopathologische Untersuchung ergab eine diffuse alveoläre Schädigung der Lunge (DAD) im überwiegenden Proliferations‑/Organisierungsstadium.
Schlussfolgerung
Die histopathologische Untersuchung der Lungen zeigte ein proliferatives Stadium der DAD mit Anzeichen einer fibrosierenden Pneumonie. Eine frühzeitige Diagnose und Krankenhauseinweisung schwangerer Frauen könnte Spätkomplikationen einer COVID-19-Infektion und die Entwicklung einer fibrosierenden Lungenentzündung sowie zukünftige Risiken tödlicher Ausgänge bei Schwangeren und in der frühen Zeit nach der Geburt verhindern.
Similar content being viewed by others
Introduction
Coronaviruses are a large family of viruses which affect both animals and humans. In humans, several coronaviruses cause respiratory infections, such as Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS) caused by SARS-CoV. The coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the outbreak of which was declared a pandemic on 11 March 2020 by the World Health Organization (WHO), has caused not only increase in hospitalizations for predominantly pneumonia all over the world, but also resulted in fatal outcomes of many patients with estimated average mortality rates up to 3.4% globally (mortality rate estimated by the WHO as of 3 March 2020, Director-General Tedros Adhanom Ghebreyesus said during a press briefing at the agency’s headquarters in Geneva). The mortality rate differs depending on the age, presence of concurrent diseases (comorbidities) and pathologies starting from 0.3% in people under 65 years old and 4.5% in people over 65 years old with the presence of concurrent chronic diseases such as hypertension, diabetes mellitus etc.
According to the Centers of Disease Control and Prevention (updated 11 November 2021) the risk of COVID-19 infection mortality in different age groups was different. Compared to patients 18–29 years old, the rate of death is 4 times higher in those 30–39 years old and 370 times higher in those who are 85 years and older. Not only old individuals are the target groups of fatal cases, but also immunocompromised people and pregnant women. In pregnant women, independent from the causative agent, pneumonias have more severe course associated with decreased lung surface, increased demand for oxygen, increased uterine volume and elevation of diaphragm.
According to the literature, in early infections SARS-CoV‑2 target cells are nasal, bronchial epithelial cells, pneumocytes. Inhaled viral particles are deposited on the nasal mucosa epithelial target cells, where the virus replicates within the cytoplasm. The cell surface receptor ACE2 is recognized by SASRS-CoV‑2 spike (S) protein, and TMPRSS2 is a serine protease that cleaves the S protein for viral entry. SARS-CoV‑2 infects the ACE2 expressing ciliated cells. SARS-CoV‑2 replicates, is released apically, and infects neighboring cells [3] after the virus enters the airways. Infection in the bronchi produces epithelial injury, cellular infiltrates and in some cases marked denudation of the epithelial surface [2, 4]. SARS-CoV‑2 then enters the alveoli, infects ACE2 expressing alveolar type II cells and replicates. Infected AT2 cells (lung epithelial cells that secret surfactant) release virus that infects bystander type II cells. Infected and bystander cells secret inflammatory cytokines and chemokines to recruit and activate immune cells. Attracted inflammatory cells affect endothelial cells. According to the literature, in later stages of infection, the virus itself infects the pulmonary capillary endothelial cells and pericytes. This process then triggers the inflammatory reaction with influx of neutrophils and other inflammatory cells, which evolves to diffuse alveolar damage. The end effect of diffuse alveolar damage is loss of functional surfactant, alveolar flooding, damage to type I cells and endothelial cells [3], with late complications including organization of inflammatory exudates and pneumofibrosis development. In severe infection, there is a fulminant activation of the coagulation cascade, with microthrombus formation, pulmonary embolism and variable size lung infarcts in critically ill patients [1, 5, 9].
Microscopic (histologic) description of DAD includes two main stages—exudative/acute phase and proliferative or organizing phase. Some authors also admit the third fibrosing phase of DAD. The phases of DAD depend on period of ARDS [6, 8, 10]. In the exudative/acute phase there are alveolar changes usually with formation of hyaline membranes in alveolar walls, deposition of fibrin, alveolar and interstitial edema, desquamation of alveolocytes and necrosis of type I pneumocytes, vascular changes such as necrosis of endotheliocytes, microthrombi and hemorrhages. In proliferation or organizing stage there are organizing changes of hyaline membrane remnants, interstitial and intra-alveolar proliferation of myofibroblasts, proliferation of type II pneumocytes, endothelial injury and thromboembolism in arteries. In the stage of pneumofibrosis, architectural remodeling of lung tissue sometimes with honeycomb lung appearance, squamous cell hyperplasia/metaplasia of alveolar and bronchiolar epithelium and remodeling of arteries is seen in many cases and superimposed bacterial pneumonia may develop [10].
Material and methods
Autopsy cases of pregnant and postpartum period women who died of COVID-19 in Armenia during the years 2020 and 2021 were examined. Women with antemortem and postmortem positive results to SARS-CoV2 were included in the study.
As an autopsy material lung tissue was examined. Tissues were fixed in 10% neutral buffered formalin, after paraffin-embedded tissue samples were submitted for routine standard staining such as hematoxylin and eosin. To reveal connective tissue overgrowth and pneumofibrosis, the tissue samples were stained by the van Gieson method (fibrous connective tissue was stained in bright red, other tissues in brownish-yellow).
Immunohistochemical staining was performed to detect viral particles (SARS-CoV‑2 S1spike protein) in lung tissue specimens.
Automated classical immunohistochemical (IHC) stains were performed using the Benchmark ULTRA (Roche, Ventana Medical Systems, Tucson, AZ, USA) on formalin-fixed and paraffin-embedded (FFPE) tissue sections. After dewaxing, tissue slides were heat pretreated using a cell conditioning buffer 1 (CC1) (pH8) buffer for 64 min at 98 °C. The slides were blocked for endogenous peroxidase activity and incubated with primary anti-2019-nCOV Spike rabbit polyclonal and anti-SARS-CoV‑2 nucleocapsid mouse monoclonal (Ready to Use, clone BSB-134, BioSB, Diagomics, Blagnac, France) antibodies. Targets were then visualized using the OptiView DAB (Tucson, AZ, USA) detection kit. The tissue slides were counterstained using hematoxylin followed by postcoloration using bluing reagent.
In the examination the age of women, week of pregnancy, postpartum period, comorbidities, forensic medical and histopathological diagnosis were considered (Tables 1 and 2).
As seen from the tables, the age of women was ranging from 23 to 44 years old, with average age of 33.9 years.
The period of pregnancy was the second and the third trimester (ranging from 19 weeks to 39–40 weeks). In 8 cases cesarean section was performed considering severe condition of pregnant women.
In 9 women comorbidities such as obesity of different degrees, arterial hypertension, thyrotoxicosis, diabetes, hypothyrosis were present.
Results and discussion
Autopsy material of lung tissue specimens were examined grossly and microscopically.
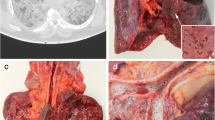
On gross examination the usual findings in the lungs included severe congestion with numerous small petechial hemorrhages under the pleura and in the parenchyma, which were seen also on the cut surfaces. The pleura was dull and on the pleural surface there was fibrinous purulent film of various thicknesses, sometimes with numerous adhesions with the surrounding tissues. The color of lung tissue was dark red or grayish-red. The consistency was firm. In some cases, there were pleural effusions. On the cut surface the lung tissue had patchy, multicolored appearance caused not only by numerous small focal, petechial hemorrhages, but also purulent exudate, abscesses, sometimes areas of atelectasis and emphysema. There was release of hemorrhagic, purulent hemorrhagic, serous hemorrhagic, sometimes foamy exudate from the cut surfaces of lung tissue (Fig. 1). Tracheal and bronchial mucosa showed marked hyperemia with small focal hemorrhages. In the lung vessels, besides postmortem clots, the usual findings included thrombi.
Microscopically the typical changes in the lungs showed signs of diffuse alveolar damage (DAD).
In the exudation stage (the first 7–10 days of the disease) of DAD the most prominent changes included:
-
Hyaline membranes of different thickness, but in many cases with much expressed thickening of alveolar walls.
-
Intra-alveolar edema with accumulation of foamy eosinophilic abundant exudate in the alveoli.
-
Small focal hemorrhages in the peribronchial and perivascular connective tissue and alveoli.
-
Superimposed bacterial pneumonia with purulent destruction of the lung parenchyma and small abscesses formation.
-
Sometimes presence of desquamated alveolocytes, few numbers of macrophages in the serous fluid of alveoli.
In the proliferation stage (starting after 7–15 days from the onset of the disease) the most prominent changes included.
-
Organization of hyaline membranes with extreme fibrous thickening of alveolar walls.
-
Resorption and organization of inflammatory intra-alveolar exudate with widespread ingrowth of granulation tissue into alveolar lumina.
-
Carnification of lung tissue with widespread fibrosis and with totally airless lung tissue formation.
-
Proliferation of alveolocytes with appearance of giant multinucleated alveolocytes in the alveoli.
-
Myxoid changes in the interstitial interalveolar tissue of lungs.
-
Squamous cell metaplasia of bronchiolar and bronchial epithelium.
In acute phase of DAD, the most common changes were associated with interalveolar and intra-alveolar edema and accumulation of foamy, spongy eosinophilic exudate in the alveoli. In the exudate there was admixture of polymorphonuclear leukocytes, fibrin strands, few number of lymphocytes, plasma cells and macrophages (Fig. 2), desquamated alveolar epithelial cells, erythrocytes. In superimposed bacterial infections, there were different types of pneumonias depending on the character of inflammatory exudate—serous, serous purulent or purulent. In severe cases purulent pneumonias were accompanied by purulent destruction of lung tissue and formation of small abscesses (abscessing pneumonias). Exudation phase of DAD was accompanied by deposition of amorphous eosinophilic masses in the alveolar walls with formation of hyaline membranes. In some cases, formation of hyaline membranes had focal character with involvement of local areas of one or both lungs, but in many cases it was of diffuse character with affection of the entire lung parenchyma of both lungs. Hyaline membranes characterized by dense eosinophilic amorphous material arranged along the alveolar walls and septa and composed of cellular debris, plasma proteins such as albumin, immunoglobulins, fibrinogen and surfactant [7], usually had different thicknesses. In examined cases expressed thickening of alveolar walls and interalveolar septa with severe narrowing of alveolar spaces were seen (Fig. 2).
In the exudative stage the common findings were also small focal hemorrhages in the interalveolar tissue and blood accumulation in the alveoli. Sometimes hemorrhages were large with involvement of wider areas of lung tissue, which resembled lung infarctions (Fig. 3). Areas of usually small focal hemorrhages were also seen in the peribronchial and perivascular tissues, under the pleura.
Local or diffuse alveolar wall thickening with hyaline membranes was not seen only in the exudation stage of DAD, but also in the proliferation stage, where along with the signs of resorption and organization of inflammatory exudate, there was also organization of hyaline membranes (Fig. 4).
In predominant cases histopathological changes in the lungs of dead women were displaying proliferative/organizing stage of DAD, rarely with admixture of exudative component.
In many cases in early proliferation stage the typical findings included organizing hyaline membranes, alveolar infiltration by macrophages, organizing fibrin deposits, the presence of giant multinucleated alveolocytes, proliferating fibroblasts (Figs. 4 and 5). The resorption and organizing processes in the lungs were accompanied by the development of fibrosing (organizing) pneumonia with severe thickening of interalveolar septa. Thickening of interalveolar septa sometimes was associated with myxoid changes or ingrowth of loose connective tissue. In many cases there was overgrowth of fibrous connective tissue with severe pneumofibrosis development. Organization of inflammatory exudate in the alveoli was manifested by ingrowth of granulation tissue into alveoli (Fig. 5) with subsequent carnification of lung tissue, which in many cases, along with simultaneous thickening and fibrosis of interalveolar septa, ended by irreversible remodeling of lung tissue architecture, formation of totally airless lung parenchyma. In the late proliferation stage there were cases of cyst-like transformation of terminal bronchioles, which along with pneumofibrosis and fibrous thickening of interalveolar septa ended to honey-comb transformation of lung parenchyma.
In the late proliferation stage of DAD in many cases there was squamous-cell metaplasia of bronchiolar and bronchiolar epithelium, which has focal or sometimes diffuse character (Fig. 6).
From the moment of COVID-19 infection diagnosis (positive COVID tests) until death the minimum time interval was 2 weeks with a maximum of about 4 weeks.
To reveal connective tissue growth in lungs during fibrosing pneumonia van Gieson staining method was used (Fig. 7).
Immunohistochemical staining of lung tissues revealed SARS-CoV‑2 viral RNA/S1 spike protein/in lung tissue of the deceased women (Fig. 8). Positive staining showed occasional macrophages, with engulfed viral proteins in the cytoplasm showing dark brown color. Few amounts of positive stained cells may be explained by the elimination of the viral particles from the cells in the proliferation stage of DAD.
Conclusion
Based on the examination results we conclude that:
-
During 2020–2021 from total 24 cases of maternal death in Armenia 14 cases (58%) were caused by COVID-19 infection or its early and late complications (COVID infection as comorbid disease was not considered).
-
From 14 deceased women 9 of them had comorbidities such as obesity of different degree, diabetes mellitus, arterial hypertension, phlebothrombosis of deep veins of lower extremities, endocrine pathologies such as hypothyrosis, thyrotoxicosis, gestational diabetes etc.
-
The average age of the deceased women was 33.9 years (ranging from 23 to 44 years).
-
Fatal outcomes were observed in the second and third trimester of pregnancy (starting from 19 weeks of pregnancy to 38–49 weeks) or in postpartum period.
-
Histological examination of lung tissue in 12 cases revealed late (proliferative), only in 2 cases early (exudation) phase of DAD, predominance of fibrosing pneumonias, structural remodeling of lung tissue and respiratory failure, which was the main immediate cause of death.
-
The risk factors for fatal COVID-19 infection in pregnant women include age over 30 years, 3rd trimester of pregnancy, comorbidities, late diagnosis and treatment of the disease.
-
Prevention of fatal complications of COVID-19 infection is possible on condition of early diagnosis and adequate immediate treatment of the disease, which could result in decrease in maternal mortality caused by coronavirus infection.
References
Montero-Fernandez AM, Pardo-Garcia R (2021) Histopathology features of the lung in COVID-19 patients. Diagn Histopathol (Oxf) 27(3):123–127. https://doi.org/10.1016/j.mpdhp.2020.11.009
Borczuk AC, Salvatore SP, Seshan SV et al (2020) COVID-19 pulmonary pathology: a multi-institutional autopsy cohort from Italy and New York City. Mod Pathol 33:2156–2168. https://doi.org/10.1038/s41379-020-00661-1
Bridges JP, Vladar EK, Huang H, Mason RJ (2022) Respiratory epithelial cell responses to SARS-CoV‑2 in COVID-19. Thorax 77(2):203–209. https://doi.org/10.1136/thoraxjnl-2021-217561
Martines RB, Ritter JM, Matkovic E et al (2020) Pathology and pathogenesis of SARS-CoV‑2 associated with fatal coronavirus disease, United States. Emerg Infect Dis 26:2005–2015. https://doi.org/10.3201/eid2609.202095
Magro C, Mulvey JJ, Berlin D (2020) Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res 220:1–13. https://doi.org/10.1016/j.trsl.2020.04.007
Beasley MB (2010) The pathologist’s approach to acute lung injury. Arch Pathol Lab Med 134(5):719–727. https://doi.org/10.5858/134.5.719
Cardinal-Fernandez P, Lerente JA, Ballen-Barragan A, Naute-Bello G (2017) Acute respiratory distress syndrome and diffuse alveolar damage. New insight on complex relationship. Ann Am Thorac Soc 14(6):844–850. https://doi.org/10.1513/AnnalsATS.201609-728 PS
Tomashefski JF Jr. (2000) Pulmonary pathology of acute respiratory distress syndrome. Clin Chest Med 21(3):435–466. https://doi.org/10.1016/s0272-5231(05)70158-1
Wiersinga W, Rhodes A, Cheng A, Peacock S, Prescott H (2020) Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. JAMA 324(8):782–793. https://doi.org/10.1001/jama.2020.12839
Yoshikawa A, Bychkov A ARDS/DAD. Pathology outlines.com website. https://www.pathologyoutlines.com/topic/lungnontumordiffusealveolardamage.html. Accessed 12 Jan 2022
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M.S. Bisharyan, K.A. Arsenyan, P.S. Khachatryan, M.Z. Muradyan and A.A. Tonoyan declare that they have no competing interests.
For this article no studies with human participants or animals were performed by any of the authors. All studies mentioned were in accordance with the ethical standards indicated in each case. The investigations were carried out according to the specifications of the Central Ethics Committee of the Federal Medical Council.
Additional information

Scan QR code & read article online
Rights and permissions
About this article
Cite this article
Bisharyan, M.S., Arsenyan, K.A., Khachatryan, P.S. et al. Histopathological autopsy findings in lungs of pregnant and postpartum women who died of COVID-19 infections. Rechtsmedizin 33, 403–409 (2023). https://doi.org/10.1007/s00194-023-00618-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00194-023-00618-z