Abstract
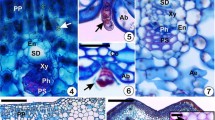
In a general way, galling insects can induce only one gall morphotype on the host plant species. However, some taxa of galling insects such as Eriococcidae can induce sexually dimorphic galls on the same host. In the present study, Lopesia mataybae (Diptera: Cecidomyiidae) induced two gall morphotypes on the leaflets of Matayba guianensis (Sapindaceae), a rare evidence of sexual dimorphism in galls induced by Cecidomyiidae. We investigated the adult emergence of galling insects and the morphological, histological, and histochemical attributes of the gall morphotypes. Both gall morphotypes showed the galling insect in the larval or pupal stage, and the insect adult emergence from different morphotypes was sexually distinct. Galls occupied by females (Globoid) were significantly larger (average height = 4.67 mm. S = 0.43/average width = 4.59 mm. S = 0.70) than galls occupied by males (cylindrical) (average height = 2.95 mm. S = 0.46/average width = 2.34 mm. S = 0.45). Both gall types were composed of elongated cells in the inner cortex and rounded cells in the outer cortex of the vessel-like structure. The globoid galls showed 42 cell layers (14 of them cylindrical, S = 3.86; 28 rounded, S = 5.89) and cylindrical galls showed 29 layers. There were no differences between morphotypes in the histochemical compounds detected. We have shown here significant morphological and histological differences between male and female galls possibly due to the different nutritional requirements of Lopesia mataybae sexes. Therefore, the expression of sexual dimorphism in gall morphotypes may be associated with developmental pathway differentiation between the sexes of L. mataybae.





Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Amorim DO, Ferreira BG, Fleury G (2017) Plant potentialities determine anatomical and histochemical diversity in Mikania glomerata Spreng. galls. Rev Bras Bot 40:517–527. https://doi.org/10.1007/s40415-016-0357-9
Bartlett L, Connor EF (2014) Exogenous phytohormones and the induction of plant galls by insects. Arthropod Plant Interact 8:339–348. https://doi.org/10.1007/s11829-014-9309-0
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Bronner R (1992) The role of nutritive cells in the nutrition of Cynipids and Cecidomyiids. In: Shorthouse JD, Rohfritsch O (eds) Biology of Insect Induced-galls. Oxford University Press, pp 118–137
Bukatsch F (1972) Bemerkungen zur Doppelfarbung: Astrablau-Safranin. Mikrokosmos 61:255
Castro AC, Oliveira DC, Moreira ASFP et al (2012) Source-sink relationship and photosynthesis in the horn-shaped gall and its host plant Copaifera langsdorffii Desf. (Fabaceae) South African. J Bot 83:121–126. https://doi.org/10.1016/j.sajb.2012.08.007
Condrashoff SF (1962) Bionomics of three closely related species of Contarinia Rond. (Diptera: Cecidomyiidae) from Douglas-fir needles. Can Entomol 94:376–394. https://doi.org/10.4039/Ent94376-4
Costa FV, Fagundes M, De Siqueira NF (2010) Arquitetura da planta e diversidade de galhas associadas à Copaifera langsdorffii (Fabaceae). Ecol Austral 20:9–17
Costa EC, Oliveira DC, Ferreira DKL, Isaias RMS (2021) Structural and nutritional peculiarities related to lifespan differences on four Lopesia induced bivalve-shaped galls on the single super-host Mimosa gemmulata. Front Plant Sci 12:1–13. https://doi.org/10.3389/fpls.2021.660557
de Silva AF, M, Kuster VC, Rezende UC, de Oliveira DC, (2019) The early developmental stages of gall-inducing insects define final gall structural and histochemical profiles: the case of Bystracoccus mataybae galls on Matayba guianensis. Botany 97:427–438. https://doi.org/10.1139/cjb-2019-0017
Dorchin N, Clarkin CE, Scott ER et al (2007) Taxonomy, life history, and population sex ratios of North American Dasineura (Diptera: Cecidomyiidae) on goldenrods (Asteraceae). Ann Entomol Soc Am 100:539–548. https://doi.org/10.1603/0013-8746(2007)100[539:TLHAPS]2.0.CO;2
Dorchin N, Hoffmann JH, Stirk WA et al (2009) Sexually dimorphic gall structures correspond to differential phytohormone contents in male and female wasp larvae. Physiol Entomol 34:359–369. https://doi.org/10.1111/j.1365-3032.2009.00702.x
Ferreira BG, Avritzer SC, Isaias RMS (2017a) Totipotent nutritive cells and indeterminate growth in galls of Ditylenchus gallaeformans (Nematoda) on reproductive apices of Miconia. Flora Morphol Distrib Funct Ecol Plants 227:36–45. https://doi.org/10.1016/j.flora.2016.12.008
Ferreira BG, Álvarez R, Avritzer SC, Isaias RMS (2017b) Revisiting the histological patterns of storage tissues: beyond the limits of gall-inducing taxa. Botany 95:173–184. https://doi.org/10.1139/cjb-2016-0189
Ferreira BG, Álvarez R, Bragança GP et al (2019) Feeding and Other Gall Facets: Patterns and Determinants in Gall Structure. Bot Rev 85:78–106. https://doi.org/10.1007/s12229-019-09207-w
Fujii T, Matsuo K, Abe Y, Yukawa J, Tokuda M (2014) An endoparasitoid avoids hyperparasitism by manipulating immobile host herbivore to modify host plant morphology. PLoS ONE 9(7):1–12. https://doi.org/10.1371/journal.pone.0102508
Gagné RJ (1994) The gall midges of the Neotropical Region. Cornell University Press
Ganaha, T., Nohara, M., Sato, S., Uechi, N., Yamagishi, K., Yamauchi, S., & Yukawa, J. (2007). Polymorphism of axillary bud galls induced by Rhopalomyia longitubifex (Diptera: Cecidomyiidae) on Artemisia princeps and A. montana (Asteraceae) in Japan and Korea, with designation of new synonyms. Entomological Science, 10(2), 157–169. https://doi.org/10.1111/j.1479-8298.2007.00210.x
Garcia CA, Urso-Guimarães MV (2018) Three new species of Lopesia Rübsaamen (Diptera: Cecidomyiidae) from Brazil. Florida Entomol 101:203–211. https://doi.org/10.1653/024.101.0208
Gonçalves SJMR, Isaias RMS, Vale FHA, Fernandes GW (2005) Sexual dimorphism of Pseudotectococcus rolliniae hodgson & gonçalves 2004 (Hemiptera: Coccoidea Eriococcidae) influences gall morphology on Rollinia laurifolia schltdl. (Annonaceae). Trop Zool 18:161–169. https://doi.org/10.1080/03946975.2005.10531219
Guedes LM, Aguilera N, Ferreira BG, Becerra J, Sáez K, Pérez C, Hernández V, Isaias RMS (2018) Factors influencing the morphogenesis of galls induced by Calophya mammifex (Calophyidae) on Schinus polygama (Anacardiaceae) leaves. Botany 96(9):589–599. https://doi.org/10.1139/cjb-2018-0078
Gullan PJ, MIller DR and Cook LG (2005) Gall-inducing scale insects (Hemiptera: Sternorrhyncha: Coccoidea). In: Raman A. et al. (ed) Biology, ecology, and evolution of gall-inducing arthropod, New Delhi: Oxford & IBH Publishing Co. Pvt Ltd., 774 pp.
Hartley SE (1998) The chemical composition of plant galls: are levels of nutrients and secondary compounds controlled by the gall former? Oecologia 113:492–501. https://doi.org/10.1007/s004420050401
Hawkins BA, Cornell HV, Hochberg ME (1997) Predators, parasitoids, and pathogens as mortality agents in phytophagous insect populations. Ecology 78:2145–2152. https://doi.org/10.1890/0012-9658(1997)078[2145:PPAPAM]2.0.CO;2
Hayward A, Stone GN (2005) Oak gall wasp communities: evolution and ecology. Basic Appl Ecol 6:435–443. https://doi.org/10.1016/j.baae.2005.07.003
Hodgson C, Isaias RMS, Oliveira DC (2013) A new gall-inducing genus and species of eriococcidae (hemiptera: Sternorrhyncha: Coccoidea) on Sapindaceae from Brazil. Zootaxa 3734:317–330. https://doi.org/10.11646/zootaxa.3734.3.2
Isaias RMS, Carneiro RGS, Santos JC, Oliveira DC (2014a) Gall morphotypes in the Neotropics and the need to standardize them. In: Fernandes GW and Santos, JC (ed) Neotropical insect galls, 1st edn. Springer, Dordrecht. pp 51–67 https://doi.org/10.1007/978-94-017-8783-3_4
Isaias RMS, Oliveira DC, Carneiro RGS, and Kraus JE (2014b) Developmental anatomy of galls in the neotropics: arthropods stimuli versus host plant constraints. In: Fernandes GW, Santos JC (eds) Neotropical Insect Galls. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-8783-3_2
Jankiewicz LS, Plich H, Antoszewski R (1970) Preliminary studies on the translocation of 14 C-labelled assimilates and 32 PO3 −towards the gall evoked by Cynips quercus-folii L. on oak leaves. Marcellia (strasburg) 36:163–172
Johansen DA (1940) Plant microtechnique. McGraw-Hill Books, New York, N.Y.
Kurzfeld-Zexer L, Lev-Yadun S, Inbar M (2015) One aphid species induces three gall types on a single plant: comparative histology of one genotype and multiple extended phenotypes. Flora Morphol Distrib Funct Ecol Plants 210:19–30. https://doi.org/10.1016/j.flora.2014.10.007
Larson KC, Whitham TG (1991) Manipulation of food resources by a gall forming aphid: the physiology of sink-source interaction. Oecologia 88:15–21. https://doi.org/10.1007/BF00328398
Lin SF, Tokuda M, Yang MM (2019) Leaf gall polymorphism and molecular phylogeny of a new Bruggmanniella species (Diptera: Cecidomyiidae: Asphondyliini) associated with Litsea acuminata (Lauraceae) in Taiwan, with ecological comparisons and a species description. Entomological Science 23(1):10–22. https://doi.org/10.1111/ens.12391
Magalhães TA, Oliveira DC, Isaias RMS (2015) Population dynamics of the gall inducer Eriogallococcus isaias (Hemiptera: Coccoidea: Eriococcidae) on Pseudobombax grandiflorum (Malvaceae). J Nat Hist 49:789–801. https://doi.org/10.1080/00222933.2014.951083
Malenovský I, Burckhardt D, Queiroz DL et al (2015) Descriptions of two new Pseudophacopteron species (Hemiptera: Psylloidea: Phacopteronidae) inducing galls on Aspidosperma (apocynaceae) in Brazil. Acta Entomol Musei Natl Pragae 55:513–538
Mani MS (1964) Ecology of plant galls. W. Junk, The Hague, p 434p
Martini VC, Raymundo D, Prado-Junior J, Oliveira DC (2021) Bottom-up and top-down forces in plant-gall relationships: testing the hypotheses of resource concentration, associational resistance, and host fitness reduction. Ecol Entomol. https://doi.org/10.1111/een.13043
Mazia D, Brewer PA, Alfert M (1953) The cytochemistry staining and measurement of protein with mercuric bromophenol blue. Biol Bull 104:57–67
Meyer J and Maresquelle HJ (1983) Anatomie des Galles. Gebrüder Borntraeger, Berlin, Deutschland
Miller DG (1998) Life history, ecology and communal gall occupation in the manzanita leaf-gall aphid, Tamalia coweni (Cockerell) (Homoptera: Aphididae). J Nat Hist 32:351–366. https://doi.org/10.1080/00222939800770181
Mishima, M., & Yukawa, J. (2007). Dimorphism of leaf galls induced by Pseudasphondylia neolitseae (Diptera: Cecidomyiidae) on Neolitsea sericea (Lauraceae) and their distributional patterns in Kyushu, Japan. Bulletin of the Kyushu University Museum, 57–64.
O’Brien TP, Feder N, McCully ME (1965) Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma 59:368–373
Oliveira DC, Isaias RMS (2010) Redifferentiation of leaflet tissues during midrib gall development in Copaifera langsdorffii (Fabaceae). S Afr J Bot 76(2):239–248. https://doi.org/10.1016/j.sajb.2009.10.011
Oliveira DC, Christiano JDCS, Soares GLG, Isaias RMDS (2006) Reações de defesas químicas e estruturais de Lonchocarpus muehlbergianus Hassl. (Fabaceae) à ação do galhador Euphalerus ostreoides Crawf. (Hemiptera: Psyllidae). Rev Bras Bot 29:657–667. https://doi.org/10.1590/S0100-84042006000400015
Oliveira DC, Drummond MM, Pinheiro Moreira ASF et al (2008) Potencialidades morfogênicas de Copaifera langsdorffii Desf. (Fabaceae): super-hospedeira de herbívoros galhadores. Rev Biol Neotrop 5:31–39. https://doi.org/10.5216/rbn.v5i1.5625
Oliveira DC, Magalhães TA, Carneiro RGS et al (2010) Do Cecidomyiidae galls of Aspidosperma spruceanum (Apocynaceae) fit the pre-established cytological and histochemical patterns? Protoplasma 242:81–93. https://doi.org/10.1007/s00709-010-0128-6
Oliveira DC, da Carneiro RGS, Magalhães TA, dos Isaias RMS (2011) Cytological and histochemical gradients on two Copaifera langsdorffii Desf. (Fabaceae)-Cecidomyiidae gall systems. Protoplasma 248:829–837. https://doi.org/10.1007/s00709-010-0258-x
Oliveira DC, Magalhães TA, Ferreira BG, Teixeira CT, Formiga AT et al (2014) Variation in the degree of pectin methylesterification during the development of Baccharis dracunculifolia kidney-shaped gall. PLoS ONE 9(4):e94588. https://doi.org/10.1371/journal.pone.0094588
Oliveira DC, Isaias RMS, Fernandes GW et al (2016) Manipulation of host plant cells and tissues by gall-inducing insects and adaptive strategies used by different feeding guilds. J Insect Physiol 84:103–113. https://doi.org/10.1016/j.jinsphys.2015.11.012
Oliveira DC, Moreira ASFP, Isaias RMS, Martini V and Rezende UC (2017) Sink status and photosynthetic rate of the leaflet galls induced by Bystracoccus mataybae (Eriococcidae) on Matayba guianensis (Sapindaceae). Front Plant Sci 8https://doi.org/10.3389/fpls.2017.01249
Pfeffer L, Rezende UC, Barônio GJ, de Oliveira DC (2018) Building two houses on a single host plant: galling insect synchronizes its life cycle with plant phenology. Oecologia Aust 22:438–448. https://doi.org/10.4257/oeco.2018.2204.07
Price PW (2005) Adaptive radiation of gall-inducing insects. Basic Appl Ecol 6:413–421. https://doi.org/10.1016/j.baae.2005.07.002
Price PW, Pschorn-Walcher H (1988) Are galling insects better protected against parasitoids than exposed feeders?: a test using tenthredinid sawflies. Ecological Entomology 13:195–205. https://doi.org/10.1111/j.1365-2311.1988.tb00347.x
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.
Rezende UC, Cardoso JCF, Kuster VC et al (2019) How the activity of natural enemies changes the structure and metabolism of the nutritive tissue in galls? Evidence from the Palaeomystella oligophaga (Lepidoptera) -Macairea radula (Metastomataceae) system. Protoplasma 256:669–677. https://doi.org/10.1007/s00709-018-1321-2
Rezende UC, Pinheiro Moreira ASF, Coelho Kuster V, Coelho De Oliveira D (2018) Structural, histochemical and photosynthetic profiles of galls induced by Eugeniamyia dispar (Diptera: Cecidomyiidae) on the leaves of Eugenia uniflora (Myrtaceae). Rev Biol Trop 66:1469–1480. https://doi.org/10.15517/rbt.v66i4.32531
Rhomberg L (1980) Causes of life history differences between the morphs of Pemphigus populitransversus. J. N.Y. Entomological Society 88:106–112
Rohfritsch O (1992) Patterns in gall development. In: Shorthouse JD, Rohfritsch O (eds) Biology of insect-induced galls. Oxford University Press, Oxford, pp 60–86
Rohfritsch O, Anthony M (1992) Strategies on gall induction by two groups of homopterans. In: Shorthouse JD, Rohfritsch O (eds) Biology of insect induced galls. Oxford University, Oxford, pp 102–117
Rohfritsch O, Shorthouse JD (1982) Insect galls, pp. 131–152. In: Kahl G and Schell JS (ed) Molecular biology of plant tumors. Academic press, pp 131–152
Roskam JC (1992) Evaluation of gall-inducing guild. pp. 34–50. In: Shorthouse JD, Rohfritsch O (eds) Biology of insect induced galls. Oxford University Press, Oxford, pp 34–50
Sass JE (1951) Botanical microtechnique. Iowa State College Press, Ames, Iowa
Shorthouse JD, Wool D, Raman A (2005) Gall-inducing insects - nature’s most sophisticated herbivores. Basic Appl Ecol 6:407–411. https://doi.org/10.1016/j.baae.2005.07.001
Stone GN, Schönrogge K (2003) The adaptive significance of insect gall morphology. Trends Ecol Evol 18:512–522. https://doi.org/10.1016/S0169-5347(03)00247-7
Tabadkani SM, Allahyari H, Farhoudi F et al (2012) Effect of male densities on sex ratio variations of the predatory gall midge, Aphidoletes aphidimyza (Diptera: Cecidomyiidae). Arthropods 1:94–100
Wool D, Burstein M (1991) A galling aphid with extra life-cycle complexity: population ecology and evolutionary considerations. Res Popul Ecol (kyoto) 33:307–322. https://doi.org/10.1007/BF02513556
Acknowledgements
We are grateful to Carolina A. Garcia and Maria V. Urso-Guimarães, who described the gall-inducing insect of this system, and to Wesley C. Silva for support with some experiments. We thank Universidade Federal de Uberlândia – UFU and Laboratório de Anatomia Vegetal e Interações – LADEVI in particular for the equipment that made possible the elaboration of this work.
Funding
This study was financed in part by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES, finance code 001) to the first author, and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG). The Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) granted a fellowship to D.C. Oliveira.
Author information
Authors and Affiliations
Contributions
Pedro Henrique Pereira Gonçalves collected and analyzed the material, searched the bibliography, and wrote the full text. Uiara Costa Rezende helped collect and analyze the material and write the text. Phabliny Martins Silva Bonfim analyzed the laboratory material and the statistical data. Valéria Cid Maia described the insect’s spatula and the overall features of the developmental stages of L. mataybae. Denis Coelho de Oliveira was responsible for the analysis of the anatomical features and the revision of the text throughout the writing process.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All authors consented to participate.
Consent for publication
All authors consented for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by: Peter Schausberger
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gonçalves, P.H.P., Rezende, U.C., Bomfim, P.M.S. et al. Sexually dimorphic galls induced on leaflets of Matayba guianensis (Sapindaceae): a rare phenomenon occurring in Diptera (Cecidomyiidae). Sci Nat 109, 10 (2022). https://doi.org/10.1007/s00114-021-01777-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-021-01777-3