Abstract
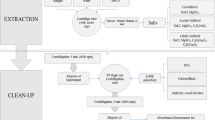
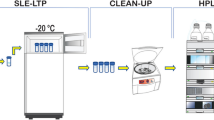
An effective analytical method for the residue analysis of a novel acaricide hexythiozox and its dissipation in beans pods were studied. Hexythiozox residues were extracted from beans pods samples and the extract was cleaned up according to QuEChERS (Quick, Easy, Cheap, Effective, Rugged and Safe) method and determined by high-performance liquid chromatography with photodiode array detector (HPLC–DAD). At fortification levels of 0.1, 0.5, and 1.0 mg kg−1 in Beans Pods, it was shown that recoveries ranged from 82.4 % to 89.6 % with relative standard deviation (RSD) of 6 %–9 %. The limit of detection (LOD) and limit of quantification (LOQ) were found to be 0.02 and 0.06 mg kg−1, respectively. The dissipation half-life time of hexythiozox residues in beans pods was 12.04 days. According to maximum residue limit (MRL) 0.5 mg kg−1, the preharvest interval (PHI) of hexythiozox on beans pods was 10 days after the treatment. Based on the results of this study and the relevant residue regulation, hexythiozox residue levels will be acceptable when applied to beans pods in Egypt.


Similar content being viewed by others
References
Abd-Alrahman SH, Almaz MM, Ahmed NS (2011a) Dissipation of fungicides, insecticides, and acaricide in tomato using HPLC-DAD and QuEChERS methodology. Food Anal Methods 5:564–570
Abd-Alrahman SH, Almaz MM, Osama IA (2011b) Determination of degradation rate of acaricide Fenpyroximate in apple, citrus, and grape by HPLC-DAD. Food Anal Methods 5:306–311
Ambrus A, Lantos J (2002) Evaluation of the studies on decline of pesticide residues. J Agric Food Chem 50:4846–4851
Blasco C, Pico Y, Manes J, Font G (2002) Determination of fungicide residues in fruits and vegetables by liquid chromatography-atmospheric pressure chemical ionization mass spectrometry. J Chromatogr A 947:227–235
Blasco C, Font G, Pico Y (2004) Multiple-stage mass spectrometric analysis of six pesticides in oranges by liquid chromatography-atmospheric pressure chemical ionization-ion trap mass spectrometry. J Chromatogr A 1043:231–238
Blasco C, Font G, Pico Y (2006) Evaluation of 10 pesticide residues in oranges and tangerines from Valencia (Spain). Food Control 17:841–846
Caboni P, Sarais G, Angioni A, Vargiu S, Pagnozzi D, Cabras P, Casida J (2008) Liquid chromatography—tandem mass spectrometric ion-switching determination of chlorantraniliprole and flubendiamide in fruits and vegetables. J Agric Food Chem 56:7696–7699
Cabras P, Meloni M, Gennari M, Cabitza F, Cubeddu M (1989) Pesticide residues in lettuce. 2. Influence of formulations. J Agric Food Chem 37:1405–1407
Cabras P, Spanedda L, Cabitza F, Cubeddu M, Martini MG, Brandolini V (1990) Pirimicarb and its metabolites residues in lettuce. Influence of cultural environment. J Agric Food Chem 38:879–882
Castillo-Sanchez J, Aguilera-Del Real A, Rodriguez-Sanchez M, Valverde-Garcia A (2000) Residue levels, decline curves, and plantation distribution of procymidone in green beans grown in greenhouse. J Agric Food Chem 48:2991–2994
Coscolla C, Yusa V, Marti P, Pastor A (2008) Analysis of currently used pesticides in fine airborne particulate matter (PM 2.5) by pressurized liquid extraction and liquid chromatography-tandem mass spectrometry. J Chromatogr A 1200:100–107
Coscolla C, Yusa V, Beser MI, Pastor A (2009) Multi-residue analysis of 30 currently used pesticides in fine airborne particulate matter (PM 2.5) by microwave-assisted extraction and liquid chromatography-tandem mass spectrometry. J Chromatogr A 1216:8817–8827
FAO/WHO J (1999) Recommended method of sampling for the determination of pesticide residues for compliance with MRLs. Food standards programme, CAC/GL: 1–18
FAO/WHO (2006) Joint FAO/WHO Food standards program. Codex committee on pesticide residues CX/PR 06/38/5:177
Fenoll J, Ruiz E, Hellín P, Lacasa A, Flores P (2009) Dissipation rates of insecticides and fungicides in peppers grown in greenhouse and under cold storage conditions. Food Chem 113:727–732
Gennari M, Zannini E, Cignetti A, Bicchi C, DAmato A, Taccheo M, Spessotto C, De Paoli M, Flori P (1985) Vinclozolin decay on different grape vines in four different Italian areas. J Agric Food Chem 33:1232–1237
Hetherton CL, Sykes MD, Fussell RJ, Goodall DM (2004) A multi-residue screening method for the determination of 73 pesticides and metabolites in fruit and vegetables using high-performance liquid chromatography/tandem mass spectrometry. Rapid Commun Mass Spectrom 18:2443–2450
Holland PT, McGhie TK (1983) Multiresidue method for determination of pesticides in kiwi fruit, apples, and berry fruits. J Assoc Off Anal Chem 66:1003–1008
Hoskin M (1961) Mathematical treatments of the rate of loss of pesticide residues. FAO P1 Prot Bull 9:163–168 http://www.epa.gov/EPA-PEST
Khay S, Choi J, Abd El-Aty M (2008) Dissipation behavior of lufenuron, benzoyphenylurea insecticide, in/on Chinese cabbage applied by foliar spraying under greenhouse condition. Bull Environ Contam Toxicol 81:369–372
Liu Y, Zhao E, Zhu W, Gao H, Zhou Z (2009) Determination of four heterocyclic insecticides by ionic liquid dispersive liquid–liquid microextraction in water samples. J Chromatogr A 1216:885–891
Omirou M, Vryzas Z, Papadopoulou-Mourkidou E, Economou A (2009) Dissipation rates of iprodione and thiacloprid during tomato production in greenhouse. Food Chem 116:499–504
Sannino A, Bolzoni L, Bandini M (2004) Application of liquid chromatography with electrospray tandem mass spectrometry to the determination of a new generation of pesticides in processed fruits and vegetables. J Chromatogr A 1036:161–169
Valenzuela AI, Pico Y, Font G (2001) Determination of five pesticide residues in oranges by matrix solid-phase dispersion and liquid chromatography to estimate daily intake of consumers. J AOAC Int 84:901–909
Yoshii K, Kaihara A, Tsumura Y, Ishimitsu S, Tonogai Y (2000) Liquid chromatographic determination of emamectin, milbemectin, ivermectin and abamectin in crops and confirmation by liquid chromatography-mass spectrometry. J Chromatogr A 896:75–85
Zhou P, Yt Lu, Liu BF, JJ Gan (2004) Dynamics of fipronil residue in vegetable-field ecosystem. Chemosphere 57:1691–1696
Acknowledgments
The project was supported by the Research Center, College of Science, King Saud University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Abd-Alrahman, S.H. Dissipation of Hexythiozox on Beans Pods by HPLC–DAD. Bull Environ Contam Toxicol 90, 504–507 (2013). https://doi.org/10.1007/s00128-012-0942-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-012-0942-y